AP Biology PowerPoints
Biology 7th edition
By Neil A. Campbell, Jane B. Reece, Lawrence G. Mitchell
Copyright Pearson/Benjamin Cummings
AP Biology PowerPoints
Biology 7th edition
By Neil A. Campbell, Jane B. Reece, Lawrence G. Mitchell
Copyright Pearson/Benjamin Cummings
| Lab. 2 – Enzyme Catalysis |  |
Introduction:
This lab will observe the conversion of hydrogen peroxide to water and oxygen gas by the enzyme catalysis. The amount of oxygen generated will be measured and used to calculate the rate of the enzyme-catalized reaction. Enzymes are proteins produced by living cells. Enzymes act as biochemical catalysts during a reaction, meaning they lower the activation energy needed for that reaction to occur. Through enzyme activity, cells gain the ability to carry out complex chemical activities at relatively low temperatures. The substance in an enzyme-catalyzed reaction that is to be acted upon is the substrate, which binds reversibly to the active site of the enzyme. The active site is the portion of the enzyme that interacts with the substrate. One result of this temporary union between the substrate and the active site is a reduction in the activation energy required to start the reaction of the substrate molecule so that products are formed. In a mathematical equation of the substrate (S) binding with the activation site (E) and forming products (P) is:
E + S —> ES –> E + P
Several ways enzyme action may be affected include:
1) Salt Concentration — For example, if salt concentration is close to zero, the charged amino acid side chains of the enzyme molecules will attract each other. The enzyme will then denature and form an inactive precipitate. If salt concentration is extremely high, the normal interaction of charged groups will be blocked, new interactions will occur, and again the enzyme will precipitate. An intermediate salt concentration such as that of human blood (0.9%) is the optimum for many enzymes.
2) pH of the environment — . The pH of a solution is a logarithmic scale that measures the acidity or H+ concentration in a solution. The scale begins at 0, being the highest in acidity, and ends at 14, containing the least amount of acidity. As the pH is lowered an enzyme will tend to gain H+ ions, disrupting the enzyme’s shape. In turn, if the pH is raised, the enzyme will lose H+ ions and eventually lose its active shape.
3) Temperature — Usually, chemical reactions speed up as the temperature is raised. When the temperature is increased, more of the reacting molecules have enough kinetic energy to undergo the reaction. However, if the temperature goes past a temperature optimum, the conformation of the enzyme molecules is disrupted.
4) Activations and Inhibitors — Many molecules other than the substrate may interact with an enzyme. If such a molecule speeds up the reaction it is an activator, but if it slows the reaction down it is an inhibitor.
The enzyme used in this lab is catalase, which has four polypeptide chains that are composed of more than 500 amino acids each. One function of this enzyme is to prevent the accumulation of toxic levels of hydrogen peroxide formed as a by-product of metabolic processes. Catalase is also involved in some of the many oxidation reactions that occur in the cells of all living things. The primary reaction catalyzed by catalase is the decomposition of hydrogen peroxide to form water and oxygen.
2H2O2 ——-> 2 H2O + O2 (gas)
Without the help of catalase, this reaction occurs spontaneously, but very slowly. Catalase helps to speed up the reaction considerably. In this lab, a rate for this reaction will be determined.
Hypothesis:
The enzyme catalase, under optimum salt conditions, temperature, and pH level will speed up the reaction as it denatures the hydrogen peroxide at a higher rate than normal.
Materials:
Exercise 2A
For the first part of the lab, 10 mL of 1.5% H2O2, a 50-mL glass beaker, and 1 mL of fresh catalase are needed. At the second stage a test tube, a hot water bath, 5 mL of catalase, 10 mL of 1.5% H2O2 are needed. Finally, in the third part, a potato, and10 mL of 1.5% H2O2 are needed.
Exercise 2B
For this experiment, 10 mL of 1.5% H2O2, 1 mL of water, 10 mL of sulfuric acid, two 25 mL beakers, 5-10 mL syringe, potassium permanganate, lab aprons and trays are needed.
Exercise 2C
In this section of the experiment, 20 mL of 1.5% H2O2, two glass beakers, 1 mL of H2O, 10 mL of sulfuric acid, a 5 mL syringe, 5-10 mL of potassium permanganate, and lab aprons and trays are used.
