Author: Biology Junction Team
OCR Document
Lab Safety Rules & Contract

Purpose Science is a hands-on laboratory class. You will be doing many laboratory activities, which require the use of hazardous chemicals. Safety in the science classroom is the #1 priority for students, teachers, and parents. To ensure a safe science classroom, a list of rules has been developed and provided to you in this student safety contract. These rules must be followed at all times. The contract are must be signed by both you and a parent or guardian before you can participate in the laboratory.
GENERAL RULES
1. Conduct yourself in a responsible manner at all times in the laboratory.
2. Follow all written and verbal instructions carefully. If you do not understand a direction or part of a procedure, ask the instructor before proceeding.
3. Never work alone. No student may work in the laboratory without an instructor present.
4. When first entering a science room, do not touch any equipment, chemicals, or other materials in the laboratory area until you are instructed to do so.
5. Do not eat food or drink beverages in the laboratory. Do not use laboratory glassware as containers for food or beverages.
6. Perform only those experiments authorized.
7. Be prepared for your work in the laboratory. Read all procedures thoroughly before starting the laboratory experiment.
8. Never fool around in the laboratory. Horseplay, practical jokes, and pranks are dangerous and prohibited.
9. Observe good housekeeping practices. Work areas should be kept clean and tidy at all times. Bring only your laboratory instructions, worksheets, and/or reports to the work area. Other materials (books, purses, backpacks, etc.) should be stored under your chairs or in the classroom area.
10. Keep aisles clear. Push your chair under the desk or table.
11. Know the locations and operating procedures of all safety equipment including the first aid kit, eyewash station, safety shower, fire extinguisher, and fire blanket. Know where the fire alarm and the exits are located.
12. Be alert and proceed with caution at all times in the laboratory. Notify the instructor immediately of any unsafe conditions you observe.
13. Dispose of all chemical waste properly. Never mix chemicals in sink drains. Sinks are to be used only for water and those solutions designated by the instructor. Solid chemicals, metals, matches, filter paper, and all other insoluble materials are to be disposed of in the proper waste containers, not in the sink.
14. Labels and equipment instructions must be read carefully before use. Set up and use prescribed apparatus as directed in the laboratory instructions or by your instructor.
15. Keep hands away from face, eyes, mouth, and body while using chemicals or preserved specimens. Wash your hands with soap and water after performing all experiments. Clean all work surfaces and apparatus at the end of the experiment. Return all equipment clean and in working order to the proper storage area.
16. Experiments must be personally monitored at all times. You will be assigned a laboratory station at which to work. Do not wander around the room, distract other students, or interfere with the laboratory experiments of others.
17. Students are never permitted in the science storage areas or preparation areas unless given permission by the instructor.
18. Know what to do if there is a fire drill during a laboratory period; containers must be closed, gas valves turned off, and any electrical equipment turned off.
19. Handle all living organisms used in a laboratory activity in a humane manner.
20. When using knives and other sharp instruments, always carry with the tips and points pointing down and away. Always cut away from your body. Never try to catch falling sharp instruments. Grasp sharp instruments only by the handles.
21. If you have a medical condition (e.g., allergies, pregnancy, etc.), check with your physician prior to working in lab.
CLOTHING
22. Any time chemicals, heat, or glassware are used, students will wear laboratory goggles. There will be no exception to this rule.
23. Dress properly during a laboratory activity. Long hair, dangling jewelry, and loose or baggy clothing are a hazard in the laboratory. Long hair must be tied back and dangling jewelry and loose or baggy clothing must be secured.
24. Lab aprons have been provided for your use and should be worn during lab activities to prevent stains on your clothing.
ACIDENTS AND INJURIES
25. Report any accident (spill, breakage, etc.) or injury (cut, burn, etc.) to the instructor immediately, not matter how trivial it may appear.
26. If you or your lab partner are hurt, immediately yell out “Code one, Code one” to get the instructor’s attention.
27. If chemical splashes in your eye(s) or on your skin, immediately flush with running water from the eyewash station or safety shower for at least 20 minutes. Notify the instructor immediately.
