Author: Biology Junction Team
Macromolecule Pictures


Lab 7 Sample 3 Fruitflies
Drosophila Genetics

Introduction
Drosophila Melanogaster, the fruit fly, is a great organism for genetic use because it has simple food requirements, occupies little space, is hardy, completes its life cycle in 12 days, makes a large number of offspring, can be knocked out easily, and it has many types of hereditary variations that can be seen with low power microscopes. Drosophila has a small number of chromosomes, four pairs. They are easily located in the large salivary glands. The Drosophila can be obtained from many places. Research of Drosophilae has led to a lot of knowledge about many of its genes.
Many factors combine to affect the length of the Drosophila life cycle. Temperature affects the life cycle the most. At room temperature the average life cycle of the Drosophila is about 12 days. Eggs of the Drosophila are small, oval shaped, and have two filaments at one end. They are usually laid on the surface of the culture medium, and with practice, can be seen with the naked eye. After one day the eggs hatch into the larva.
The larval stage of the Drosophila eats all the time. Larvae tunnel into the culture medium when they eat. The larva will shed its skin as it increases in size. In the last of the three larval stages, the cells of the salivary glands contain giant chromosomes that can be seen under low power in a microscope.
The pupal stage. Before a larva becomes a pupa it climbs the side of the container. The last larval covering then becomes harder and darker, forming the pupal case. Through this case the later stages of metamorphosis to an adult fly can be seen. In particular, the eyes, the wings, and the legs become visible.
The adult stage. When metamorphosis is over, the adult fly emerges form the pupal case. They are fragile and light in color and their wings are not fully expanded. They get darker in about an hour. They live about a month and then die. A female refrains from mating for about 12 days after she emerges from the pupal case. After she mates her receptacles contain large amounts of sperm and she lays her eggs. Make sure that the first flies you use are virgins.
The experiment will take several weeks. You will be assigned Drosophila with well-defined mutant traits by your teacher. You will keep a close record of what happens as each of these flies mate and pass there traits off to their offspring over a few generations.
There are three types of crosses that are studied in this lab. In monohybrid crosses the mode of inheritance is determined when a single contrasting pair of characteristics is involved. In a dihybrid cross the mode of inheritance is determined when the two pairs of contrasting of characteristics are considered simultaneously. In a sex-linked cross the mode of inheritance is determined when the mutant characteristic is associated with the X chromosome.
Hypothesis
In the sex linked cross of Drosophila Melanogaster, a phenotypic ratio of 1:1 will be obtained.
Materials
The materials used in this lab are as follows: a vile of Drosophilia with c designated trait, vials containing a medium, a refrigerator, ice packs, Petri dishes, a light microscope, a vial of wild type flies, an incubator, a pencil and paper.
Methods
Begin by obtaining a vial of wild type flies. Practice immobilizing and sexing these flies. Make sure to examine the flies and determine the characteristics of their eyes, wings, bristles, and antennae. Next, these are the steps for immobilizing the flies. Hold the vial containing the flies at an angle and place it in a refrigerator for several minutes. When the flies are immobilized, place them into a small plastic Petri dish. Then place the Petri dish on top of the icepack in order to maintain the cool temperature necessary to keep flies immobilized. Use the dissecting microscope to view the flies. Make sure to top the petri dish on when viewing the flies.
You can easily distinguish male flies from females by looking for the following characteristics: males are usually smaller than the females, males have dark blunt abdomens and females have lighter pointed abdomens. The males have sex combs, which are black bristles on the uppermost joint of the forelegs. Next, get a vial containing experimental flies. Make sure to write down the number of the vial that you have. The flies you now have are the P1 generation. The females should have laid eggs. The eggs and larvae are the F1 generation. Then after there are eggs present knock out remove the adult flies from the vial. Sex the adult flies and write down any mutations. Place the flies in the morgue that contains alcohol. Make sure to label the vial with the symbols for the mating.
After about another week has passed knock out and record characteristics of the remaining F1 flies and record the results in table 7.1. Then place the six pairs of these flies in a new vial and place the remaining flies in the morgue. Label the new vial F1, and tell the cross, date and your name.