Exercise 2D
In the final part of the lab, 6 plastic cups labeled 10, 30, 60, 120, 180, 360, 6 plastic cups labeled acid, 60 mL of 1.5% H2O2, a 50-mL beaker, 6 mL of catalase extract, two 5-mL syringes, potassium permanganate, a timer (clock), lab aprons and trays are needed.
Methods:
Exercise 2A
Transfer 10 mL of 1.5% H2O2 into a 50-mL glass beaker and add 1 mL of freshly made catalase solution. The fresh catalase should be kept on ice until ready to be used. Observe the reaction. Then transfer 5 mL of purified catalase extract to a test tube and place it in a hot water bath for five minutes. Transfer 10 mL of 1.5% H2O2 into a 50-mL beaker and add 1 mL of the boiled catalase solution, after it has cooled. Observe the changes in the reaction. To demonstrate the presence of catalase in living tissue, cut 1 cubic cm of potato, macerate it, and transfer it into a 50-mL glass beaker containing 10 mL of 1.5% H2O2. Observe the results.
Exercise 2B
Put 10 ml of 1.5% H2O2 into a clean glass beaker. Add 1 mL of H2O. Add 10 mL of sulfuric acid (1.0 M). USE EXTREME CARE IN HANDLING ACIDS. Mix the solution well. Remove a 5 mL sample. Place this 5 mL sample in another beaker, and assay for the amount of H2O2 as follows: Place the beaker containing the sample over white paper. Use a burette or 5 mL pipette to add potassium permanganate a drop at a time to the solution until a persistent pink or brown color is obtained. Remember to gently swirl the solution after adding each drop.
Exercise 2C
To determine the rate of spontaneous conversion of H2O2 to H2O and O2in an uncatalyzed reaction, put about 20 mL of 1.5% H2O2 in a beaker. Store it uncovered at room temperature for approximately 24 hours. Put 10 mL of 1.5% H2O2 into a clean glass beaker (using the uncatalyzed H2O2 that set out). Add 1 mL of H2O2 and then add 10 mL of sulfuric acid (1.0 M). Be careful when using acid. Mix this solution well. Remove a 5 mL sample and place it into another beaker. Assay for the amount of H2O2 as follows: Use a 5 mL syringe to add one drop of potassium permanganate at a time to the solution until it becomes a persistent pink or brown color. Gently swirl the solution after adding each drop. Record all results.
Exercise 2D
If a day or more has passed since Exercise B was performed, a baseline must be reestablish. Repeat the assay from Exercise B and record the results. Compare with other groups to check that results are similar. To determine the course of an enzymatic reaction, how much substrate is disappearing over time must be measured. The first thing to be done is to set up the six cups labeled with times, and the other six, one directly in front of each cup with a time on it. Then put 10 mL of 1.5% H2O2 into the cup marked 10 sec. Add 1 mL of catalase extract to this cup. Swirl gently for 10 seconds using a timer or clock for help. At 10 seconds, add 10 mL of sulfuric acid. Remove 5 mL and place in the cup directly in front of the cup marked 10 sec. Assay the 5 mL sample by adding one drop of potassium permanganate at a time until the solution turns a pink or brown. Repeat the previous steps, with clean cups using the times 30, 60, 120, 180, and 360. Record all results and observations.