28. When mercury thermometers are broken, mercury must not be touched. Notify the instructor immediately.
HANDLING CHEMICALS
29. All chemicals in the laboratory are to be considered dangerous. Do not touch, taste, or smell any chemicals unless specifically instructed to do so. The proper technique for smelling chemical fumes will be demonstrated to you.
30. Check the label on chemical bottles twice before removing any of the contents. Take only as much chemical as you need.
31. Never return unused chemicals to their original containers.
32. Never use mouth suction to fill a pipet. Use a rubber bulb or pipet pump.
33. When transferring reagents from one container to another, hold the containers away from your body.
34. Acids must be handled with extreme care. You will be shown the proper method for diluting strong acids. Always add acid to water, swirl or stir the solution and be careful of the heat produced, particularly with sulfuric acid.
35. Handle flammable hazardous liquids over a pan to contain spills. Never dispense flammable liquids anywhere near an open flame or source of heat.
36. Never remove chemicals or other materials from the laboratory area.
37. Take great care when transporting acids and other chemicals from one part of the laboratory to another. Hold them securely and walk carefully.
HANDLING GLASSWARE AND EQUIPMENT
38. Carry glass tubing, especially long pieces, in t vertical position to minimize the likelihood of breakage and injury.
39. Never handle broken glass with your bare hands. Use a brush and dustpan to clean up broken glass. Place broken or waste glassware in the designated glass disposal container.
40. Inserting and removing lass tubing from rubber stoppers can be dangerous. Always lubricate glassware (tubing, thistle tubes, thermometers, etc.) before attempting to insert it in a stopper. Always protect your hands with towels or cotton gloves when inserting glass tubing into, or removing it from, a rubber stopper. If a piece of glassware becomes “frozen” in a stopper, take it to your instructor for removal.
41. Examine glassware before each use. Never use chipped or cracked glassware. Never use dirty glassware.
42. Do not immerse hot glassware in cold water; it may shatter.
43. When removing an electrical plug from its socket, grasp the plug, not the electrical cord. Hands must be completely dry before touching an electrical switch, plug, or outlet.
44. Report damaged electrical equipment immediately. Look for things such as frayed cords, exposed wires, and loose connections. Do not use damaged electrical equipment.
45. If you do not understand how to use a piece of equipment, ask the instructor for help.
HEATING SUBSTANCES
46. Exercise extreme caution when using a gas burner. Take care that hair, clothing, and hands are a safe distance from the flame at all times. Do not put any substance into the flame unless specifically instructed to do so. Never reach over an exposed flame. Light gas burners only as instructed by the teacher.
47. Never leave a lit burner unattended. Never leave anything that is being heated or is visibly reacting unattended. Always turn the burner or hot plate off when not in use.
48. You will be instructed in the proper method of heating and boiling liquids in test tubes. Do not point the open end of a test tube being heated at yourself or anyone else.
49. Heated metals and lass remain very hot for a long time. They should be set aside to cool and picked up with caution. Use tongs or heat-protective gloves if necessary.
50. Never look into a container that is being heated.
51. Do not place hot apparatus directly on the laboratory desk. Always use an insulating pad. Allow plenty of time for hot apparatus to cool before touching it.
52. When bending glass, allow time for the glass to cool before further handling. Hot and cold glass has the same visual appearance. Determine if an object is hot by bringing the back of your hand close to it prior to grasping it.
QUESTIONS
53. Do you wear contact lenses?
□ YES □ NO
54. Are you color blind?
□ YES □ NO
55. Do you have allergies?
□ YES □ NO
If so, list specific allergies:
____________________________________________________________________________________________________________________________________________________________________
Web Page Permission
Our science classes have a web page on the internet found at: http://sps.k12.ar.us/massengale
We often include pictures of students doing classroom activities. In order for a student’s picture to appear on the web page, both the student and parent/guardian, must sign the form below.
STUDENT
I, ____________________________________ (print student name) give my permission to have my picture displayed on the class web page.
____________________________________________
Student Signature/Date
PARENT OR GUARDIAN
___________________________________________
Parent/Guardian Signature & Date
Safety Contract Agreement STUDENT
I, ___________________________________
(Student’s name) has read and agrees to follow all of the safety rules set forth in this contract. I realize that I must obey these rules to insure my own safety, and that of my fellow students and instructors. I will cooperate to the fullest extent with my instructor and fellow students to maintain a safe lab environment. I will also closely follow the oral and written instructions provided by the instructor. I am aware that any violation of this safety contract that results in unsafe conduct in the laboratory or misbehavior on my part, may result in being removed from the laboratory, detention, and/or receiving a failing grade.