After another week has passed, remove the F1 flies and put them in the morgue. The F2 generation are the eggs and larvae in the vial. Place the vial back into the incubator. Once again, after another week has passed remove the F2 flies and record their sex and characteristics and place the results in Table 7.2. Recording a greater number of F2 flies will make your results more accurate. Try to collect at least 200 flies. In order to analyze your data you will first have to be able to be able to complete Chi-Square Analysis.
Results
Table 7.1 F1 Generation
| Phenotype | females | males | ||
| Red eyes | 33 | 0 | ||
| White eyes | 0 | 31 | ||
|
Table 7.2 F2 Generation |
||||
| Phenotype | Male | Female | ||
| Red eyes | 50 | 52 | ||
| White eyes | 56 | 67 | ||
1. Describe the observed mutations? In the F1 generation the males had white eyes and the females had red eyes. In the F2 generation the males and females could have had either red or white eyes.
2. Write a hypothesis which describes the mode of inheritance of the trait you studied. This is your null hypothesis ( as described in the Statistical Analysis Section). For a sex linked cross there will always be a one to one ratio of the phenotypes. In the F1 generation there will be a one to one ratio of red eyed females to the number of white eyed males. In the F2 generation there will be a one to one ratio of red eyed females to white eyed females. There will also be a one to one ratio of red eyed males to white eyed males.
3. Refer to a textbook and review Punnett squares. In the space below construct two Punnett squares to predict the expected results of both the parental and F1 crosses from your null hypothesis.
Parental cross
| Y | Xr | |
| Xr | YXr | Xr Xr |
| Xr | YXr | Xr Xr |
F1 cross
| Y | Xr | Y | XR | |
| XR | YXR | XR Xr | YXR | XRXR |
| XR | YXR | XR Xr | YXr | XR Xr |
| Xr | YXr | XrXr | YXr | X RXr |
| Xr | YXr | XrXr | YXr | XRXr |
4. Refer to the Punnett squares above. Record the expected ratios for the genotypes and phenotypes of the F1 and F2 in the experiment below.
| Expected Genotypic Ratio | Expected Phenotypic ratio | ||
| F1 | 1:1 | 1:1 | |
| F2 | 1:1 | 1:1 | |
5. Do the actual results deviate from what was expected? If so, explain how.
No my results do no deviate much from what was expected. However in the F2 generation there were 67 white females and 52 red females.
6. For the results describe your cross? My cross is a sex linked cross.
7. Are the deviations for the phenotypic ratio of the F2 generation within the limits expected by chance? To answer this question, statistically analyze the data using the Chi-Square-Analysis. Calculate the Chi-Square for the F2 generation in the chart below. Refer to the critical values of the Chi Square distribution table to determine the P value that is associated with your statistic.
| Observed Phenotypes (o) | Expected (e) | (o-e) | (o-e)2 | (o-e)2
e |
|
| 67 Xr Xr | 56 | 11 | 121 | 2.16 | |
| 52 XR XR | 56 | -4 | 16 | .28 | |
| 50 YXR | 56 | -6 | 36 | .64 | |
| 56YXr | 56 | 0 | 0 | 0 | |
| 3.02 | |||||
(a) Calculate the Chi-Square value for these data.
1. How many degrees of freedom are there? 3 degrees of freedom
2. Chi Square=2.52
3. Referring to the critical values chart what is the probability value for these data? Greater than .05 probabilities that the null hypothesis is right.
(b) According to the probability value, can you accept or reject your null hypothesis? Explain why. I can accept the null hypothesis because my Chi-Square answer is less than the critical value form the table. I have 3 degrees of freedom and my Chi- Square answer was3.0 which is less than 7.82.
1. Why was it necessary for the females of the parental generation to be virgins? The females store sperm in their receptacles and if they were not virgins we would not be able to tell who the fathers were.
2. Why was it not necessary to isolate virgin females for the F1 cross? The females store sperm in their receptacles and if they were not virgins we would not be able to tell who the fathers were
3. Why the adult flies were removed from the vials at weeks 2 and 4? So, they are not be able to mate with the next generation.
Chi-Square Analysis
Introduction
Statistics can be used to determine if differences among groups are significant, or simply the result of predictable error. The statistical test most frequently used to determine whether data obtained experimentally provide a good fit, or approximation to the expected or theoretical data is the Chi-square test. This test can be used to determine if deviations from the expected values are due to chance alone or to comeother circumstance.