Results:
Table 1
Enzyme Activity
| Activity | Observations |
Enzyme activity |
The reaction caused oxygen gas as a product, which made the wooden splint glow bright red. |
Effect of Extreme temperature
|
Boiling the catalase caused it to denature and it resulted in no bubbling in the solution. |
Presence of catalase |
The catalase being present in a living thing (potato), caused an extreme reaction with tons of products (02) produced. |
Table 2
Establishing a Baseline
| Volume | |
Initial reading
|
10 mL |
Final reading
|
6.7 mL |
| Baseline ( final volume – initial volume)
|
3.3 mL
Potassium permanganate |
Table 3
Rate of Hydrogen Peroxide Spontaneous Decomposition
| Volume | |
| Initial KMnO4
|
5 mL |
Final KMnO4
|
.3 mL |
| Amount of KMnO4 used after 24 hours
|
4.7 mL |
| Amount of H2O2 spontaneously decomposed
( ml baseline – ml after 24 hours) |
1.4 mL |
Percent of H2O2 spontaneously decomposed( ml baseline – ml after 24 hours/ baseline) |
57.6% |
Table 4
Reestablishing a Baseline
| Volume | |
Initial reading
|
5 mL |
Final reading
|
.8 mL |
| Baseline ( final volume – initial volume)
|
4.2 mL
Potassium permanganate |
Table 5
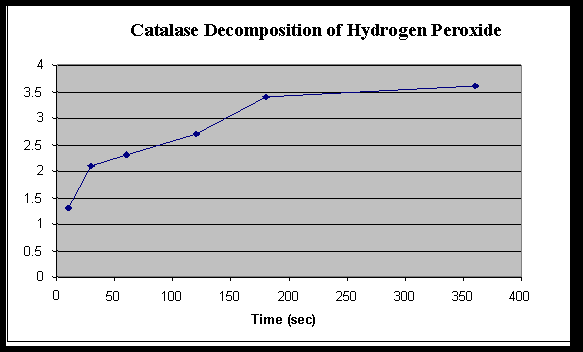
Rate of Hydrogen Peroxide Decomposition by Catalase
|
Time ( Seconds) |
||||||
| 10 | 30 | 60 | 120 | 180 | 360 | |
| Baseline KMnO4 | 4.2 | 4.2 | 4.2 | 4.2 | 4.2 | 4.2 |
| Initial volume KMnO4
|
10 | 10 | 10 | 10 | 10 | 10 |
| Final volume KMnO4
|
7.1 | 7.9 | 8.1 | 8.5 | 9.2 | 9.4 |
| Amount KMnO4 used
(baseline – final) |
2.9 | 2.1 | 1.9 | 1.5 | .8 | .6 |
Amount H2O2 used(KMnO4 – initial)
|
1.3 | 2.1 | 2.3 | 2.7 | 3.4 | 3.6 |
Graph 1
![]()
Exercise 2A
1. a) What is the enzyme in this reaction? Catalase is the enzyme in the reaction.
b) What is the substrate in this reaction? The substrate is hydrogen peroxide.
c) What is the product in this reaction? The products are oxygen (gas) and water.
d) How could you show that the gas evolved is oxygen? Using the example of holding a burning wooden splint over the reaction, the splint glows bright red, therefore showing that oxygen is being let out of the solution.
2. How does the reaction compare to the one using the unboiled catalase? Explain the reason for this difference. When the boiled catalase was used, there was no bubbling in the solution, which proved that there was no reaction occurring because the extreme heat had denatured the catalase.
3. What do you observe? What do you think would happen if the potato or liver was boiled before being added to the hydrogen peroxide? The catalase shows a lot of reaction with the potato, causing many bubbles to form in the solution. Also, if the potato were boiled there wouldn’t be any bubbles, because the heat would denature the potato.
Analysis of Results
1. Determine the initial rate of the reaction and the rates between each of the time points. Record the rates in the table below.
| Time Intervals (seconds) | ||||||
| Initial to 10 | 10 to 30 | 30 to 60 | 60 to 120 | 120 to 180 | 180 to 360 | |
| Rates | .13 | .04 | .007 | .007 | .0012 | .0011 |
2. When is the rate the highest? Explain why. The rate is the highest at the beginning of the reaction, because the hydrogen peroxide had been exposed to the air for the least amount of time.
3. When is the rate the lowest? For what reasons is the rate low? At the longer times the rate was the lowest because the peroxide had been exposed to the air longer.
4. Explain the inhibiting effect of sulfuric acid on the function of catalase. Relate this to enzyme structure and chemistry. The sulfuric acid lowered the pH level of the solution, which caused the catalase to denature by gaining hydrogen ions and it stopped the reaction immediately.
5. Predict the effect lowering the temperature would have on the rate of enzyme activity. Explain you prediction. Enzymes work best at optimum temperature, therefore increasing, or in this case, decreasing the temperature would extremely change the rate of the reaction. Lowering the temperature would cause the reaction to slow down.
