| Grasshopper Dissection |  |
Introduction:
Insects are arthropods with jointed appendages, segmented bodies, and an exoskeleton composed of chitin. Insects are in the class Insecta, & are the largest and most diverse group of animals on earth. The genus Romalea is a large grasshopper common in the southeastern United States. Insects have three body regions (head, thorax, & abdomen), 3 pairs of legs attached to the thorax, a single pair of antenna attached to the head, mouthparts adapted for chewing or sucking, and two pairs of wings. Some insects may have a single pair of wings or be wingless. Insect legs are often adapted for digging, crawling, jumping, or swimming. The insects are mostly terrestrial, they breathe air which enters small lateral openings on the body called spiracles and circulates in a system of ducts to all organs and tissues. Their chewing or sucking mouth parts are adapted for feeding on plant or animal materials.
Classification:
Kingdom – Animalia
Phylum – Arthropoda
Class – Insecta
Order – Orthoptera
Objective:
Identify & label the internal & external anatomy of a grasshopper.
Materials:
Lab apron, gloves, eyeglasses, dissecting pan, dissecting kit with forceps & scalpel, t-pins, magnifying glass, preserved grasshopper, paper, pencil.
Procedure (External Anatomy): Examine the entire grasshopper and identify the major subdivisions and parts of the body.
- Obtain a preserved grasshopper & rinse off any preservative with water. Place grasshopper in the dissecting pan.
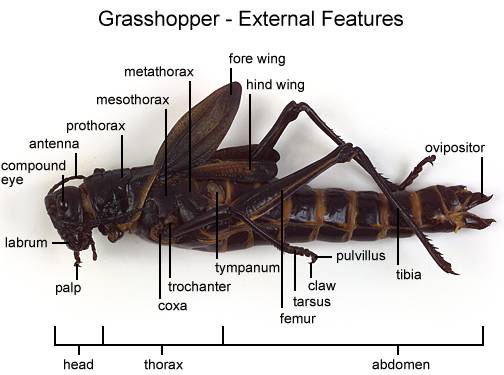
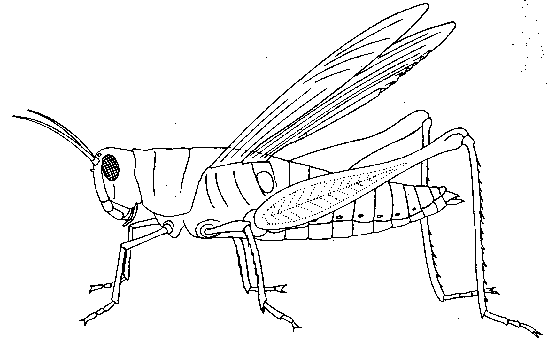
- Observe that the body of the grasshopper is divided into 3 regions — the head, the thorax, and abdomen. Label these on Figure 2.
- Examine the head and locate the following parts:
HEAD
Antennae (two, slender appendages)
Compound eyes (2, large lateral)
Ocelli (or simple eyes) – 3, small, between compound eyes
Mouth parts – Labrum (upper lip), mandibles (jaws) below the labrum, maxillae located behind the mandibles to help cut & hold food, and the lower lip or labium



| 1. Labrum | 4. Labium |
| 2. Mandibles | 5. Maxillary Palps |
| 3. Labial Palps | 6. Maxillae |
| 7. compound eye | 8. ocelli |
- Label the mouthparts, eyes, and antenna on Figure 1.
- Using forceps, remove each of the appendages from the head, and attached them to table 1.
- Examine the following appendages on the thorax (middle section of the grasshopper’s body):
THORAX
Legs (first 2 pairs are for walking & the last pair are for jumping)
Wings (forewings have a leathery appearance & protect the hind wings)

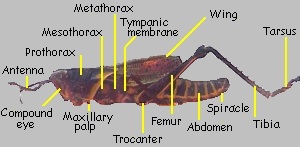
- Using forceps, remove one of the walking legs and identify these parts — the coxa connects the femur (the thickest part of the leg) to the grasshopper’s body; a slender, spiny tibia connects the femur to the tarsal segments (lowest part of the leg). Label these on Figure 2.
- Remove a jumping leg and attach the walking leg & jumping leg to Table 1.
- Raise both pairs of wings and locate the first abdominal segment.
- Locate the tympanic membrane or eardrum on the first abdominal segment. Label this on Figure 2.

- Using a magnifying glass, locate the spiracles or tiny pores for respiration on each side of the abdominal segments. Label these on Figure 2.
- Determine if your grasshopper is a male or female by looking at the end of the abdomen. Females have a tapered abdomen that ends in a pointed egg laying tube called the ovipositor. Male have a more rounded abdomen that turns upward.

- Label the ovipositor on Figure 2.

ABDOMEN
Spiracles (small openings on the side of somites or body segments)
Auditory Organs (two located laterally on the 1st body somite or segment)
Ovipositor (on female)
Observations & Conclusion:
Figure 1 – Grasshopper Head (Label ALL parts.)
Figure 2 – External Grasshopper anatomy (Label ALL parts.)
Table 1 – External Appendages of the Grasshopper (Attach ALL parts.)
| Antenna
|
|
| Labrum
|
|
| Mandible
|
|
| Maxilla
|
|
| Labium
|
|
| Forewing
|
|
| Hindwing
|
|
| Walking Leg
|
|
| Jumping Leg
|
|
| Sex of Grasshopper |
1. Which region of the insect’s body is specialized for sensory functions? Explain your answer.
2. Which region of the insect’s body is specialized for movement & explain why?
3. What is the purpose of compound eyes? of simple eyes?
4. List the grasshopper’s mouthparts & their functions.
5. How are the ends of the legs adapted for holding onto plants?
6. How is the third pair of legs adapted for jumping?
7. Describe the differences between the two pairs of wings (appearance & function).
8. How does the tympanic membrane help a grasshopper?
9. What system do spiracles open into on a grasshopper?
10. Do all abdominal segments have spiracles? Are there any spiracles on the thoraic segments?
11. How did you determine the sex of your grasshopper?
12. Explain how grasshoppers dig holes to lay their eggs.