STRUCTURE AND FUNCTION OF THE CELL

All Materials © Cmassengale
I. All Organisms are Made of Cells
A. The cell is the basic unit of structure & function
B. The cell is the smallest unit that can still carry on all life processes
C. Both unicellular (one celled) and multicellular (many celled) organisms are composed of cells
D. Before the 17th century, no one knew cells existed
E. Most cells are too small to be seen with the unaided eye
F. In the early 17th century microscopes were invented & cells were seen for the 1st time
G. Anton Von Leeuwenhoek, a Dutchman, made the 1st hand-held microscope & viewed microscopic organisms in water & bacteria from his teeth
| Leeuwenhoek’s microscope consisted simply of:

- A) a screw for adjusting the height of the object being examined
- B) a metal plate serving as the body
- C) a skewer to impale the object and rotate it
- D) the lens itself, which was spherical
|
H. In 1665, an English scientist named Robert Hooke made an improved microscope and viewed thin slices of cork viewing plant cell walls
I. Hooke named what he saw “cells”

J. In the 1830’s, Matthias Schleiden (botanist studying plants) & Theodore Schwann (zoologist studying animals) stated that all living things were made of cells
K. In 1855, Rudolf Virchow stated that cells only arise from pre-existing cells
L. Virchow’s idea contradicted the idea of spontaneous generation (idea that nonliving things could give rise to organisms)
M. The combined work of Schleiden, Schwann, & Virchow is known as the Cell Theory
 |
 |
 |
| Schwann |
Schleiden |
Virchow |
II. Principles of the Cell Theory
A. All living things are made of one or more cells
B. Cells are the basic unit of structure & function in organisms
C. Cells come only from the reproduction of existing cells
III. Cell Diversity
A. Not all cells are alike
B. Cells differ in size, shape, and function
C. The female egg cell is the largest cell in the body & can be seen without a microscope

D. Bacterial cells are some of the smallest cells & are only visible with a microscope

E.coli Bacterial Cells
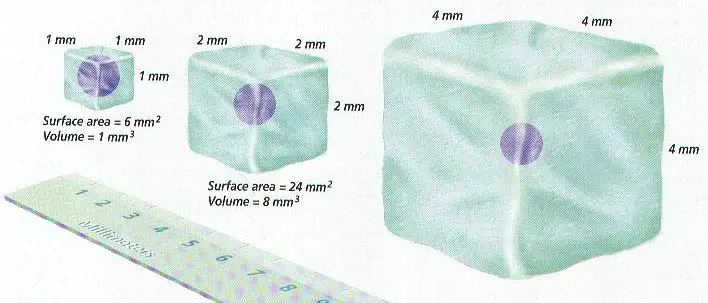
E. Cells need surface area of their cell membrane large enough to adequately exchange materials with the environment (wastes, gases such as O2 & CO2, and nutrients)
F. Cells are limited in size by the ratio between their outer surface area & their volume
G. Small cells have more surface area for their volume of cytoplasm than large cells
H. As cells grow, the amount of surface area becomes too small to allow materials to enter & leave the cell quickly enough
I. Cell size is also limited by the amount of cytoplasmic activity that the cell’s nucleus can control
J. Cells come in a variety of shapes, & the shape helps determine the function of the cell (e.g. Nerve cells are long to transmit messages in the body, while red blood cells are disk shaped to move through blood vessels)

IV. Prokaryotes
A. Prokaryotic cells are less complex
B. Unicellular
C. Do not have a nucleus & no membrane-bound organelles
D. Most have a cell wall surrounding the cell membrane & a single, looped chromosome (genetic material) in the cytoplasm
E. Include bacteria & blue-green bacteria
F. Found in the kingdom Monera

