The States of Water 
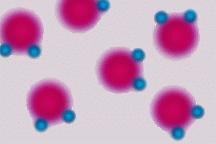
Most liquids contract (get smaller) when they get colder. Water is different. Water contracts until it reaches 4 C then it expands until it is solid. Solid water is less dense that liquid water because of this. If water worked like other liquids, then there would be no such thing as an ice berg, the ice in your soft drink would sink to the bottom of the glass, and ponds would freeze from the bottom up! Water is found on Earth in all three forms. This is because Earth is a very special planet with just the right range of temperatures and air pressures. Adhesion and Cohesion Water is attracted to other water. This is called cohesion. Water can also be attracted to other materials. This is called adhesion.
Surface Tension Surface tension is the name we give to the cohesion of water molecules at the surface of a body of water. Try this at home: place a drop of water onto a piece of wax paper. Look closely at the drop. What shape is it? Why do you think it is this shape? What is happening? Water is not attracted to wax paper (there is no adhesion between the drop and the wax paper). Each molecule in the water drop is attracted to the other water molecules in the drop. This causes the water to pull itself into a shape with the smallest amount of surface area, a bead (sphere). All the water molecules on the surface of the bead are ‘holding’ each other together or creating surface tension.  Surface tension is not the force that keeps boats floating. If you want to know why a boat floats look here: Why do boats float?
Capillary Action
Plants take advantage of capillary action to pull water from the into themselves. From the roots water is drawn through the plant by another force, transpiration. |