6. Design a controlled experiment to test the effect of varying pH, temperature, or enzyme concentration. Since the results of room temperature and heated have already been recorded, using catalase that was completely frozen would test the other end of the spectrum as far as temperature goes.
Error Analysis:
Any errors occurring in this experiment could have been caused by misreading of a syringe, miscalculating the data on the tables, or when the 1.5% H2O2 was mixed that was used in almost all parts of the experiment. Also, in Exercise 2D, if the getting the timing just right with all parts of the lab were a source of error in the experiment.
Discussion and Conclusion:
The main purpose of this lab was to show how enzymes can be affected in reactions with other substances by factors such as pH, temperature, and exposure to the surrounding environment. This lab proved that an extreme increase in temperature (boiling) can cause absolutely no reaction by using the boiled enzyme catalase. Also, by using a potato, it shows that catalase is speeding up the decomposition of hydrogen peroxide in living things, helping all living things survive another day.
| Lab 5 Cellular Respiration |
Introduction:
Cellular respiration is an ATP-producing catabolic process in which the ultimate electron acceptor is an inorganic molecule, such as oxygen. It is the release of energy from organic compounds by metabolic chemical oxidation in the mitochondria within each cell. Carbohydrates, proteins, and fats can all be metabolized as fuel, but cellular respiration is most often described as the oxidation of glucose, as follows:
C6H12O6 + 6O2 → 6CO2 + 6H2O + 686 kilocalories of energy/mole of glucose oxidized
Cellular respiration involves glycolysis, the Krebs cycle, and the electron transport chain. Glycolysis is a catabolic pathway that occurs in the cytosol and partially oxidizes glucose into two pyruvate (3-C). The Krebs cycle is also a catabolic pathway that occurs in the mitochondrial matrix and completes glucose oxidation by breaking down a pyruvate derivative (Acetyl-CoA) into carbon dioxide. These two cycles both produce a small amount of ATP by substrate-level phosphorylation and NADH by transferring electrons from substrate to NAD+ (Krebs cycle also produces FADH2 by transferring electrons to FAD). The electron transport chain is located at the inner membrane of the mitochondrion, accepts energized electrons from reduced coenzymes that are harvested during glycolysis and Krebs cycle, and couples this exergonic slide of electrons to ATP synthesis or oxidative phosphorylation. This process produces 90% of the ATP.
Cells respond to changing metabolic needs by controlling reaction rates. Anabolic pathways are switched off when their products are in ample supply. The most common mechanism of control is feedback inhibition. Catabolic pathways, such as glycolysis and the Krebs cycle, are controlled by regulating enzyme activity at strategic points. A key control point of catabolism is the third step of glycolysis, which is catalyzed by an allosteric enzyme, phosphofructokinase. The ratio of ATP to ADP and AMP reflects the energy status of the cell, and phosphofructokinase is sensitive to changes in this ratio. Citrate and ATP are allosteric inhibitors of phosphofructokinase, so when their concentration rise, the enzyme slows glycolysis. As the rate of glycolysis slows, the Krebs cycle also slows since the supply of Acetyl-CoA is reduced. This synchronizes the rates of glycolysis and the Krebs cycle. ADP and AMP are allosteric activators for phosphofructokinase, so when their concentrations relative to ATP rise, the enzyme speeds up glycolysis, which speeds of the Krebs cycle.
Cellular respiration is measure in three manners: the consumption of O2 (how many moles of O2 are consumed in cellular respiration?), production of CO2 (how many moles of CO2 are produced in cellular respiration?), and the release of energy during cellular respiration.
PV = nRT is the formula for the inert gas law, where P is the pressure of the gas, V is the volume of the gas, n is the number of molecules of gas, R is the gas constant, and T is the temperature of the gas in degrees K. This law implies several important things about gases. If temperature and pressure are kept constant then the volume of the gas is directly proportional to the number of molecules of the gas. If the temperature and volume remain constant, then the pressure of the gas changes in direct proportion to the number of molecules of gas. If the number of gas molecules and the temperature remain constant, then the pressure is inversely proportional to the volume. If the temperature changes and the number of gas molecules is kept constant, then either pressure or volume or both will change in direct proportion to the temperature.
