| Protozoa Animal like Protists All Materials © Cmassengale |
 |
Characteristics:
- Eukaryotes
- Found in kingdom Protista
- Most are unicellular
- Heterotrophs that ingest small food particles & digest it inside food vacuoles containing digestive enzymes
- Classified by the way they move (cilia, flagella, pseudopodia…)

- Microscopic in size
- 65,000 identified species with almost half extinct
- Found in freshwater, marine, and moist terrestrial habitats
- Make up part of the zooplankton & serve as food for animals in marine & freshwater systems
- First seen by Leeuwenhoek in 1675
- Many species are free living
- Some species are parasitic living in the bloodstream of their host & cause malaria, amebic dysentery, or giardiasis
- Many serve as food for other organisms in aquatic habitats; called zooplankton
Reproduction:
- All reproduce asexually by binary fission (single protozoan divides into two individuals)
- Some species reproduce by multiple fission producing more than two individuals
- Some species reproduce sexually by conjugation (opposite mating strains join & exchange genetic material)
Adaptations:
- Eyespots in some protozoans can detect changes in light

- Many can form harden covering called cyst when conditions become unfavorable (no water, pH or temperature changes, nutrient deficiency, decreased oxygen supplies…)
- Metabolic activity of protozoans resumes when conditions become favorable again
- Some protozoans can detect & avoid obstacles and harmful chemicals in their environment
- Freshwater protozoa have contractile vacuoles to pump out excess water
Classification:
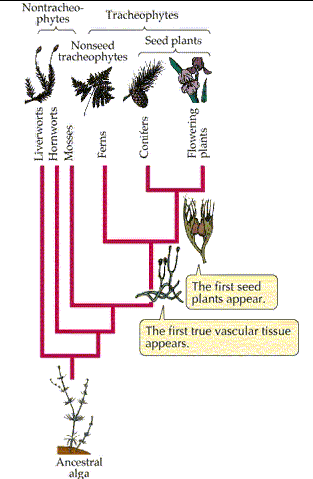
- Divided into 4 phyla based on their method of movement — Sarcodina, Ciliophora, Zoomastigina, & Sporozoa
- Found in the kingdom Protista along with algae, slime molds, & water molds
- Sarcodinians move by extending their cytoplasm or pseudopodia (fingerlike projections of the cytoplasm)
- Zooflagellates move by whip like flagella
- Ciliophorans or ciliates move by hair like cilia move
- Sporozoans are nonmotile
| Phylum | Common Name | Locomotion | Type of Nutrition | Examples |
| Sarcodina | sarcodines | pseudopodia | heterotrophic; some parasitic |
Amoeba Radiolaria Naegleria |
| Ciliophora | ciliates | cilia | heterotrophic; some parasitic |
Paramecium Tetrahymena Balantidium |
| Zoomastigina | zooflagellates | flagella | heterotrophic; some parasitic |
Trypanosoma Leishmania Giardia Trichonympha |
| Sporozoa | sporozoans | (None in Adults) | heterotrophic; some parasitic |
Plasmodium Toxoplasma |
Protozoan Evolution:
- First eukaryotic organism thought to have evolved about 1.5 billion years ago
- Protozoans possible evolved from the 1st eukaryotes by Endosymbiosis
- Endosymbiosis – process where one prokaryote lives inside another becoming dependent upon each other
Phylum Sarcodina:
- Includes hundreds of species of amebas
- Found in freshwater, marine, & moist soil habitats
- Usually reproduce asexually
- Their cytoplasm consists of clear, outer ectoplasm and granular, inner endoplasm
- Move by extending cytoplasm (cytoplasmic streaming)
- Cytoplasm extensions are called “false foot” or pseudopods
- Pseudopods form when the inner cytoplasm or endoplasm pushes the outer cytoplasm or ectoplasm forward to make a blunt, armlike extension
- Ameba move by cytoplasmic streaming to produce pseudopods; process called ameboid movement

- Sarcodines also use their pseudopods for feeding by surrounding & engulfing food particles & other protists; called phagocytosis
- Food is surrounded by a pseudopod & then this part of the cell membrane pinches together forming a food vacuole; called endocytosis
- Cytoplasmic enzymes enter the food vacuole & digest the food
- Undigested food & wastes leave by exocytosis

- Most Sarcodinians have contractile vacuoles to pump out excess water

- Oxygen & carbon dioxide diffuse through the cell membrane
- Sarcodinians may form hard, protective, inactive cysts when conditions become unfavorable (drought, lack of nutrients, heat…)
- React to stimuli such as light
- Some Sarcodinians have hard shells called the test made of silica or calcium carbonate
- Radiolarians found in warm, marine waters have a test made of silica & have sticky pseudopodia to trap food

- Marine Foraminiferans have a test made of calcium carbonate with holes through which pseudopodia extend

- Foraminiferan tests build up and form limestone or chalk (e.g. White Cliffs of Dover)
- Important food source in marine habitats
- Entameba histolytica cysts in untreated water supplies cause amebic dysentery which can be fatal

Phylum Ciliophora:
- Called ciliates because they move by short, hairlike cilia lining the cell membrane
- Cilia may be modified into teeth, paddles, or feet

- Largest group of protozoans
- Most found in freshwater, but some are marine
- Called plankton & serve as a food source
- Form protective cysts to survive unfavorable conditions
- Members include the Paramecium, Vorticella, & Stentor
- Have 2 types of nuclei — smaller micronuclei & larger macronuclei
- Macronucleus controls asexual reproduction by mitosis
- Can reproduce sexually by conjugation (two paramecia join together & exchange DNA)
- Gases diffuse across cell membrane
Stentor:
- Trumpet shaped protozoan with cilia around the top
- Attaches to feed & then detaches to swim around

Vorticella:
- Cup shaped protozoan with cilia at the top
- Has a coiled stalk to raise & lower the organism
- Can attach to surfaces

Paramecium caudatum:
- Slipper shaped protozoan found in freshwater

- Clear, elastic covering of cell membrane called pellicle
- Pellicle made of protein for protection
- Use cilia to swim & obtain food (algae & bacteria)
- Have 2 contractile vacuoles to pump out excess water
- Cilia sweep food into oral groove where mouth located at the bottom
- Food enters short tube called gullet into food vacuoles where it’s digested
- Wastes leave through anal pore

- Have trichocysts (tiny, toxic darts to help capture prey or anchor to a surface)
- Respond to light & learn by trial & error
- Reproduce asexually by mitosis & sexually by conjugation

Phylum Zoomastigina:
- Called Zooflagellates because have one or more whiplike flagella to move
- Flagella made of bundles of microtubules

- May be freshwater or marine
- Some are parasites such as Trypanosoma that destroy red blood cells & causes fatal African sleeping sickness

- Trichonympha lives symbiotically inside termites & digests cellulose

Phylum Sporozoa:
- Adult sporozoans have no structures for movement
- Form spores

- Most are parasitic using one or more hosts
- Immature sporozoans are called sporozoites & live in body fluids of hosts
- Plasmodium is transmitted by mosquitoes & causes malaria
- Plasmodium sporozoites enter the bloodstream, travel to the liver, divide & form spores called merozoites
- Merozoites attack red blood cells & later form eggs & sperm that fertilize
- New sporozoites migrate to the salivary glands of mosquitoes where they can be passed on to another person
- Malaria can be controlled by controlling mosquito populations & it is treated with a drug called quinine derived from the Cinchona Tree

























.gif)