| Chapter 2 Chemical Context of Life | |
| Objectives | |
Elements and Compounds
Atoms and Molecules
|
|
| BACK | |
| Chapter 2 Chemical Context of Life | |
| Objectives | |
Elements and Compounds
Atoms and Molecules
|
|
| BACK | |
Chemistry
All Materials © Cmassengale
Composition of Matter
Ø Everything in the universe is made of matter
Ø Matter takes up space & has mass
Ø Mass is a measure of the amount of matter in the substance
Ø Mass & weight are NOT the same
Ø Weight is a measure of the pull of gravity on an object
Question: Is the mass of an object the same on the moon as it is on the Earth? Is its weight the same? (Hint: Gravitational pull on the moon is 1/6 of that on the Earth.)
Ø Matter exists in 4 states – solid, liquid, gas, & plasma

Ø Solids have both a definite volume & definite shape (rock)
Ø Liquids have a definite volume but no definite shape; they can be poured (water)
Ø Gases do not have a definite volume or definite shape, but they take the volume & shape of their container
Ø Plasmas have no definite volume, no definite shape, and only exist at extremely high temperatures such as the sun
Ø Chemical Changes in matter are essential to all life processes
Ø Biologists study chemistry because all living things are made of the same kinds of matter that make up nonliving things
Elements
Ø Elements are pure substances which cannot be chemically broken down into simpler kinds of matter
Ø More than 100 elements have been identified, but only about 30 are important in living things
Ø All of the Elements are arranged on a chart known as the Periodic Table
Ø Periodic charts tell the atomic number, atomic mass, & chemical symbol for every element
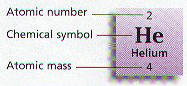
Ø Four elements, Carbon – C, Hydrogen – H, Oxygen – O, and Nitrogen – N make up almost 90% of the mass of living things
Ø Every element has a different chemical symbol composed of one to two letters
Ø Chemical symbols usually come from the first letter or letters of an element like C for Carbon and Cl for Chlorine
Ø Some chemical symbols come form their Latin or Greek name such as Na for Sodium (natrium) or K for Potassium (Kalium)
Ø Elements in the same horizontal period on the periodic table have the same number of energy levels (e.g. H & He in period 1 have only a K energy level)
![[Periodic Table]](https://biologyjunction.com/images/periodic.jpg)
All Period 2 elements have 2 energy levels
(K & L)
Ø Elements in the same vertical Family on the periodic table have the same number of electrons in their outermost energy level & react similar (e.g. Family IV, the Carbon family all have 4 electrons in their outermost energy level)
Atoms
Ø Atoms are the simplest part of an element that keeps all of the element’s properties
Ø Atoms are too small to be seen so scientists have developed models that show their structure & properties
Ø Atoms consist of 3 kinds of subatomic particles – protons & neutrons in the center or nucleus, and electrons spinning in energy levels around the center
Ø The nucleus is the center of an atom where most of the mass is concentrated
Ø Protons are positively charged ( p+ ), have a mass of 1 amu (atomic mass unit) , are found in the nucleus, and determine the atomic number of the element
Example: Carbon has 6 protons so its atomic number is 6
Ø Neutrons are neutral or have no electrical charge (n), have a mass of 1 amu, are found in the nucleus, and when added to the number of protons, determine the atomic mass of the element
Example: Sodium has 11 protons and 12 neutrons so its atomic mass is 11+12=23 amu
Ø Electrons (e-) are negatively charged, high energy particles with little mass that spin around the nucleus in energy levels
Ø Seven energy levels (K, L, M, N, O, P, & Q) exist around the nucleus and each holds a certain number of electrons
Ø The K energy level is closest to the nucleus & only holds 2 electrons, while the L – Q energy levels can hold 8 electrons

Ø Electrons in outer energy level are traveling faster & contain more energy than electrons in inner levels

Ø The number of protons (positive charges) and electrons (negative charges in an atom are equal so the net electrical charge on a atom is zero making it electrically neutral
Ø Stable or non-reactive atoms have an outer energy level that is filled with electrons