_______________________________________________
Student signature/Date
_______________________________________________
Date
PARENT OR GUARDIAN
Dear Parent or Guardian:
We feel that you should be informed regarding the school’s effort to create and maintain a safe science classroom/laboratory environment. With the cooperation of the instructors, parents, and students, a safety instruction program can eliminate, prevent, and correct possible hazards. You should be aware of the safety instructions your son/daughter will receive before engaging in any laboratory work. Please read the list of safety rules above. No student will be permitted to perform laboratory activities unless this contract is signed by both the student and parent/guardian and is on file with the teacher. Your signature on this contract indicates that you have read this Student Safety Contract, are aware of the measures taken to insure the safety of your son/daughter in the science laboratory, and will instruct your son/daughter to uphold his/her agreement to follow these rules and procedure in the laboratory.
_______________________________________________
Parent/Guardian Signature & Date
Sponge Coloring Diagram and Questions
Found at the Biology Corner Name __________________ Period ______
 Sponges – A Coloring Worksheet
Sponges – A Coloring Worksheet
Since sponges look like plants, it is understandable why early biologists thought they were plants. Today, we know that sponges are simple, multicellular animals in the Kingdom Animalia, Phylum Porifera. This phylum is thought to represent the transition from unicellular animals to multicellular animals. Most (but not all) sponges are asymmetrical and have no definite shape. Sponges, like all animals, are eukaryotic – meaning their cells have a nucleus. Porifera in Latin means “pore-bearer” and refers to the many pores or openings in these animals. Because of these pores, a sponge can soak up and release water. At one time, real sponges were used for cleaning and bathing. Today, most are artificially made.
All adult sponges are sessile, meaning they are attached to some surface. Since they cannot move, sponges cannot pursue their food. Instead, they are filter feeders, meaning they obtain their food by straining the water for small bits of food like bacteria, algae or protozoans.
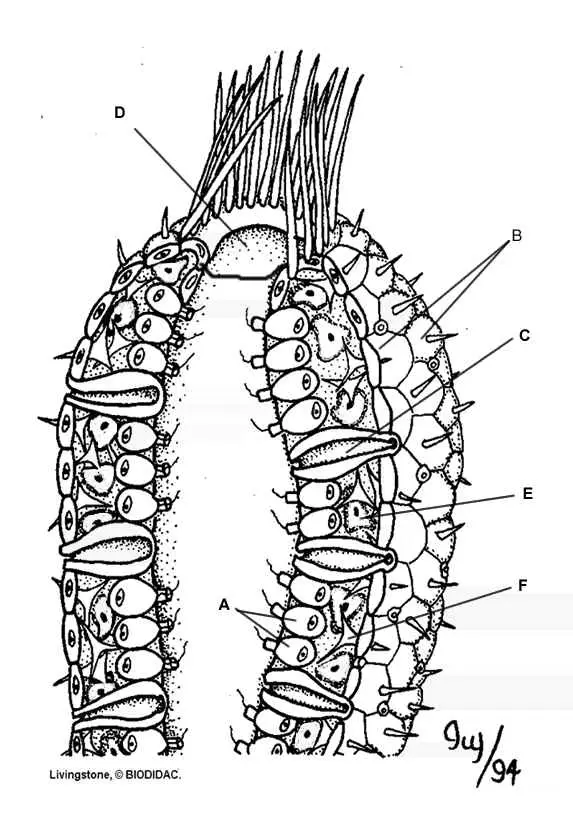
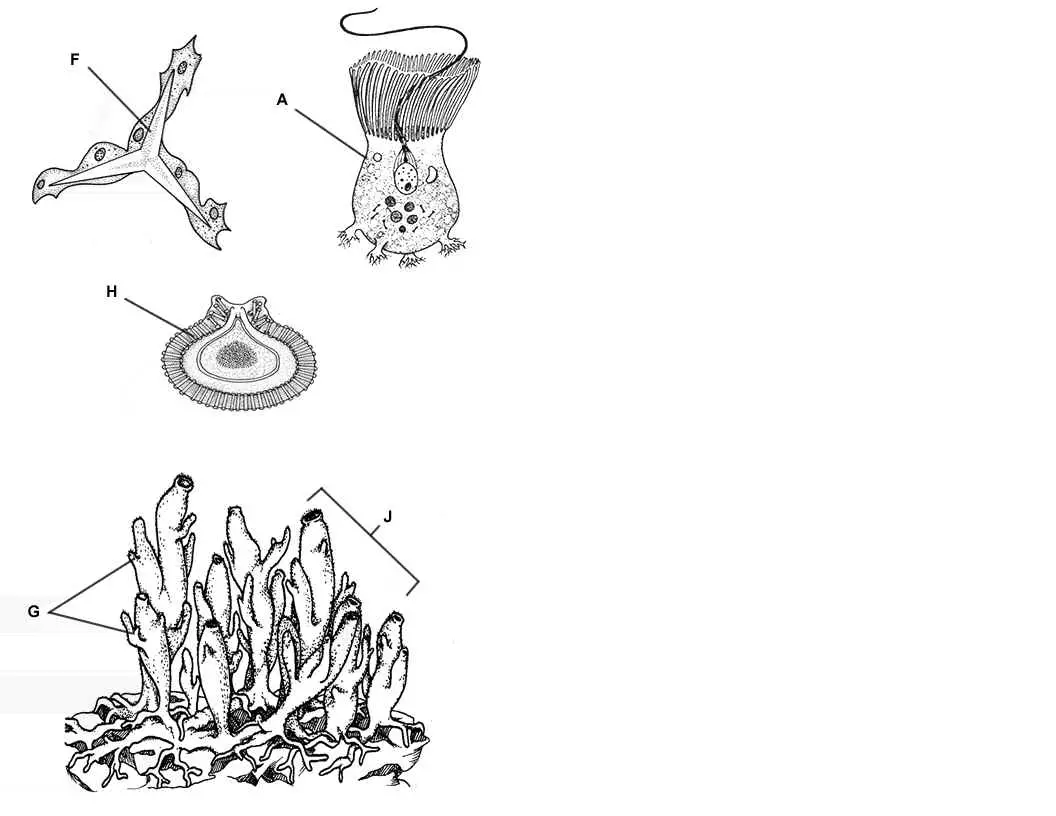