To determine if the observed data fall with in acceptable limits, a Chi-Square analysis is performed to test the validity of a null hypothesis; that there is no statistically significant difference between the observed and expected data. If the Chi-Square analysis indicates that the data vary too much from the expected 3: 1 an alternative hypothesis is accepted.
Methods
The formula for Chi-square is:
X2=E(o-e)2
E
O= observed number of individuals
e= expected number of individuals
E= the sum of the values
The (df) are determined by taking the number of possible phenotypes and subtracting one from it. If the Chi- Square answer is greater than the number from the critical values chart then the null hypothesis is incorrect. The results are said to be significant at .05. This means that only 5 % of the time you would expect to see similar data if the null hypothesis were correct. The probability can also be rejected at .001. This time it means that less than 1 % of the time would you expect to see similar data.
Results
Critical Values Chart
|
Degrees of Freedom (df) |
|||||
| 1 | 2 | 3 | 4 | 5 | |
| .05 | 3.84 | 5.99 | 7.82 | 9.49 | 11.1 |
| .01 | 6.64 | 9.21 | 11.3 | 13.2 | 15.1 |
| .001 | 10.8 | 13.8 | 16.3 | 18.5 | 20.5 |
Practice Problem
An investigator observes that when pure-breeding long winged Drosophila are mated with pure breeding short wing flies the F1 have an intermediate wing length. When several intermediate wing length flies are allowed to interbreed the following results are obtained. 230 long wings. 510 intermediate length wings. 260 short wings.
a. What is the genotype of the F intermediate wing length flies? The genotype is Ll.
b. Write a hypothesis describing the mode of inheritance of wing length in Drosophila. There will be 333 long winged flies. 666 intermediate winged flies. There will be 33 short wing flies.
c. Complete the table
Table 7.8
| Observed Phenotypes | Expected (e) | (o-e) | (o-e)2 | (o-e)2/ e |
| LL | 333 | -103 | 10609 | 31.86 |
| Ll | 666 | -156 | 24336 | 36.54 |
| ll | 333 | -73 | 5329 | 16.00 |
| 84.4 | ||||
1. How many degrees of freedom are there? There are 2 degrees of freedom.
2. Chi-Square= 84.4
3. Referring to the critical values chart, what is the probability value for these data? Less than .001.
4. According to the probability value can you except or reject the null hypothesis?
I can reject the null hypothesis because the Chi-square answer is greater than the critical value from the table.
Error Analysis
Results from this lab could have been affected by many things. The constant knocking out of flies could have caused some of the larvae to not hatch therefore affecting our numbers. Also, incorrectly identifying the characteristics of the flies could have also greatly affected the results received. Improper calculation of numbers could have also caused inaccurate results. Finally, some flies could have gotten stuck in the medium and could have been identified.
Conclusion
From the results of the experiment I can conclude that I received results that were close to a 1:1 ratio. The Chi- Square worked from my data was accepted at a possibility greater than .05. The null hypothesis in this case can be accepted.
Lab 7 Genetics of Organisms
Lab 7 Genetics of Organisms
Introduction:
Drosophila melanogaster, the fruit fly, is an excellent organism for genetics studies because it has simple food requirements, occupies little space, is hardy, completes its life cycle in about 12 days at room temperature, produces large numbers of offspring, can be immobilized readily for examination and sorting, and has many types of heredity variations that can be observed with low power magnification. Drosophila has a small number of chromosomes ( four pairs). These chromosomes are easily located in the large salivary gland cells.
The Life Cycle of Drosophila
- The eggs. The eggs are small, oval shaped, and have two filaments at one end. They are usually laid on the surface of the culture medium and, with practice, can be seen with the naked eye. The eggs hatch into larvae after about a day.
- The larval stage. The worm like larvae eats most continuously, and its black mouth parts can be seen moving back and forth even when the larvae is less distinct. Larvae tunnel through the culture medium when eating; thus channels are a good indication of a successful growth culture. The larvae molt twice as it increases in size. In the last of the three larval stages, the cells of the salivary glands contain giant chromosomes, which may be seen readily under low-power magnification after proper staining.
