Physical Properties of Water
All of water’s unique physical properties are caused by water’s polarity.
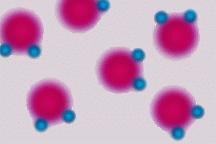
- Cohesion: water molecules stick to each other. This is caused by hydrogen bonds that form between the slightly positive and negative ends of neighboring molecules. This is the reason why water is found in drops; perfect spheres. It’s hard to imagine water behaving any other way.

- Adhesion: water molecules stick to other surfaces. This causes water to move upward against gravity in plant stems and to be absorbed by paper towels. It also causes water to adhere to spider webs.

- Surface Tension: water has the ability to support small objects. The hydrogen bonds between neighboring molecules cause a “film” to develop at the surface.

Organisms like the water strider can be seen taking advantage of this property.

4. Water has a high boiling point. Water is one of the few substances that remain a liquid at such a large range of temperatures (O-100 °C). A large amount of energy must be invested to overcome the hydrogen bonds in liquid water to change it to the gas phase.


Liquid Water Molecules with hydrogen bonds Water Vapor Molecules
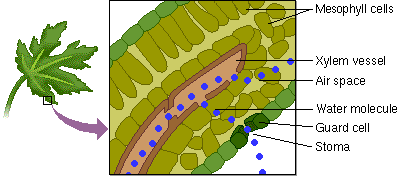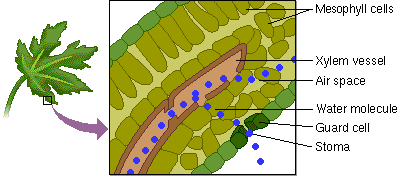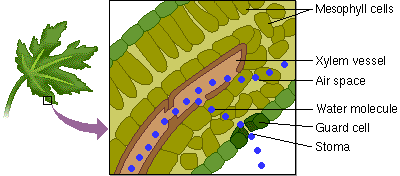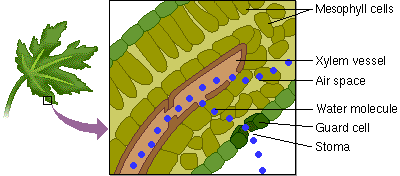
5. Capillary Action: water has the ability to “climb” structures. Think about what happens when you stick the tip of a straw in a glass of water.

6. Ability to Dissolve: water is consider to be the universal solvent. More substances will dissolve in water than any other liquid. This includes other polar substances (such as sugar) and ionic compounds (such as salt).
When a sugar crystal is placed in water, the slightly positive and negative ends of the water molecule attract the sugar molecules in the crystal (they are also polar) and pull them into solution.

When an salt crystals are placed in water, the slightly positive and negative ends of the water molecules attract the ions in the crystal. The ionic bonds holding the sodium and chlorine ions together are broken and the ions are pulled into solution.


7. High Heat of Vaporization: