| AP Lab 5 Cell Respiration |  |
Introduction:
Cellular respiration is the release of energy from organic compounds by metabolic chemical oxidation in the mitochondria in each cell. Cellular respiration involves a number of enzyme mediated reactions. The equation for the oxidation glucose is C6H12O6 + O2 à CO2 + H2O + 686 kilocalories per mole of glucose oxidized. There are three ways cellular respiration could be measured. The consumption of O2 (how many moles of O2 are consumed in cellular respiration). Production of CO2 (how many moles of CO2 are produced in cellular respiration?) and the release of energy during cellular respiration. In this lab, the volume of O2 consumed by germinating and non-germinating peas at two different temperatures will be measured.
PV=nRT is the inert gas law. P is the pressure of the gas. V is the volume of the gas. n is the number of molecules of gas. R is the gas constant. T is the temperature of the gas in degrees K. This law tells us several important things about gases. If temperature and pressure are kept constant then the volume of the gas is directly proportional to the number of molecules of the gas. If the temperature and volume remain constant, then the pressure of the gas changes in direct proportion to the number of molecules of gas. If the number of gas molecules and the temperature remain constant, then the pressure is inversely proportional to the volume. IF the temperature changes and the number of gas molecules is kept constant, then either pressure or volume or both will change in direct proportion to the temperature.
In this lab, CO2 , made during cellular respiration will be removed by potassium hydroxide (KOH) and will make potassium carbonate (K2CO3). Carbon dioxide is removed so the change in the volume of gas in the respirometer will be directly proportional to the amount of oxygen that is consumed. In the experiment water will move toward the region of lower pressure. During respiration, oxygen will be consumed and its volume will be reduced to a solid. The result is a decrease in gas volume within the tube, and a related decrease in pressure in the tube. The respirometer with just the glass beads will allow changes in volume due to changes in atmospheric pressure or temperature changes.
Hypothesis:
The respirometer with only germinating peas will have a larger consumption of oxygen and will have a larger amount of CO2 that is converted into K2CO3 than the respirometer with beads and dry peas and the respirometer with beads alone.
Materials:
The materials used in the lab are as follows: a thermometer, 2 water baths, tap water, masking tape, germinating peas, non-germinating (dry) peas, 100 mL graduated n cylinder, 6 vials, 6 rubber stoppers, absorbent and non absorbent cotton, KOH, 5 mL syringe, 6 pipettes, ice, and 6 washers.
Methods:
First, set up both a room temperature 25oC and a 10oC water bath. Make sure you allow time to adjust the temperature in each bath. To obtain a temperature of 10oC add ice to of the baths until the temperature in the bath is 10oC. Next, obtain a 100 mL graduated cylinder and fill it with 50 mL of water. Drop in 25 germinating peas and determine the amount of water that is displaced. Record the volume of the 25 germinating peas. Then remove these peas and place them on a paper towel. They will be used in respirometer 1. Next, refill the graduated cylinder with 50 mL of water and drop 25 non-germinating peas into it. Then drop glass beads into the respirometer until the volume is equivalent to that of the expanded germinating peas. Remove the beads and peas. They will be used in respirometer 2. Next, refill the graduated cylinder with 50 mL of water. Determine how many glass beads would be required to attain a volume that is equivalent to that of the germinating peas. Remove the beads. They will be used in respirometer 3. Then repeat the procedures used above to prepare a second set of germinating peas, dry peas + beads, and beads to be used in respirometers 4,5,and 6.
Assemble the six respirometers by obtaining 6 vials, each with an attached stopper and pipette. Then put a small wad of absorbent cotton in the bottom of each vial and, using the syringe, saturate the cotton with 15 % KOH making sure not to get the KOH on the sides of the respirometer. Then place a small wad of dry cotton on top of the KOH-soaked absorbent cotton. Repeat these steps to make the other five respirometers. Make sure to use about the same amount of cotton in each vial.
Next, place the first set of germinating peas, dry peas + beads and beads in vials 1,2, and 3. Place the second set of germinating peas, dry peas + beads, and beads in vials 4,5, and 6. Insert the stoppers in each vial with the proper pipette. Place a washer on each of the pipettes to be used as a weight.
Make a sling using the masking tape and attach it to each side of the water baths to hold the pipettes out of the water during the equilibration period of 10 minutes. Vials 1,2, and 3, should be in the bath containing water of 25o C. Vials 4, 5, and 6 should be in the bath containing water that is 10oC. After the equilibration period completely immerse all six respirometers in the water completely. Water will enter the pipette for a short distance and stop. If it does not stop, there is a leak. Make sure the pipettes are facing so you can read them. The vials should not be shifted during the experiment and your hands should not be placed in the water during the experiment.
Allow the respirometers to equilibrate for three more minutes and then record the initial water in each pipette time 0. Check the temperature in both baths and record in table 5.1. Every five minutes for 20 minutes, take readings of the water’s position in each pipette, and record the data in table 5.1.