Hypothesis:
The respirometer with only germinating peas will consume the largest amount of oxygen and will convert the largest amount of CO2 into K2CO3 than the respirometers with beads and dry peas and with beads alone. The temperature of the water baths directly effects the rate of oxygen consumption by the contents in the respirometers (the higher the temperature, the higher the rate of consumption).
Materials:
The following materials are necessary for the lab: 2 thermometers, 2 shallow baths, tap water, ice, paper towels, masking tape, germinating peas, non-germinating (dry) peas, glass beads, 100 mL graduated cylinder, 6 vials, 6 rubber stoppers, absorbent and non- absorbent cotton, KOH, a 5-mL pipette, silicon glue, paper, pencil, a timer, and 6 washers.
Methods:
Prepare a room temperature and a 10oC water bath. Time to adjust the temperature of each bath will be necessary. Add ice cubes to one bath until the desired temperature of 10oC is obtained.
Fill a 100 mL graduated cylinder with 50 mL of water. Add 25 germinating peas and determine the amount of water that is displaced. Record this volume of the 25 germinating peas, then remove the peas and place them on a paper towel. They will be used for respirometer 1. Next, refill the graduated cylinder with 50 mL of water and add 25 non-germinating peas to it. Add glass beads to the graduated cylinder until the volume is equivalent to that of the expanded germinating peas. Remove the beads and peas and place on a paper towel. They will be used in respirometer 2. Now, refill the graduated cylinder with 50 mL of water. Determine how many glass beads would be required to attain a volume that is equivalent to that of the germinating peas. Remove the beads. They will be used in respirometer 3. Then repeat the procedures used above to prepare a second set of germinating peas, dry peas and beads, and beads to be used in respirometers 4,5,and 6.
Assemble the six respirometers by obtaining 6 vials, each with an attached stopper and pipette. Then place a small wad of absorbent cotton in the bottom of each vial and, using the pipette or syringe, saturate the cotton with 15 % KOH. Be sure not to get the KOH on the sides of the respirometer. Then place a small wad of non-absorbent cotton on top of the KOH-soaked absorbent cotton. Repeat these steps to make the other five respirometers. It is important to use about the same amount of cotton and KOH in each vial.
Next, place the first set of germinating peas, dry peas and beads and beads alone in vials 1,2, and 3. Place the second set of germinating peas, dry peas and beads, and glass beads in vials 4,5, and 6. Insert the stoppers in each vial with the proper pipette. Place a washer on each of the pipettes to be used as a weight.
| Respirometer | Temperature | Contents |
| 1 | Room | Germinating Peas |
| 2 | Room | Dry Seeds + Beads |
| 3 | Room | Beads |
| 4 | 10oC | Germinating Peas |
| 5 | 10oC | Dry Seeds + Beads |
| 6 | 10oC | Beads |
Make a sling using masking tape and attach it to each side of the water baths to hold the pipettes out of the water during the equilibration period of 10 minutes. Vials 1,2, and 3 should be in the bath containing water at room temperature. Vials 4, 5, and 6 should be in the bath containing water that is 10oC. After the equilibration period, immerse all six respirometers into the water completely. Water will enter the pipette for a short distance and stop. If the water does not stop, there is a leak. Make sure the pipettes are facing a direction from where you can read them. The vials should not be shifted during the experiment and your hands should not be placed in the water during the experiment.
Allow the respirometers to equilibrate for three more minutes and then record the initial water reading in each pipette at time 0. Check the temperature in both baths and record the data. Every five minutes for 20 minutes take readings of the water’s position in each pipette, and record.