- The pupal stage. When a mature larvae in a laboratory culture is about to become a pupa, it usually climbs up the side of the container or on to a paper strip provided in the culture bottle. The last larval covering becomes harder and darker, forming a pupal case.
- The adult stage. When metamorphosis is complete, the adult flies emerge from the pupal case. They are fragile and light in color and their wings are not full expanded. These flies darken in a few hours and take on the appearance of an adult fly. They live a month or more and then die. A female does not mate for 10-12 hours after emerging from the pupa. Once she has mated, she stores a considerable quantity of sperm in receptacles and fertilized her eggs as she lays them. To ensure a controlled mating, it is necessary to use females that have not been mated (virgins).
Figure 7.1 The Life Cycle of Drosophila melanogaster

Design of the Exercise
This genetics experiment will be carried on for several weeks. Drosophila with well-defined mutant traits will be assigned to you by your teacher. You are responsible for making observations and keeping records concerning what happens as mutant traits are passed from generation to the next.
You will be assigned to study a certain mode of inheritance using particular genetic crosses of flies having one or two mutations. The modes of inheritance most commonly used are:
- Monohybrid. In these experiments, the mode of inheritance is determined when a single contrasting pair of traits is involved.
- Dihybrid. In these experiments, the mode of inheritance is determined when two pairs of contrasting traits are considered at the same time.
- Sex-linked. In these experiments, the mode of inheritance is determined when the mutant characteristic is associated with the X chromosome.
Procedure:
1. Obtain a vial of wild- type flies. Practice immobilizing and sexing these flies. Examine these flies and note the characteristics of their eyes, wings, bristles, and antennae.
2. To make handling easier, immobilize the flies with fly-nap, or by twirling the vial in ice packs for several minutes. Place the immobilized flies on a piece of filter paper inside a petri dish. Place this under a dissecting microscope to view the flies.
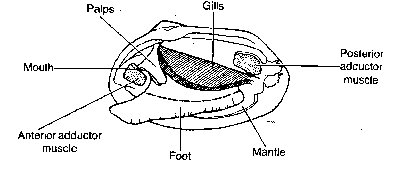
3. Distinguish male flies from female flies by looking for the following characteristics:
a). Males are usually smaller than females.
b). Males have dark, blunt abdomens, and females have lighter, pointed abdomens.
c). Only males have sex combs. which are groups of black bristles on the upper most joint of the forelegs.
Figure 7.2: Female and Male Drosophila
Female

Male
Male with vestigial wings
4. Obtain a vial containing pairs of experimental flies. Record the cross number of the vial. This number will serve as a record as to which cross you obtained. These flies are the parental generation(P1) and have already mated. The female should have already laid eggs on the surface of the culture medium. The eggs represent the first filial, F1 generation and will be emerging from their pupal cases in about a week.
5. First week (today): Immobilize and remove the adult flies. Observe them carefully under the dissecting microscope. Separate the males from the females and look for the mutation(s). Note whether the mutation(s) is/are associated with the males or females. Identify the mutation(s) and give them a made up name and symbol. Record the phenotype and symbol in Table 7.1. The findings should be confirmed by your teacher.
6. Place the parents in the morgue(jar containing alcohol). Label the vial containing the eggs or larvae with the symbols for the mating. Also label the vial with your name and date. Place the vial in a warm location.
7. Second week: Begin by observing the F1 flies. Immobilize and examine all the flies. Record their sex and characteristics. Consider the conclusion s that can be drawn from these data. Place five or six pairs of F1 flies in a fresh culture bottle and the rest of the flies in the morgue. For this cross the females need not be virgins. Label the vial with the symbols , name, and date.
8. Third week: Remove the F1 flies from the vials and place them into the morgue. The F2 generation are the eggs and /or larvae in the vial. Place the vial in a warm place.
9. Fourth week: Begin removing the F2 flies. Record their sex and the presence or absence of mutation(s). The more F2 flies collected, the more reliable the data will be. You may have to collect flies over a three-or four day period. Try to collect at least 200 flies.
10. To analyze your data, you will need to learn how to use the Chi-Square Test. Go to the Statistical Analysis Section to review the technique.