Results:
Table 5.1: the Measurement of Oxygen Consumption by Soaked and Dry Pea Seeds at Room Temperature 25o C and 10oC Using Volumetric methods.
| Temp o C | Time (min) | Reading at time X | Diff. | Reading at time X | Diff. | Corrected Diff. | Reading at time X | Diff. | Corrected Diff. |
| 25 | Initial- 0 | 14.4 | 13.9 | 14.2 | |||||
| 25 | 0 to 5 | 14.1 | .3 | 13 | .9 | .6 | 14.1 | .1 | -.2 |
| 25 | 5 to 10 | 14.0 | .4 | 11.1 | 2.8 | 2.4 | 13.9 | .3 | -.1 |
| 25 | 10 to 15 | 13.9 | .5 | 10.3 | 3.6 | 3.1 | 13.7 | .5 | 0 |
| 24 | 15 to 20 | 13.9 | .5 | 8.8 | 5.1 | 4.6 | 13.5 | .7 | .2 |
| 10 | Initial – 0 | 14.2 | 14.2 | 14.7 | |||||
| 10 | 0 to 5 | 14.8 | -.6 | 14.0 | .2 | .8 | 15.2 | -.5 | .1 |
| 10 | 5 to 10 | 14.6 | -.4 | 13.5 | .7 | 1.1 | 15 | -.7 | -.3 |
| 10 | 10 to 15 | 14.8 | -.6 | 13.2 | .9 | 1.5 | 15 | -.7 | -.1 |
| 10 | 15 to 20 | 14.9 | -.7 | 12.6 | 1.6 | 2.3 | 15 | -.7 | 0 |
Graph: Consumption of Oxygen for Germinating Peas and Dry Peas at 10oC and 25o C.
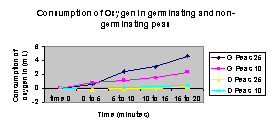
Questions:
1. In this activity, you are investigating both the effect of germination versus non-germination and warm temperature versus cold temperature on respiration rate. Identify the hypothesis being tested in this activity.
The hypothesis being tested in this activity is that the germinating peas in a water bath of 25 o C will have a higher respiration rate than the other vials.
2. This activity uses a number of controls. Identify at least three of the controls, and describe the purpose of each control.
One control is each vial had the same volume. This showed that the volume of the vial did not effect respiration rate. Another control was the vial with beads alone. The beads carried out no respiration. The final control was the 10 minute equilibration period. This allowed the contents of the vials to carry out respiration for a short period of time before they were completely immersed in the water.
3.Graph the results from the corrected difference column for the germinating and dry peas at both room temperature and at 10oC.
4. Explain the relationship between the amount of oxygen consumed and time.
As time increased oxygen consumption increased.
5. From the slope of the four lines on the graph, determine the rate of oxygen consumption of germinating and dry peas during the experiments at room temperature and at 10o C.
| Condition | Show Calculations | Rate in mL O / minute |
| Germinating peas at 10oC | 2.3-1.5=.8/5 | .16mL O2 /minute |
| Germinating peas at room temperature | 4.6-3.1/5 | .3mL O2 /minute |
| Dry peas at 10oC | (.1)/5= | .02 mL O2 /minute |
| Dry peas at room temperature | (.2-0 )/5= | .04 mL O 2 /minute |
6. Why is it necessary to correct the readings from the peas with the readings from the beads?
The beads carried out no cellular respiration. The peas did. Changes in atmospheric pressure could have caused changes in respiration rate and correcting the readings provided the most accurate results under the given conditions.
7. Explain the effect of germination versus non-germination on pea seed respiration.
Germination causes a higher rate of respiration than the non-germinating peas.
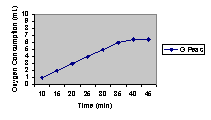
8. Graph the predicted results through 45o C. Explain your prediction.
As the temperature increased cellular respiration increased, but after a certain temperature the respiration rate will start to go down. The peak is the optimal temperature.

9. What is the purpose of KOH in this experiment?
KOH removes carbon dioxide formed during cellular respiration.
10. Why did the vial have to be completely sealed around the stopper.
The stopper was completely sealed to prevent water from entering the respirometer.
11. If you used the same experimental design to compare the rates of respiration of a 25g. reptile and a 25 g. mammal at 10oC what results would you expect? Explain your reasoning.
The mammal would carry out a higher rate of cellular respiration. This is because the mammal maintains a constant temperature that is higher than the temperature of the cold blooded reptiles that will have a temperature of 10 C.
12. If respiration in a small mammal were studied at both room temperature 21 o C and 10oC what results would you predict? Explain your reasoning.
The rate of cellular respiration would be higher at 21 degrees C because the 10 degrees C temperature could cause the overall body of the mammal temperature to drop the most.
13. Explain why water moved into the respirometers’ pipettes.
Water moved into the pipettes because oxygen was being consumed and allowed water to move only partially into the pipette.
14. Design an experiment to examine the rates of cellular respiration in peas that have been germinating for 0, 24, 48, and 72 hours. What results would you expect? Why?
I would use the same format using respirometers to measure the cellular respiration rate of the peas. The peas that had been germinating for 72 hours would have a higher respiration rate because they have a higher energy demand.
Error Analysis:
Several factors could have caused inaccurate results in this experiment. First, not maintaining a constant temperature in the water bath could have caused inaccurate results. Also moving the vials in the water after the experiment began could have caused inaccurate results. Putting your hands in the water bath while the vials were in the water could have caused inaccurate results. Allowing the peas to come into contact with the KOH could have also caused inaccurate results. Finally not having the same amount of cotton in each vial could have caused an error in the results.
Conclusion:
In this experiment the vial with just germinating peas had the greatest consumption of oxygen. This is because germinating peas carried out a more rapid process of cellular respiration than the non-germinating peas. The beads carried out no cellular respiration. The non-germinating peas require less energy than the germinating peas so the dry peas carry out a slower process of cellular respiration. This in turn caused less oxygen to be consumed in the vials with non-germinating peas than the vials with germinating peas. The higher temperature caused cellular respiration to occur at a higher rate which in turn caused a greater consumption of oxygen.