Results:
Table 1: Measurement of O2 Consumption by Soaked and Dry Pea Seeds at Room Temperature and 10˚C Using Volumetric Methods
|
Beads Alone |
Germinating Peas |
Dry Peas and Beads |
|||||||||||||||
|
Reading at time X |
Diff. |
Reading at time X |
Diff. |
Corrected Diff.∆ |
Reading at time X |
Diff. |
Corrected Diff.∆ |
||||||||||
|
Initial-0 |
1.38 | 1.35 | 1.47 | ||||||||||||||
|
0-5 |
1.38 | 0 | 1.16 | .19 | .19 | 1.46 | .01 | .01 | |||||||||
|
5-10 |
1.38 | 0 | 1.04 | .31 | .31 | 1.44 | .03 | .03 | |||||||||
|
10-15 |
1.38 | 0 | 0.93 | .42 | .42 | 1.43 | .04 | .04 | |||||||||
|
15-20 |
1.38 | 0 | 0.57 | .78 | .78 | 1.42 | .05 | .05 | |||||||||
|
Initial-0 |
1.40 | 1.32 | 1.40 | ||||||||||||||
|
0-5 |
1.39 | .01 | 1.20 | .12 | .11 | 1.40 | 0 | .01 | |||||||||
|
5-10 |
1.38 | .02 | 1.11 | .21 | .19 | 1.40 | 0 | .02 | |||||||||
|
10-15 |
1.38 | .02 | 1.00 | .32 | .30 | 1.39 | .01 | .01 | |||||||||
|
15-20 |
1.38 | .02 | 0.95 | .37 | .93 | 1.38 | .02 | 0 | |||||||||
In this activity, you are investigating both the effects of germination versus non-germination and warm temperature versus cold temperature on respiration rate. Identify the hypothesis being tested on this activity. The rate of cellular respiration is higher in the germinating peas in cold than in the beads or non-germinating peas; the cooler temperature in the cold water baths slows the process of cellular respiration in the both germinating and non-germinating peas.

This activity uses a number of controls. Identify at least three of the controls, and describe the purpose of each. The constant temperature in the water baths yielding stable readings, the unvarying volume of KOH from vial to vial leading to equal amounts of carbon dioxide consumption, identical equilibration periods for all the respirometers, precise time intervals between measurements, and glass beads acting as a control for barometric pressure all served as controls.
Describe and explain the relationship between the amount of oxygen consumed and time. There was a constant, gradual incline in the amount of oxygen consumed over precise passage of time.
|
Condition |
Calculations |
Rate in mL O2/ minute |
|
Germinating Peas/ 10 oC
|
(1.40-1.38)
20 min. |
.001 |
|
Germinating Peas/ 20 oC
|
(1.35-.57)
20 min. |
.040 |
|
Dry Peas/ 10 oC
|
(1.40-1.38)
20 min. |
.001 |
|
Dry Peas/ 20 oC |
(1.47-1.42)
20 min. |
.003 |
Why is it necessary to correct the readings from the peas with the readings from the beads? The beads served as a control variable, therefore, the beads experienced no change in gas volume.
Explain the effects of germination (versus non-germination) on pea seed respiration. The germinating seeds have a higher metabolic rate and needed more oxygen for growth and survival. The non-germinating peas, though alive, needed to consume far less oxygen for continued subsistence.
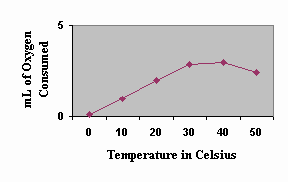
Above is a sample graph of possible data obtained for oxygen consumption by germinating peas up to about 8 oC. Draw in predicted results through 45 oC. Explain your prediction. Once the temperature reached a certain point, the enzymes necessary for cellular respiration denatured and germination (and large amounts of oxygen consumption) was inhibited.
What is the purpose of KOH in this experiment? The KOH drops absorbed the carbon dioxide and caused it to precipitate at the bottom of the vial and no longer able to effect the readings.
Why did the vial have to be completely sealed under the stopper? The stopper at the top of the vial had to be completely sealed so that no gas could leak out of the vial and no water would be allowed into the vial.
If you used the same experimental design to compare the rates of respiration of a 35g mammal at 10 oC, what results would you expect? Explain your reasoning. Respiration would be higher in the mammal since they are warm-blooded and endothermic.
If respiration in a small mammal were studied at both room temperature (21 oC) and 10 oC, what results would you predict? Explain your reasoning. Respiration would be higher at 21 degrees because it would be necessary for the animal to maintain a higher body temperature. The results would proliferate at 10 degrees because the mammal would be required to retain its body temperature at an even lower temperature in comparison to room temperature.
Explain why water moved into the respirometer pipettes. While the peas underwent cellular respiration, they consumed oxygen and released carbon dioxide, which reacted with the KOH in the vial, resulting in a decrease of gas in the pipette. The water moved into the pipette because the vial and pipette were completely submerged into the bath.