Table 7.1: F1 Generation Data
Date _________________
| Phenotype and Symbol | Females | Males |
Table 7.1: F2Generation Data
Date _________________
| Phenotype and Symbol | Females | Males |
Analysis of Results:
1. Describe and name the observed mutation(s).
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
2. Write a hypothesis which describes the mode of inheritance of the trait(s) you studied. This is your null hypothesis ( as described in the statistical analysis section).
____________________________________________________________________
____________________________________________________________________
3. Refer to a textbook and review Punnett squares. In the space below, construct two Punnett squares to predict the expected results of both the parental and F1 crosses from your null hypothesis.
| Parental Cross | F1 Cross |
| f | r |
4. Refer to the Punnett squares above. Record the expected ratios for the genotypes and phenotypes of the F1 and F2 in the experiment below.
| Expected Genotypic Ratio | Expected Phenotypic Ratio | |
| F1 | ||
| F2 |
5. Do the actual results deviate from what was expected? If so explain how.
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
6. From the results describe your cross: is it
Sex-linked or autosomal?_________________________
A dominant mutation or a recessive mutation? ________________________
Monohybrid or Dihybrid? ________________________
7. Are the deviations for the phenotypic ratio of the F2 generation within the limits expected by chance? To answer this question, statistically analyze the data using the Chi-square analysis. Calculate the Chi-square statistic for the F2 generation in the chart below.
| Observed Phenotypes (o) | Expected (e) | (o-e) | (o-e)2 | (o-e)2
e |
| X2 = |
a). Calculate the Chi-square value for these data.
1. How many degrees of freedom are there? ___________________
2. Chi-square (X2) = __________________
3. Referring to the critical values chart, what is the probability value for these data? _____________
b). According to the probability value, can you accept or reject your null hypothesis? Explain why.
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
Topics for Discussion:
1. Why was it necessary for the females of the parental generation to be virgin?
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
2. Why was it not necessary to isolate virgin females for the F1 cross?
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
3. Why were the adult flies removed from the vials at weeks 2 and 4?
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
_____________________________________________________________________
Lab 6 & Molecular Biology
| AP Lab 6
Molecular Biology |

Introduction: Exercise 6A: Recombinant DNA Technology
To study the structure and function of a single protein-coding gene, one must prepare the gene in a purified form. Vertebrate cells contain enough DNA to code for more than I 00,000 proteins; therefore it is not very practical to isolate a gene by conventional biochemical procedures. This is why recombinant DNA technology is so important; it can be used to isolate and amplify a specific gene relatively simply.
Plasmids, small circular DNA molecules, are usually extra chromosomal; they exist apart from the chromosomes in most bacterial species. Plasmids are not necessary for the survival of the host bacteria, but they can contain genes that enable the bacteria to survive in certain environments. If a bacterial cell contains a plasmid carrying a gene that confers resistance to antibiotics, then that cell could survive in the presence of the drug.
Plasmids can be introduced into bacterial cells by the process of transformation. Bacteria placed in a calcium chloride solution can take in plasmid DNA molecules. In this way, large amounts of specific plasmid DNA can be prepared, because one transformed cell gives rise to duplicate cells also containing the plasmid DNA molecule. Plasmids are very important for the molecular biologist because they serve as gene- carrier molecules called cloning vectors. A gene of interest can be joined to vector DNA to form a hybrid or recombinant molecule that can replicate in bacteria. When preparing a recombinant DNA molecule, a procedure is required for cutting cloning vectors and cellular DNA molecules in precise positions.
Restriction nucleases are important for recombinant DNA technology because they cut DNA at specific sites. These enzymes are usually made by bacterial species in which they degrade invading foreign DNA within the bacterial cell. Most restriction enzymes recognize a specific sequence of nucleotides in DNA and cut a long DNA double helix into restriction fragments, which are measured in the process of agarose gel electrophoresis.
Introduction: Exercise 6B: DNA Fingerprinting
Electrophoresis is the movement of charged particles in solution under the influence of an electric field. In gel electrophoresis, agarose gel is the stabilizing medium that serves as a matrix for the buffer in which the sample molecules travel. The gel is submerged in buffer within the electrophoretic gel cell. The samples are loaded into the sample wells in the gel, and electric current is passed through the gel.