Sponges exhibit less specialization (adaptation of a cell for a particular function) of cells than most invertebrates. The primitive structure of a sponge consists of only two layers of cells separated by a non-living jelly like substance. The outer layer of the sponge is the epidermis which is made of flat cells called epithelial cells. Color all the epithelial cells (B) of the epidermis peach or pink.
The inner layer consists of collar cells (A) whose function is to circulate water through the sponge. They do this by swishing their flagella which pulls water through the incurrent pore – water then travels out the osculum at the top of the sponge. As water passes through the sponge in this way, cells absorb food and oxygen and waste is excreted. Color the osculum (D) dark blue, the incurrent pores (C) light blue. Color the inside of the sponge where water circulates the same light blue as you colored the incurrent pores. Color all the collar cells (A) red.
In the jelly-like substance between the epidermis and the collar cells are cells called amebocytes – because they look like amebas. The job of the amebocytes is to travel around distributing food and oxygen to the cells of the epidermis. Because of the amebocytes, scientists believe that sponges evolved from protists. Color all of the amebocytes (E) green – look for them carefully.
The body of the sponge would collapse if it did not have some type of supporting structure. Some sponges have a soft network of protein fibers called spongin. Others have tiny, hard particles called spicules. Many of these spicules also stick out of the epidermis and provide the sponge with protection. Most sponges have a combination of spicules and spongin, the ratio often determines how soft or hard the sponge is. Search for and color all the pointy spicules (F) brown.
Reproduction for sponges can be accomplished both sexually and asexually. There are three ways for a sponge to reproduce asexually: budding, gemmules, and regeneration. Sponges can simply reproduce by budding, where a new sponge grows from older ones and eventually break off. Color the adult sponge (J) pink and all the buds (G) you can find red. Sponges can also reproduce by regeneration, where missing body parts are regrown. People who harvest sponges often take advantage of this by breaking off pieces of their catch and throwing them back in the water, to be harvested later. Finally, sponges can reproduce by creating gemmules – which is a group of amebocytes covered by a hard outer covering. Color the gemmule (H) yellow.