Design an experiment to examine the rates of cellular respiration in peas that have been germinating for 0, 24, 48, and 72 hours. What results would you expect? Why? Respirometers could be set up with respirometer 1 containing non-germinating peas, respirometer 2 holding peas that have been germinating 24 hours, 3 would contain the peas that germinated 48 hours, and 4 would hold the peas that germinated 72 hours. All the respirometers should have the KOH added to the bottom in the same manner as in lab described earlier. The respirometers should be placed in baths with the same temperature for all the respirometers. The seeds that have not begun germination would consume very little oxygen. The peas that have been germinating for 72 hours will have the greatest amount of oxygen consumption, while the other two samples will consume a medium (in comparison to respirometers 1 and 4 results) amount of oxygen.
Error Analysis:
Numerous errors could have occurred throughout the lab. The temperature of the baths may have been allowed to fluctuate, the amounts of peas, beads, KOH, and cotton may have varied from vial to vial damaging the results, and these problems would have occurred only during set up. Air may have been allowed to creep into the vial via a leaky stopper or poorly sealed pipette. Timing for the equilibration of the respirometers and the five-minute time intervals may have been erroneous. It was somewhat difficult to read the markings on the pipettes and so errors are always likely. Mathematical inaccuracies may have taken place when filling out the table and finding the corrected difference by using the formula provided.
Discussion and Conclusion:
The lab and the results gained from this lab demonstrated many important things relating to cellular respiration. It showed that the rates of cellular respiration are greater in germinating peas than in non-germinating peas. It also showed that temperature and respiration rates are directly proportional; as temperature increases, respiration rates increase as well. Because of this fact, the peas contained by the respirometers placed in the water at 10 oC carried on cellular respiration at a lower rate than the peas in respirometers placed in the room temperature water. The non-germinating peas consumed far less oxygen than the germinating peas. This is because, though germinating and non-germinating peas are both alive, germinating peas require a larger amount of oxygen to be consumed so that the seed will continue to grow and survive.
In the lab, CO2 made during cellular respiration was removed by the potassium hydroxide (KOH) and created potassium carbonate (K2CO3). It was necessary that the carbon dioxide be removed so that the change in the volume of gas in the respirometer was directly proportional to the amount of oxygen that was consumed. In the experiment water will moved toward the region of lower pressure. During respiration, oxygen will be consumed and its volume will be reduced to a solid. The result was a decrease in gas volume within the tube, and a related decrease in pressure in the tube. The respirometer with just the glass beads served as a control, allowing changes in volume due to changes in atmospheric pressure and/or temperature.
1. A rooster with gray feathers is mated with a hen of the same phenotype. Among their offspring, 15 chicks are gray, 6 are black, and 8 are white.
2. In some plants, a true-breeding, red-flowered strain gives all pink flowers when crossed with a white-flowered strain: RR (red) x rr (white) —> Rr (pink). If flower position (axial or terminal) is inherited as it is in peas what will be the ratios of genotypes and phenotypes of the generation resulting from the following cross: axial-red (true-breeding) x terminal-white? What will be the ratios in the F2 generation?
3. Flower position, stem length, and seed shape were three characters that Mendel studied. Each is controlled by an independently assorting gene and has dominant and recessive expression as follows:
| Character | Dominant | Recessive |
| Flower position | Axial (A ) | Terminal (a ) |
| Stem length | Tall (T ) | Dwarf (t ) |
| Seed shape | Round (R ) | Wrinkled (r) |
If a plant that is heterozygous for all three characters were allowed to self-fertilize, what proportion of the offspring would be expected to be as follows: (Note – use the rules of probability (and show your work) instead of huge Punnett squares)
4. A black guinea pig crossed with an albino guinea pig produced 12 black offspring. When the albino was crossed with a second one, 7 blacks and 5 albinos were obtained.
