Lab 4 Plant Pigments & Photosynthesis

Introduction:
The purpose of this lab experiment was to separate plant pigments using paper chromatography, and to measure the rate of photosynthesis in isolated chloroplasts. Because of capillary action the solvent moves up the paper causing the pigments to become visible at certain distances.
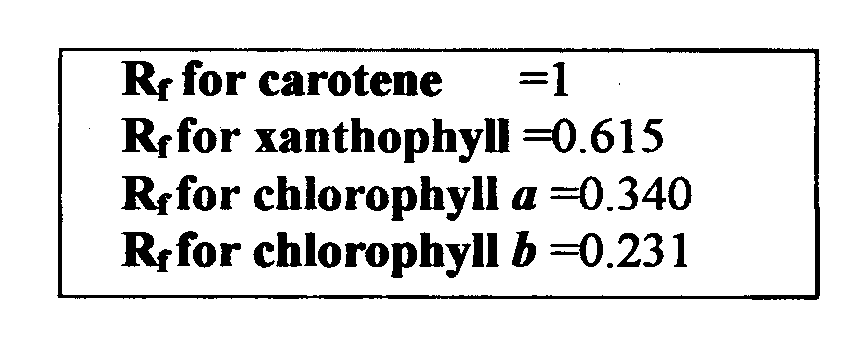
The substances visible on the paper are called pigments. Chlorophyll a is the main pigment that makes up about 75% of the pigmentation in plants. Chlorophyll b makes up about 25% of the pigmentation. And carotenes and xanthophylls are accessory pigments that make up the rest of the pigmentation. Carotene is the most soluble of the pigments and as a result will be carried the farthest by the solvent. The paper will display a spectrum of the pigments found in the spinach leaves. Using the formula Rf one can determine the relationship between the distance the solvent traveled to the distance the pigment traveled.
Rf=distance l2igment migrated (mm) distance solvent front migrated
Light is a part of a continuum of radiation or energy waves. The energy from visible light is used in the photosynthetic process. Light is absorbed in the leaf pigments, electrons within each photosystem are boosted to a higher energy level to produce ATP and to reduce NADP and NADPH. The ATP is then used in carbon fixation. This is the incorporation of CO2 into organic molecules.
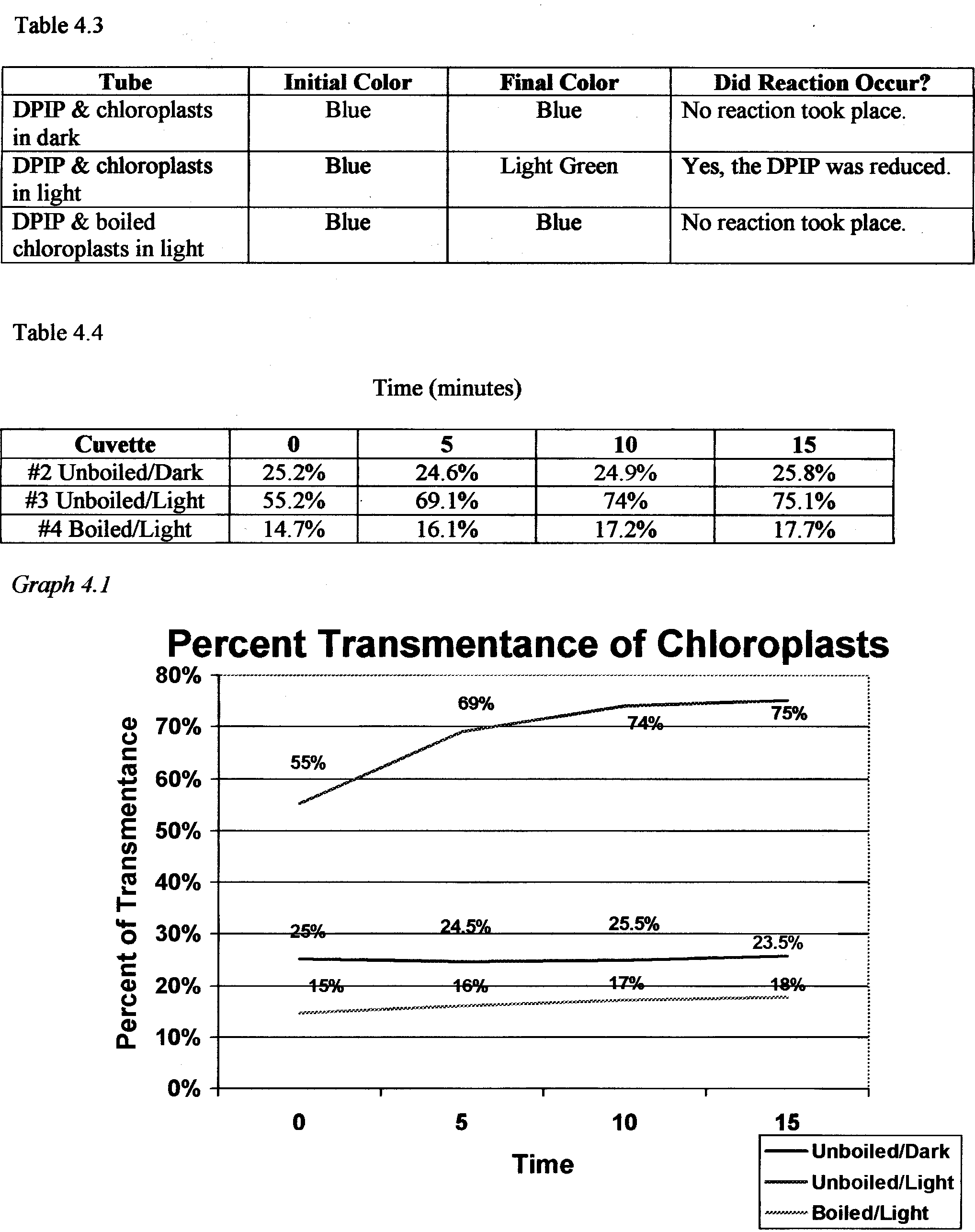
To measure light transmittance in chloroplasts a spectrophotometer will be used. The reason behind measuring the light transmittance is to calculate the rate of photosynthesis in the chloroplasts. A solution called DPIP will be used in place of NADP to judge the color change of the chloroplast solutions. This technique is known as dye- reduction and it tests the hypothesis that light and chloroplasts are required for light reactions to occur.