V. Eukaryotes
A. More complex cells
B. Includes both unicellular & multicellular organisms
C. Do have a true nucleus & membrane-bound organelles
D. Organelles are internal structures in cell’s that perform specific functions
|

|
| a. Nucleus |
b. Chloroplast |
c. Golgi |
d. Mitochondria |
E. Organelles are surrounded by a single or double membrane
F. Entire eukaryotic cell surrounded by a thin cell membrane that controls what enters & leaves the cell
G. Nucleus is located in the center of the cell
H. The nucleus contains the genetic material (DNA) & controls the cell’s activities
I. Eukaryotes include plant cells, animal cells, fungi, algae, & protists
J. Prokaryotes or bacteria lack a nucleus

K. Found in the kingdoms Protista, Fungi, Plantae, & Animalia

VI. Cell Membrane
A. Separates the cytoplasm of the cell from its environment
B. Protects the cell & controls what enters and leaves
C. Cell membranes are selectively permeable only allowing certain materials to enter or leave
D. Composed of a lipid bilayer made of phospholipid molecules

E. The hydrophilic head of a phospholipid is polar & composed of a glycerol & phosphate group and points to the aqueous cytoplasm and external environment.
F. The two hydrophobic tails are nonpolar point toward each other in the center of the membrane & are composed of two fatty acids

G. When phospholipids are placed in water, they line up on the water’s surface with their heads sticking into the water & their tails pointing upward from the surface.
H. The inside of the cell or cytoplasm is an aqueous or watery environment & so is the outside of the cell. Phospholipid “heads” point toward the water.

I. Phospholipid “tails” are sandwiched inside the lipid bilayer.

J. The cell membrane is constantly breaking down & being reformed inside living cells.
K. Certain small molecules such as CO2, H2O, & O2 can easily pass through the phospholipids
VII. Membrane Proteins
A. A variety of protein molecules are embedded in the cell’s lipid bilayer.

B. Some proteins called peripheral proteins are attached to the external & internal surface of the cell membrane
C. Integral proteins or transmembrane proteins are embedded & extend across the entire cell membrane. These are exposed to both the inside of the cell & the exterior environment.
D. Other integral proteins extend only to the inside or only to the exterior surface.
E. Cell membrane proteins help move materials into & out of the cell.
F. Some integral proteins called channel proteins have holes or pores through them so certain substances can cross the cell membrane.
G. Channel proteins help move ions (charged particles) such as Na+, Ca+, & K+ across the cell membrane
H. Transmembrane proteins bind to a substance on one side of the membrane & carry it to the other side. e.g. glucose
I. Some embedded, integral proteins have carbohydrate chains attached to them to serve as chemical signals to help cells recognize each other or for hormones or viruses to attach

VIII. Fluid Mosaic Model
A. The phospholipids & proteins in a cell membrane can drift or move side to side making the membrane appear “fluid”.
B. The proteins embedded in the cell membrane form patterns or mosaics.

C. Because the membrane is fluid with a pattern or mosaic of proteins, the modern view of the cell membrane is called the fluid mosaic model.
IX. Internal Cell Structure & Organelles of Eukaryotes
A. Cytoplasm includes everything between the nucleus and cell membrane.
B. Cytoplasm is composed of organelles & cytosol (jellylike material consisting of mainly water along with proteins.
C. Eukaryotes have membrane-bound organelles; prokaryotes do not

D. Mitochondria are large organelles with double membranes where cellular respiration (breaking down glucose to get energy) occurs
1. Energy from glucose is used to make ATP or adenosine triphosphate
2. Cells use the ATP molecule for energy
3. More active cells like muscle cells have more mitochondria
4. Outer membrane is smooth, while inner membrane has long folds called cristae
5. Have their own DNA to make more mitochondria when needed

E. Ribosomes are not surrounded by a membrane & are where proteins are made in the cytoplasm (protein synthesis)
1. Most numerous organelle
2. May be free in the cytoplasm or attached to the rough ER (endoplasmic reticulum)
F. Endoplasmic reticulum are membranous tubules & sacs that transport molecules from one part of the cell to another

