| Constructing Monomers of Organic Molecules | 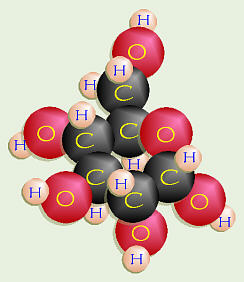 |
|
Introduction Almost all molecules made by cells are composed of carbon atoms bonded to one another and to atoms of other elements (especially H, O, N). Compounds made by cells and containing carbon are known as organic compounds. Carbon can form four covalent bonds with other carbons or other types of atoms. Cells make a huge number of large molecules from a small set of small molecules. These smaller molecules are called monomers and are linked together to make larger molecules known as polymers. Four main groups of organic compounds exist — carbohydrates, lipids, proteins, and nucleic acids. The monomers of carbohydrates are called monosaccharides or simple sugars. the 3 monosaccharides, glucose, fructose, and galactose, all have the same chemical formula, C6H12O6, but their structural formulas are different.
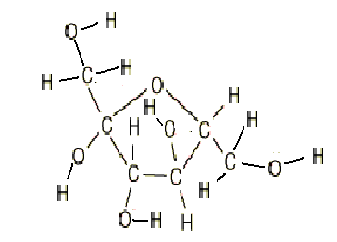
The monomers monomers that make up lipids are glycerol and fatty acid chains. Saturated fatty acid chains have all single bonds between carbons, while unsaturated fatty acids will have 1 or more double bonds between carbons in the chain.
The monomers making up proteins are called amino acids. Twenty different amino acids exist, but they all have the same basic structure — a central carbon has a single hydrogen , an amine group (-NH2), a carboxyl group (-COOH), and a side or R group attached. The side group determines the difference in properties.
Two Simple Amino Acids
Nucleic acids include DNA and RNA. The monomers of nucleic acids are called nucleotides and are composed of a pentose (5-sided sugar), a nitrogen containing base, and a phosphate group (-PO4). The sugar found in DNA is called deoxyribose, while the sugar in RNA is called ribose.
Objective Each student will construct a monomer of an organic compound found within organisms. You may construct one of the following — glucose, fructose, galactose, glycine, alanine, glycerol, saturated or unsaturated fatty acid, deoxyribose, or ribose. Materials Various materials may be used for the atoms that make up a nucleotide such as styrofoam balls, plastic coke bottle caps, beads, etc. Bonds between atoms may be made from toothpicks, plastic stirring sticks, popsicle sticks, etc. Single & double bonds must be represented by the correct number of “sticks”. The atoms and bonds may NOT be made of any food item. Your model should be glued together to make the model rigid for hanging. Attach string and a label with the nucleotide’s name to your model. Models must be sturdy, light weight, and small enough to hang from the ceiling. Color Code for atoms:
|
||||||||||||||||||||||||||||||



![[Glycerol]](https://biologyjunction.com/images/glycerol.jpg)