Molecules of DNA are negatively charged because of negative charges on the phosphate group. In this exercise, nucleic acids migrate through the pores of the gel from the negative end of towards the positive end. The large DNA molecules move more slowly than smaller molecules, therefore molecules are sorted according to size.
Objective: Exercise 6A
Investigate basic genetic concepts by transforming bacterial cells by inserting an ampicillin-resistant gene into E. coli cells.
Objective: Exercise 6B
Investigate basic genetic concepts by using restrictive enzymes to digest phage lambda DNA and separate and identify the DNA fragments using gel electrophoresis.
Materials and Methods: Exercise 6A
The materials used in this exercise included: 2 Luria agar plates, 2 Luria agar plates with ampicillin, 2 microcentrifuge tubes, 1 inoculating loop, 1 Bacti-Spreader, sterile micropipets, calcium chloride, Luria broth, plasmid pUC8, Bunsen burner, hotplate, ice, waterbath.
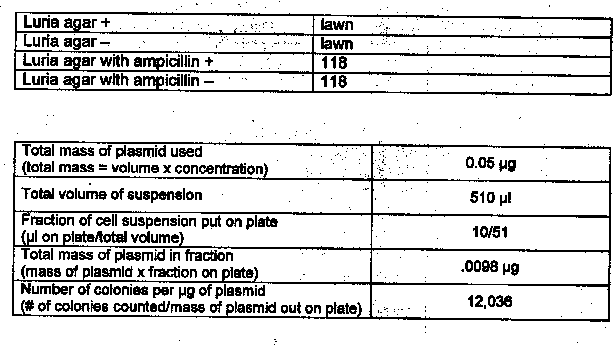
The two microcentrifuge tubes were marked !I+” and !1-!1, and 250IJI cold calcium chloride was added to each using a pipet. A large colony of bacteria was added to each tube with a sterile inoculating loop. A micropipet was used to transfer 10IJI of the plasmid pUCS solution to the !I+” tube. Both tubes were incubated on ice for 15 minutes, and meanwhile, the two Luria agar plates were labeled “+” and “-” and so were the two Luria plates with ampicillin. The tubes were removed from ice and placed in a 42°C hot waterbath for 90 seconds. The tubes were then removed from the waterbath and placed on ice for two minutes. A micropipet was used to add 250IJI Luria broth to each tube. Another micropipet was used to add 100IJI of the !I+” solution to the two !I+” plates and 1oomicroliters of the solution to the two “-” plates. The bacteria was flamed to sterilize, and after cooling, was used to spread the cells over the entire surface of the plates. After five minutes, the plates were placed in a 37°C incubator, inverted, overnight.
Materials and Methods: Exercise 6B
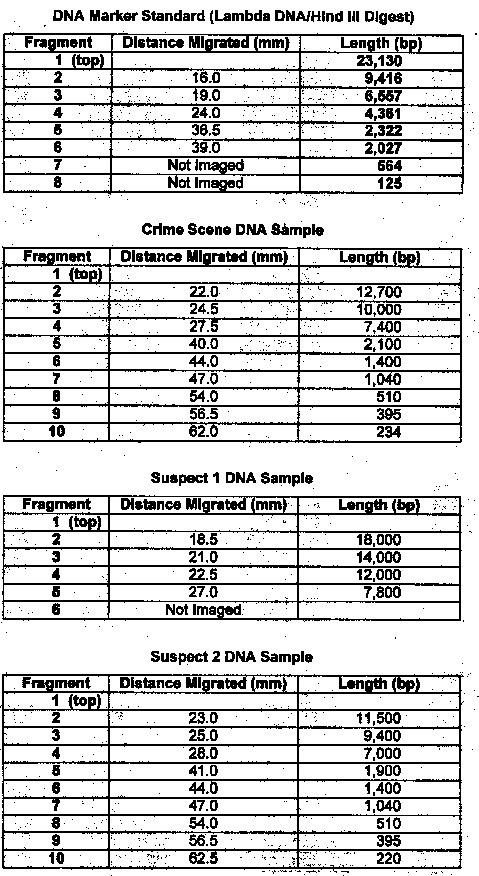
The materials used in this exercise included: 8% agarose gel, 2 electrophoresis chambers, power pack, running buffer- Tris, micropipetter and tips, staining tray, methylene blue dye, gloves, aprons, 4 DNA samples cut with restrictive enzymes, vial tray, microcentrifuge, paper, pencil, distilled water, spatula, plastic container for destaining, masking tape, light box, ruler, semi-Iog graph paper. The gel, on the gel tray, was placed in the center of the chamber, with the well-side of the gel near the black electrode. Approximately 350ml of running buffer were added to the chamber. Of each DNA sample, 10 microliters was loaded into the corresponding gel lane with a micropipet. The power cords were attached to the appropriate connections, and the power supply was turned on, set to 50 volts. The samples were allowed to migrate for three hours. The gel was then removed, stained, and destained overnight. The gel was viewed on a light board, and the band migration distances were measured.