1. Rough ER has embedded ribosomes on its surfaces for making proteins

2. Smooth ER lacks ribosomes & helps break down poisons, wastes, & other toxic chemicals
3. Smooth ER also helps process carbohydrates & lipids (fats)
4. The ER network connects the nucleus with the cell membrane
G. Golgi Apparatus modifies, packages, & helps secrete cell products such as proteins and hormones
1. Consists of a stack of flattened sacs called cisternae
2. Receives products made by the ER

H. Lysosomes are small organelles containing hydrolytic enzymes to digest materials for the cell
1. Single membrane
2. Formed from the ends of Golgi that pinch off
3. Found in most cells except plant cells
I. Cytoskeleton consists of a network of long protein tubes & strands in the cytoplasm to give cells shape and helps move organelles
1. Composed of 2 protein structures — microtubules, intermediate filaments, & microfilaments
2. Microfilaments are ropelike structures made of 2 twisted strands of the protein actin capable of contracting to cause cellular movement (muscle cells have many microfilaments)
3. Microtubules are larger, hollow tubules of the protein called tubulin that maintain cell shape, serve as tracks for organelle movement, & help cells divide by forming spindle fibers that separate chromosome pairs
| Cytoskeleton Element |
General Function |
| Microtubules |
Move materials within the cell
Move the cilia and flagella |
| Actin Filaments |
Move the cell |
| Intermediate Filaments |
Provides mechanical support |
J. Cilia are short, more numerous hair like structures made of bundles of microtubules to help cells move
1. Line respiratory tract to remove dust & move paramecia

Cross section of Cilia & Flagella
K. Flagella are long whip like tails of microtubules bundles used for movement (usually 1-3 in number)
1. Help sperm cells swim to egg
L. Nucleus (nuclei) in the middle of the cell contains DNA (hereditary material of the cell) & acts as the control center
1. Most cells have 1 nucleolus, but some have several
2. Has a protein skeleton to keep its shape
3. Surrounded by a double layer called the nuclear envelope containing pores
4. Chromatin is the long strand of DNA in the nucleus, which coils during cell division to make chromosomes
5. Nucleolus (nucleoli) inside the nucleus makes ribosomes & disappears during cell division

M. Cell walls are nonliving, protective layers around the cell membrane in plants, bacteria, & fungi

1. Fungal cell walls are made of chitin, while plant cell walls are made of cellulose
2. Consist of a primary cell wall made first and a woody secondary cell wall in some plants
N. Vacuoles are the largest organelle in plants taking up most of the space
1. Serves as a storage area for proteins, ions, wastes, and cell products such as glucose
2. May contain poisons to keep animals from eating them
3. Animal vacuoles are smaller & used for digestion
O. Plastids in plants make or store food & contain pigments to trap sunlight
1. Chloroplast is a plastid that captures sunlight to make O2 and glucose during photosynthesis; contains chlorophyll
a. Double membrane organelle with an inner system of membranous sacs called thylakoids
b. Thylakoids made of stacks of grana containing chlorophyll
2. Other plastids contain red, orange, and yellow pigments

3. Found in plants, algae, & seaweed
X. Multicellular Organization
A. Cells are specialized to perform one or a few functions in multicellular organisms
B. Cells in multicellular organisms depend on each other
C. The levels of organization include:
Cells –> Tissues –> Organs –> Systems –> Organism

D. Tissues are groups of cells that performs a particular function (e.g. Muscle)
E. Organs are groups of tissues working together to do a job (e.g. heart, lungs, kidneys, brain)
F. Systems are made of several organs working together to carry out a life process (e.g. Respiratory system for breathing)
G. Plants have specialized tissues & organs different from animals
1. Dermal tissue forms the outer covering of plants
2. Ground tissue makes up roots & stems
3. Vascular tissue transports food & water
4. The four plant organs are the root, stem, leaf, & flower
H. Colonial organisms are made of cells living closely together in a connected group but without tissues & organs (e.g. Volvox)







