Sexual reproduction occurs when one sponge releases sperm into the water. This sperm travels to another sponge and fertilizes its eggs. The larva form will then swim to another location using its flagella where it will grow into an adult sponge. Most sponge species are hermaphrodites, they can produce both eggs and sperm.
Questions:
1. What did early biologists think sponges were? ______________________
2. Sponges belong to the Kingdom _________________ and the Phylum _______________
3. Sponges are [ unicellular or multicelluar ] and [ prokaryotic or eukaryotic ]
4. What type of symmetry do sponges have? ___________________________________
5. What does it mean to be sessile? ____________________________________
6. How do sponges get their food? ___________________________________
7. Water enters the sponge through the _____________________ and leaves through the
_____________________.
8. What is the job of the amebocyte? ________________________________________
9. What two substances give the sponge support? _________________________________
10. Tiny sponges growing from the main body of the sponge are called _________________
11. What is a gemmule? ___________________________________________________
12. What is a hermaphrodite? ______________________________________________
Label the letters on the diagrams
Label the letters on the diagrams
Found at www.biologycorner.com
2006 1st Semester Test Guide
First Semester Test 2006 Study Guide
1. What is the study of life called?
2. Instructions for traits passed from parent to offspring?
3. Keeping things stable or the same in cells?
4. Smallest units that can carry on life?
5. All living things require _________ for metabolism.
6. All living things are made of __________.
7. Salamanders with curved tails in polluted water are an example of which part of the scientific method?
8. The smallest part of carbon with all the same properties is called?
9. Where are electrons found in an atom & what is their charge?
10. When electrons gain energy they move to _____________________.
11. How many covalent bonds can carbon form?
12. Compounds may form from the transfer or __________ of electrons.
13. What happens to ionic compounds when placed in water?
14. Ionic bonds form from the ____________ of electrons.
15. Covalent bonds form from the ___________ of electrons.
16. What element do all organic compounds contain?
17. Give several examples of carbohydrates.
18. In what from do animals store glucose?
19. What are the monomers for proteins?
20. Is the following model a carbohydrate, lipid, or protein?

21. Sketch a fatty acid chain found in lipids.
22. What type of fatty acids contains double bonds?
23. Name the 2 nucleic acids.
24. When the volume of a cell increases, what happens to the surface area?
25. How does a prokaryotic cell differ from a eukaryotic cell?
26. What’s the job of the plasma membrane?
27. Parts of cells performing specific functions are called?
28. The ER sends proteins & lipids it makes to the __________ to be modified.
29. The Golgi ships & receives cell products in transport ___________.
30. In what organelle is ATP produced?
31. Ribosomes make ____________.
32. All eukaryotic cells have a ___________ containing the genetic material.
33. Diffusion occurs in what direction?
34. What is osmosis?
35. Ink dissolving in water is an example of _____________.
36. Riding of cell wastes in sacs is called _____________.
37. What is the effect of placing a plant into a hypertonic solution?
38. What is the ultimate energy for life on Earth?
39. Grana are suspended in the _________ of chloroplasts.
40. ____________ absorbs light energy for plants.
41. What happens to chlorophyll’s electrons when they absorb sunlight?
42. What is the source of oxygen in photosynthesis?
43. What gas is a byproduct of photosynthesis?
44. What type of skeleton do insects have?
45. The Calvin cycle occurs in what process?
46. The breaking down of food to release energy is called?
47. __________ builds up in heavily exercised muscles.
48. Name the 3 parts of cellular respiration.
49. How many chromosomes are in a human egg or sperm cell?
50. DNA compacts itself by wrapping around ____________.
51. How do insects help crops?
Translation Animation
RNA Translation: Protein Synthesis
You need to go to www.macromedia.com to download the correct Shockwave plug-in to view this animation.
| Key | |||||||||
|---|---|---|---|---|---|---|---|---|---|
| C | G | A | T |  |
U | ||||
|
|
tRNA with attatched amino acid |
||||||||

