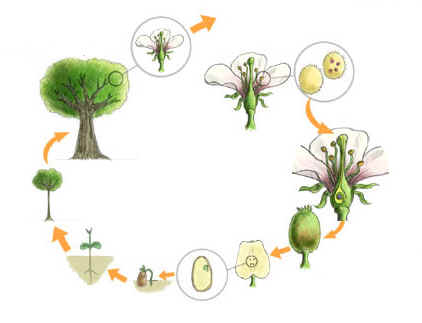
Arkansas is essentially a forest state because more than half of the state is covered with trees. The climate and soils of Arkansas also support a great variety of trees, both conifers and deciduous. Trees are one of Arkansas’ most important crops. Forests are also valuable in preventing erosion, in offering parks and recreational areas, and in providing homes for wildlife. In addition, many trees have been introduced into the state as ornamentals.
Leaf collecting is a good way to learn the trees native to your area. Collecting leaves will also help you to learn leaf margins, shapes, and venations and how to use different taxonomic keys to identify trees.

Materials needed:
- leaf press
- black ink pen
- pencil
- small notebook
- scissors
- Elmer’s glue
- art paper, poster board, etc. for mounting
- labels
- taxonomic keys (Trees of Arkansas published by the Arkansas Forestry Commission)
Directions for making a leaf press:
1. Cut 15 – 20 pieces of corrugated cardboard 30 cm by 50 cm in size.
2. Cut several sheets of newspaper the same size as the cardboard.
3. Lay 10 or 12 sheets of newspaper between each cardboard layer sandwich style. These sheets will need to be changed every couple of days as they absorb moisture from your leaves; therefore, cut extra sheets.
4. Use one, preferably two, stretch belts to bind the press together.
5. Leave the press in an area so that air can circulate & more quickly dry the leaves.
Getting started with your collection:
1. Study the shapes, margins, venations, tips, bases, etc. in your Trees of Arkansas book.
Click here to view reference page
2. Learn to distinguish simple leaves from compound leaves and conifers from deciduous trees.
3. Learn to distinguish a tree from a shrub.
4. Gather your collecting materials together – press, pencil, scissors, & small notebook.
6. Always get permission before collecting leaves on someone else’s property.
7. Be sure to collect at least two of each type of leaf so both the bottom & top side of the leaf can be shown in your collection.
8. Place leaves in your press immediately after collecting them so they do not start to dry out and wrinkle.
9. Record the name of each leaf, date collected, and place collected in your notebook as you collect. Also record tree characteristics such as shape of the crown, color and type of bark, etc.
Collecting:
1. Remember to collect two of every type of leaf!
2. Carefully remove an entire leaf, not a leaflet, from the tree, and place this in your press between newspaper layers.
3. If leaves are damaged or torn, don’t use them because you will not receive credit.
4. Make sure that none of the leaf parts extend beyond the edge of the press.
5. You may also collect &press seeds and/or fruits from some trees if they fit in your press.
6. Leave the leaf in the press for 3 – 5 days depending on its thickness and moisture content. Remember to change the newspaper when needed.
7. Keep the press in an area where air is circulating (in front of a fan).
Labeling and identifying:
1. Obtain printed labels from your teacher.
2. Use only black ink to write labels, & do not mark out or white out mistakes on the labels; rewrite them.
3. Use taxonomic keys to identify each leaf, and include both the scientific & common name of the tree on the label.
4. Determine the shape, margin, tip, base, and venation of your leaf and whether it is a simple or compound leaf; record this on your label.
5. Use you key to give a description of the tree, not the leaf.
6. Research uses for the tree, its fruit, etc. and record on your label.
7. Tell if the leaf is deciduous or coniferous.
Mounting leaves:
1. Use pieces of cut poster board or art paper to mount your leaves. Make sure all sheets are uniform in size! (The size of your sheets will be determined by your largest leaf.)
2. Use Elmer’s glue to adhere two leaves to each page — one showing the upper surface of the leaf and the other showing the underside of the leaf.
3. Each page should have only one type of leaf on it.
4. Arrange the leaves so they do not overlap each other and so there is room to glue the label in the lower right hand corner. The leaves should look nice on the page.
5. On compound leaves, mount the topside of the complete leaf and then mount the underside of a single leaflet. Make sure the leaflet comes from another leaf to receive credit!
6. Use a small amount of Elmer’s glue to adhere the completed label in the lower right hand corner of the page.
7. LET THE PAGES DRY COMPLETELY BEFORE ASSEMBLING THEM TOGETHER IN YOUR COLLECTION OR THE PAGES WILL STICK TOGETHER!!!!!
8. Once the pages are dry, lay them in the correct order (see your list of required leaves), and then number the pages in the lower right corner with black ink.
9. Make a stiff front and back cover for your collection from poster board, cardboard, wood, etc. Include the following items on your cover:
- title (Tree Identification Through Leaves)
- your complete name
- date collection turned into teacher
- class period
- subject
- teacher’s name
10. Use ribbon, string, etc. to bind the pages together or assemble the collection in a scrapbook. DO NOT COVER THE LEAVES WITH PLASTIC!!!
Required leaves:
1. Only native, Arkansas trees may be used. Refer to your Trees of Arkansas book.
2. Leaves must be in perfect condition without damage or tears.
3. No more then 4 oaks are allowed in the collection.
4. No fruit trees such as apple, pear, orange, peach, etc. are allowed.
5. Place the following leaves in your collection first and in this order:
- sweet gum
- American sycamore
- pine (any type)
- flowering dogwood
- redbud
- ash (any type)
- persimmon
- Eastern red cedar
- red or silver maple
- hickory (any type)
- pecan
- pin oak
- willow oak
- water oak
- elm (any type)
6. The remaining leaves that you include must be trees native to Arkansas!
*Pre AP Biology is required to collect 30 leaves including the 15 required.
*Biology I is required to collect 20 leaves including the 15 required.