Compounds
Ø Most elements do not exist by themselves; Most elements combine with other elements
Ø Compounds are made of atoms of two or more elements chemically combined
Ø Chemical Formulas represent a compound & show the kind & number of atoms of each element (e.g. H2O has 2 hydrogen & 1 oxygen)
Ø Compounds have different physical & chemical properties than the atoms that compose them (e.g. hydrogen & oxygen are gases but H2O is a liquid)
Ø The number & arrangement of electrons in an atom determines if it will combine to form compounds
Ø Chemical reactions occur whenever unstable atoms (outer energy level not filled) combine to form more stable compounds
Ø Chemical bonds form between atoms during chemical reactions
Types of Chemical Bonds
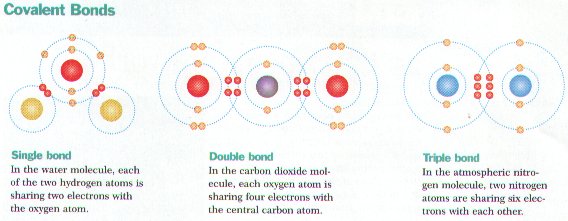
Ø Covalent bonds form between atoms whenever they share 1 or more pairs of electrons (e.g. H2O)
Ø Molecules form from covalent bonding & are the simplest part of a compound (e.g. NaCl, H2O, O2)
Ø Ionic bonding occurs between a positively & negatively charged atom or ion
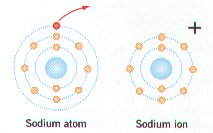
Ø Positively charged ions have more electrons (-) than protons (+); negatively charged ions have more protons than electrons
Ø Table salt (NaCl) forms when the 1 outer electron of Na is transferred to the outer energy level of chlorine that has 7 electrons (e-)
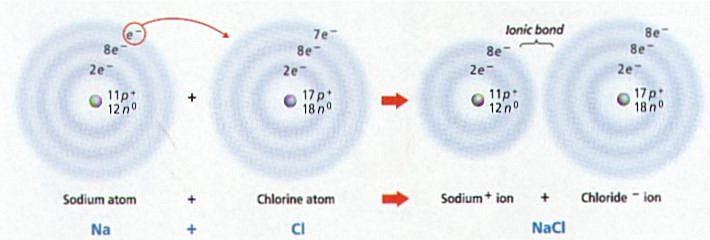
Ø Sodium (Na) with 1 less e- becomes positively charged, while Chlorine (Cl) with 1 more e- becomes negatively charged; the + and – charges attract & form the ionic bond holding NaCl together
Ø Other types of chemical bonding include hydrogen bonding
Energy
Ø Energy is the ability to do work
Ø Energy occurs in several forms & may be converted from one form to another
Ø Sunlight is the ultimate energy for all life on earth
Ø Forms of energy include chemical, electrical, mechanical, thermal, light, & sound
Ø Free energy is the energy available for work (e.g. cells have energy to carry out cell processes)
Ø Cells convert the chemical energy stored in food into other types of energy such as thermal & mechanical
Ø Energy is used to change matter form one state into another (e.g. liquid into a gas)
Chemical Reactions
Ø Living things undergo thousands of chemical reactions
Ø Chemical equations represent chemical reactions
Ø CO2 + H20—–goes to—–H2CO3 (carbonic acid) is a sample Chemical Reaction in living things
Ø Reactants are on the left side of the equation, while products are on the right side

Ø Activation energy is required to start many reactions

Ø Chemical bonds are broken, atoms rearranged, and new bonds form in chemical reaction
Ø Plants use sunlight to produce sugars such as C6H12O6 glucose; the chemical energy from the sun is stored in the chemical bonds of glucose
Ø Organisms eat plants, break down the sugars, and release energy along with CO2 & H2O
Ø Exergonic reactions involve a net release of energy; while endergonic reactions involve a net absorption of energy
Ø Energy must be added to the reactants for most chemical reactions to occur; called activation energy
Ø Enzymes are chemical substances in living things that act as catalysts & reduce the amount of activation energy needed
Ø Organisms contain thousands of different enzymes
Ø Most enzymes end with –ase (e.g. lipase is the enzyme that acts on lipids)

Reduction-Oxidation (Redox) reactions
Ø Reactions in which e- are transferred between atoms is a redox or reduction-oxidation reaction (e.g. formation of table salt NaCl)
Ø In oxidation reactions, a reactant loses 1 or more e- & becomes positively (+) charged (e.g. Sodium atom becomes a Na+ ion)
Ø In a reduction reaction, a reactant gains 1 or more e- and becomes negatively (-) charged (e.g. Chlorine atom becomes a Cl- ion)
Ø REDOX reactions always occur together; the electron(s) from the oxidation reaction are then accepted by another substance in the reduction reaction
Solutions
Ø A large percentage of the mass of organisms is water & many of the chemical reactions of life occur in water
Ø A solution is a uniform mixture of one substance in anther
Ø Solutions may be mixtures of solids, liquids, or gases
Ø The solute is the substance uniformly dissolved in the solution & may be ions, molecules, or atoms
Ø The solvent is the substance in which the solute is dissolved
Ø Water is known as the universal solvent
Ø Dissolving one substance in another does not alter their chemical properties
Ø The concentration of a solution is a measure of the amount of solute dissolved in a given volume of solvent
Ø Increasing the amount of solute increases the solution’s concentration
Ø Aqueous solutions are solutions in which water is the solvent; these are important in living things (e.g. blood, cytoplasm of cell…)
Acids and Bases
Ø The degree of acidity or alkalinity (basic) is important in organisms
Ø The force of attraction between molecules is so strong that the oxygen atom of one molecule can actually remove the hydrogen from other water molecules; called Dissociation
Ø H20—–GOES TO—– H+ + OH-
Ø OH- called hydroxide ion; H+ called hydrogen ion
Ø Free H+ ion can react with another water molecule to form H3O+ (hydronium ion)
Ø Acidity or alkalinity is a measure of the relative amount of H+ and OH- ions dissolved in a solution
Ø Neutral solutions have an equal number of H+ and OH- ions
Ø Acids have more H3O+ ions than OH- ions; taste sour; and can be corrosive
Ø Bases contain more OH- ions than H3O+ ions; taste bitter; & feel slippery
Examples of Common Acids
|
Examples of Common Bases
|
PH Scale
Ø Compares the relative concentration of H3O+ ions and OH- ions
Ø Scale ranges from 0 to 14; 0-3 is very acidic; 7 is neutral; 11-14 is very basic or alkaline

