| Chemistry Review |  |
1. Everything in the universe is made of __________________________________.
2. The measurement of the amount of matter in an object is called ___________________.
3. What are the Three States of matter?
A.____________________________________
B.____________________________________
C.____________________________________
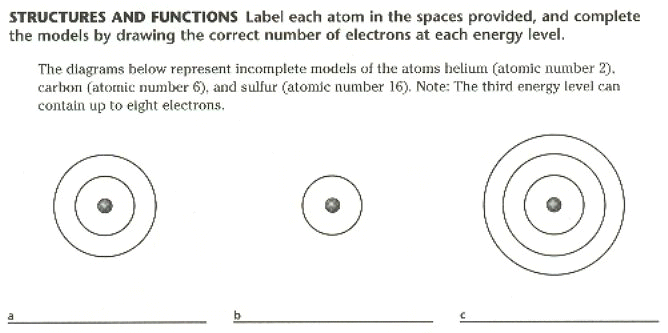
4. Charged particles that move around an atom’s nucleus are called ________________________.
5. Chemical bonds are broken, atoms are rearranged, and new bonds are formed during _______________________________ ______________________________.
6. Atoms with filled outermost energy levels tend _____________ to participate in chemical reactions.
7. A pure substance that cannot be broken down is called an _____________________.
8. The simplest part of an element is an ____________________.
9. The central core of an atom is called the _____________________________.
10. In an ionic bond, __________ atoms of ________________ charge are held together by _________________________ attraction.
11. The part of an atom that has a neutral charge is a _______________________.
12. Most of the mass of an atom is found in the _____________________.
13. A pure substance made up of atoms of one or more elements is called a ____________________________.
14. Most atoms tend to undergo ____________________ _________________, combining in ways that cause their atoms to become more ____________________.
15.When two atoms share one or more electrons, it is called ____________________________ ______________________.
16. A bond formed by electrical attraction between two opposite charged ions is called ______________________ ____________________.
17. The ability to do work or cause change is _____________________.
18. A redox reaction involves the _____________________ of ___________________ between atoms.
19. The amount of energy needed to start a chemical reaction is the reaction’s _________________________ ___________________.
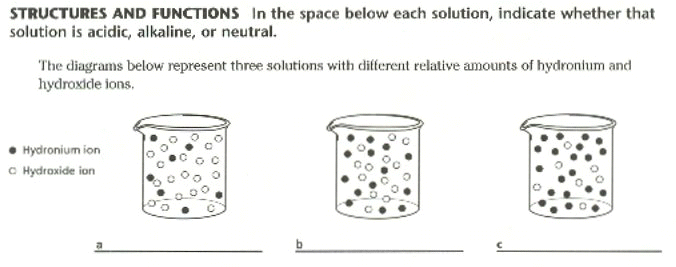
20. A substance that neutralizes small amounts of acids or bases added to a solution is a _______________________.
21. A chemical reaction that can proceed forward or backward is a ______________________ ______________________.
22. Sodium chloride (table salt) is an example of a compound formed by ______________ _________________.
23.The positive charge part of an atom is called a ___________________________.
24. A particle composed of one or more atoms is a ________________________.
25. Chemical reactions that release free energy are called ____________________________ ____________________________.
26. Chemical reactions that absorb free energy are called ____________________________ _______________________________.
27. The loss of one or more electrons is called ______________________.
28. The gaining of one or more electrons is called _______________________.
29. The breaking apart of water molecules into two ions of opposite charge is called ___________________________________.
30. An atom has six electrons, what is it atomic number? ____________ Name?___________ It is a stable or unstable atom? _________________.
DIRECTIONS: Read Chapter 2, Chemistry, and Answer the questions below as completely and as thoroughly as possible. Answer the question in essay form (not outline form), using complete sentences. You may use diagrams or pictures to supplement your answers, but a diagram or picture alone without appropriate discussion is inadequate.
1. Describe the dissociation of water.
2. Define acid and base. What is a buffer?
3. List TWO characteristics of Acids and TWO Characteristics of Bases.
4. Describe the relationship between the solute, the solvent, and the concentration of a solution.
5. How does an ionic bond differ from a covalent bond?
6. Why is it necessary for oxidation and reduction reactions to occur in pairs?
7. Define the Three States of Matter?
8. State the difference between endergonic and exergonic reactions.
9. What is the role of enzymes in chemical reactions occurring in living things? Explain how a catalyst affects a reaction.
10. What is the pH Scale, and what does its range of values mean?
11. Draw and Label a model of a Chlorine (Cl) atom. Is this atom stable? Why or Why not?
12. Describe the difference between an oxidation and reduction reaction.
13. An oxygen atom has six electrons in its outermost energy level. Explain why two oxygen atoms must share four electrons when they form a covalent bond.