Results: Exercise 6A
Questions
1. Based on your experimental results, did transformation occur? Why or why not?
Yes, transformation did occur. Colonies of E. coli grew in the presence of ampicillin.
2. What other methods can be used to verify that transformation occurred?
DNA fingerprinting
Results: Exercise 6B

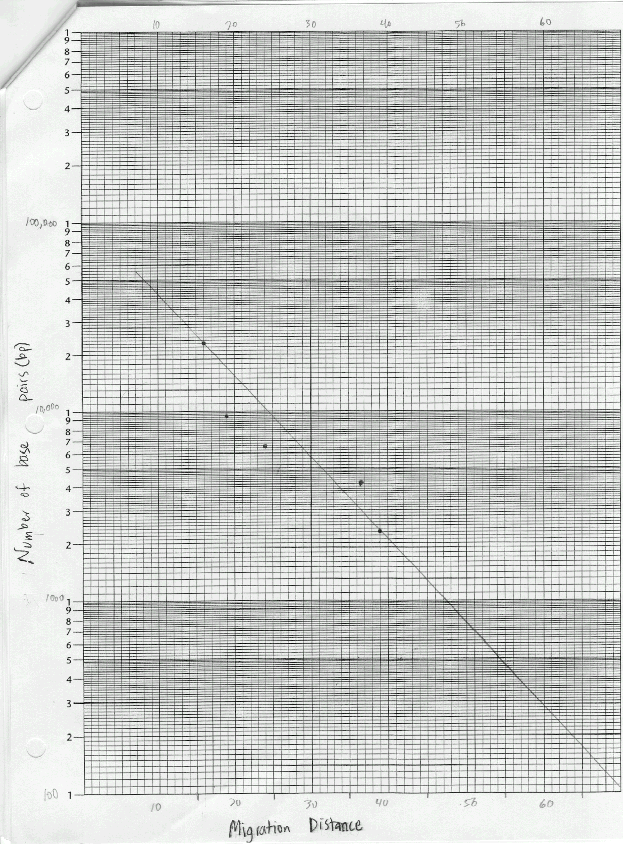
Questions
1. Compare the banding patterns. Do you think the DNA samples were the same?
No, the samples were different sizes
2. Which of the two suspects were the real burglar? Suspect #2
3. Explain the function of each of these steps in DNA fingerprinting:
a. Restriction Enzyme digest – used to cut DNA
b. Gel electrophoresis – used to separate different size pieces of DNA
c. Denaturing into single-stranded DNA – process used to view only one strand of the DNA double helix
d. Southern Blot – DNA bands transferred to a nitrocellulose paper
e. Radioactive DNA probe – used to find & bind to the complementary sequence in one or more RLFP’s
f. Autoradiograph – used to show similarities in DNA samples
Error Analysis
Lab 6A: Not enough agar was poured on the plates.
Lab 6B: Base pair counts for the DNA bands could have been inaccurate as suggested by the best-fit line on the graph.
Conclusion
Lab 6A: This lab showed that genes can be inserted into living bacterial cells thus transforming the cells and giving them new properties such as ampicillin-resistance.
Lab 6B: Through gel electrophoresis of the DNA samples, it was determined that suspect #2 was guilty. Their DNA banding pattern matched the banding pattern taken of the DNA at the crime scene.
| BACK |