Hypothesis:
In this experiment it is hypothesized that the cuvette with boiled chloroplasts and the cuvette kept in the dark containing unboiled chloroplasts will have very slight changes in light transmittance, whereas the cuvette containing unboiled chloroplasts that have been exposed to light will have an increasingly higher % transmittance over the course of time.
Materials and Methods:
Lab 4A:
The materials used in this section of the lab were: filter paper, glass vial, small amount of solvent, a quarter, and spinach leaves. The first step was to cut a point on one end of the filter paper and draw a pencil line 1.5cm from the tip of this point. Next a spinach leave was placed on the strip of paper and rolled over with a quarter on top of the pencil line. This gives a green line across the paper, which contains the pigments of the leave. Then the strip of paper was placed into the vial with the point down in the bottom. When the pigment reached the point 1 cm from the top of the vial then it was removed. The solvent front was then quickly marked with a pencil and then each pigment front was marked as well. From the distance the pigment traveled and the distance the solvent traveled the Rf value was calculated.
Lab 4B:
The materials used in this lab were: a spectrophotometer, 4 cuvettes, phosphate buffer, distilled water, boiled chloroplasts, unboiled chloroplasts, and DPIP .First the cuvettes were labeled 1-4 and cleaned with lens paper because even the oil from your hands can affect the transmittance of light through the cuvette. Cuvette 2 was then wrapped with foil to keep the contents in the dark. Next 1 ml of phosphate buffer was added to all four cuvettes, 4ml of distilled water was added to cuvette 1, 3rn1 of distilled water was added to cuvettes 2,3, and 4, and Iml of DPIP was added to cuvettes 2,3, and 4. Then 3 drops of unboiled chloroplasts were added to cuvette 1, it was covered with parafilm, placed into the spectrophotometer, and set to 100% transmittance. This cuvette was used to recalibrate between readings as well. Three drops of unboiled chloroplasts were placed in cuvette 2 and 3, and three drops of boiled chloroplasts were placed in cuvette 4. The cuvettes were then covered with parafilm. Each was placed in the spectrophotometer and the % transmittance of each, every five minutes for 15 minutes, was recorded.
Data:
Table 4.1

Questions:
1. Which pigment migrated the furthest and why? Carotene, it was the most soluble and didn’t form bonds with the filter paper.
2. Which of the 2 types of chlorophyll is more soluble? Chlorophyll a
3. Why do leaves change color in autumn? The chlorophyll production in the leaves slows down.
4. What is the function of the chlorophyll in photosynthesis? They absorb red and blue light rays.
5 .What are the accessory pigments and what are their functions? Carotene and xanthophylls both absorb different wavelengths of light than chlorophyll does.
6. What are some other ways chromatography is used to separate plant pigments? There are three types: Column, Paper, and Thin Layer chromatography.
7 .What does the & value represent? The distance the pigment traveled and the distance the solvent traveled expressed as one value.
8. What factors involved in the separation of the pigments? In this test it was solubility.
9 .Would you expect the Rf value of a pigment to be same if a different solvent were used? Explain. No, for a different solute there would be a different solvent rate.
10. What kind of chlorophyll does the reaction center contain? What are the roles of the other pigments? Chlorophyll a” the other pigments catch different wavelengths of light.
Error Analysis:
Fingerprints on either the filter paper or the cuvettes may have affected the experiment because the oil from your hands can get on these things and affect the results. The spectrophotometer may have not been calibrated correctly because this was the first time this particular one had been used. Other than these there were few places for error in this lab.
Conclusion:
From lab 4a we discovered that the many pigments found in chloroplasts are all involved in gathering energy from sunlight. The spectrum of color displayed on the filter paper showed the pigments and the solubility of each. In lab 4b the spectrophotometer measured the light transmittance through the various cuvettes and the chloroplast solutions in each. The actual purpose of this was to observe the DPIP go from a blue color to a clear color. This indicated that photosynthesis was occurring and at what rate it was occurring. The cuvette with the unboiled chloroplasts that had been exposed to light showed the biggest change in % transmittance, which indicates that the amount of light available has a very big effect on the rate at which the light reactions of photosynthesis occur .