5. In sesame plants, the one-pod condition (P ) is dominant to the three-pod condition (p ), and normal leaf (L ) is dominant to wrinkled leaf (l) . Pod type and leaf type are inherited independently. Determine the genotypes for the two parents for all possible matings producing the following offspring:
6. A man with group A blood marries a woman with group B blood. Their child has group O blood.
7. Color pattern in a species of duck is determined by one gene with three alleles. Alleles H and I are codominant, and allele i is recessive to both. How many phenotypes are possible in a flock of ducks that contains all the possible combinations of these three alleles?
8. Phenylketonuria (PKU) is an inherited disease caused by a recessive allele. If a woman and her husband are both carriers, what is the probability of each of the following?
(Note: Remember that the probabilities of all possible outcomes always add up to 1)
9. The genotype of F1 individuals in a tetrahybrid cross is AaBbCcDd. Assuming independent assortment of these four genes, what are the probabilities that F2 offspring would have the following genotypes?
10. In 1981, a stray black cat with unusual rounded curled-back ears was adopted by a family in California. Hundreds of descendants of the cat have since been born, and cat fanciers hope to develop the “curl” cat into a show breed. Suppose you owned the first curl cat and wanted to develop a true breeding variety.
11. What is the probability that each of the following pairs of parents will produce the indicated offspring (assume independent assortment of all gene pairs?
12. Karen and Steve each have a sibling with sickle-cell disease. Neither Karen, Steve, nor any of their parents has the disease, and none of them has been tested to reveal sickle-cell trait. Based on this incomplete information, calculate the probability that if this couple should have another child, the child will have sickle-cell anemia.
13. Imagine that a newly discovered, recessively inherited disease is expressed only in individuals with type O blood, although the disease and blood group are independently inherited. A normal man with type A blood and a normal woman with type B blood have already had one child with the disease. The woman is now pregnant for a second time. What is the probability that the second child will also have the disease? Assume both parents are heterozygous for the “disease” gene.
14. In tigers, a recessive allele causes an absence of fur pigmentation (a “white tiger”) and a cross-eyed condition. If two phenotypically normal tigers that are heterozygous at this locus are mated, what percentage of their offspring will be cross-eyed? What percentage will be white?
15. In corn plants, a dominant allele I inhibits kernel color, while the recessive allele i permits color when homozygous. At a different locus, the dominant gene P causes purple kernel color, while the homozygous recessive genotype pp causes red kernels. If plants heterozygous at both loci are crossed, what will be the phenotypic ratio of the F1 generation?
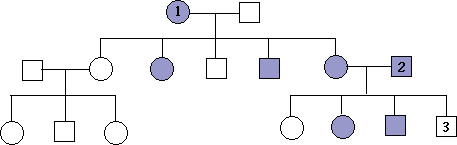
16. The pedigree below traces the inheritance of alkaptonuria, a biochemical disorder. Affected individuals, indicated here by the filled-in circles and squares, are unable to break down a substance called alkapton, which colors the urine and stains body tissues. Does alkaptonuria appear to be caused by a dominant or recessive allele? Fill in the genotypes of the individuals whose genotypes you know. What genotypes are possible for each of the other individuals?
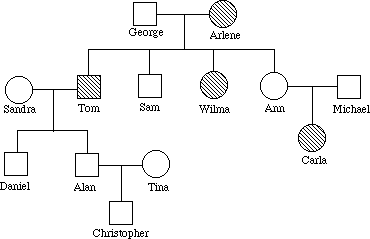
18. Imagine you are a genetic counselor, and a couple planning to start a family came to you for information. Charles was married once before, and he and his first wife had a child who has cystic fibrosis. The brother of his current wife Elaine died of cystic fibrosis. What is the probability that Charles and Elaine will have a baby with cystic fibrosis? (Neither Charles nor Elaine has the disease)
19. In mice, black color (B ) is dominant to white (b ). At a different locus, a dominant allele (A ) produces a band of yellow just below the tip of each hair in mice with black fur. This gives a frosted appearance known as agouti. Expression of the recessive allele (a ) results in a solid coat color. If mice that are heterozygous at both loci are crossed, what will be the expected phenotypic ratio of their offspring?
20. The pedigree below traces the inheritance of a vary rare biochemical disorder in humans. Affected individuals are indicated by filled-in circles and squares. Is the allele for this disorder dominant or recessive? What genotypes are possible for the individuals marked 1, 2, and 3.