Ø Litmus paper, phenolphthalein, pH paper, & other indicators that change color can be used to measure pH

Buffers
Ø Control of pH is important to organisms
Ø Enzymes function only within a narrow pH range; usually neutral
Ø Buffers neutral acids or bases in organisms to help control pH
| Chemistry Study Guide | Chemistry On-line |
| BACK |
| Chemistry Worksheet |  |
Section 2-1 Composition of Matter
1. Define matter.
2. Define mass.
3. Explain the difference between mass & weight.
4. Why do biologists study chemistry?
5. Define element.
6. Name the 4 elements that make up 90% of the mass of living things. Give the symbol for each of these elements.
7. Explain why some elements such as sodium have odd symbols.
8. Sketch a block from the periodic table and label the atomic number, atomic mass, & symbol for the element.
9. Define atom and tell whether they can be seen.
10. What is the center of an atom called & what 2 subatomic particles are found there?
11. How does the charge of a proton differ from the charge of a neutron?
12. Where is most of the mass of an atom concentrated?
13. How is the atomic number of an element determined?
14. What is the charge on an electron?
15. Explain why the overall or net charge on an atom is zero.
16. Where are electrons found in an atom & describe their movement?
17. In which energy levels do the electrons have more energy?
18. How many electrons can these energy levels hold — a. first? b. second?
19. Define compound and write a formula for water, carbon dioxide, & sodium chloride (table salt).
20. Do compounds have the same chemical properties as the elements that compose them?
21. When would an atom be chemically stable (not react)?
22. What occurs in a chemical reaction?
23. What is a covalent bond?
24. Define molecule.
25. Give an example of a gas that exists as a molecule.
26. Define ionic bond.
27. What is an ion?
28. Name a compound formed from — a. covalent bonding? b. ionic bonding?
29. If electrons are shared, a(n) ______________ compound forms.
30. If electrons are transferred, a(n) _____________ compound forms.
31. Forming ionic or covalent bonds helps make atoms more ________________.
Section 2-2 Energy 
32. All living things require _____________ to do work.
33. Energy can’t be created or _____________ in a chemical reaction, but it can be _____________ from one form into another.
34. Name 4 forms of energy important to living things.
35. What is free energy?
36. Give an example of energy changing form in an organism.
37. Atoms & molecules are in constant _______________.
38. Name the 3 main states of matter.
39. Explain how the shape and volume of a solid, liquid, and gas differ.
40. Organisms undergo thousands of ____________ as part of their life processes.
41. Where are the reactants and products in a chemical equation?
42. What does a two-direction arrow mean in a chemical equation?
43. _______________ are broken down in chemical reactions in your body to release ___________ and produce _______________ and ______________.
44. What is the difference between an endergonic & exergonic reaction?
45. What is activation energy?
46. What effect does a catalyst have on activation energy?
47. What are biological catalysts called?
48. Redox is the abbreviation for what type of reaction?
49. Redox reactions involve the transfer of energy and _________ between atoms.
50. What happens during oxidation?
51. What happens during reduction?
52. Give an example of oxidation.
53. Give an example of reduction.
Section 2-3 Solutions
54. Many of the chemical reactions in organisms take place in __________.
55. What is a solution?
56. Give an example of a complex solution in your body.
57. Name & describe the 2 parts of a solution.
58. What is meant by concentration of the solution?
59. How do you get a saturated solution?
60. What are aqueous solutions?
61. Explain dissociation of water molecules.
62. Name and give the charge for the 2 ions formed whenever water dissociates.
63. Write the final equation for the dissociation of water.
64. What is the hydronium ion?
65. How are acidity and alkalinity measured?
66. When would a solution be neutral? Give an example of a neutral solution.
67. When would solutions be considered as acidic?
68. Acids have what taste?
69. Acids form what ion in water?
70. Give an example of an acid in your stomach.
71. When would solutions be considered as a base?
72. What adjective refers to basic solutions?
73. Give an example of a base.
74. What ion forms whenever a base is dissolved in water?
75. How does a base taste and feel?
76. How is soap made?
77. What is the pH scale used for?
78. What is the range for the pH scale?
79. At what pH would you find each of these solutions on a pH scale: a. acids? b. Bases? c. neutral?
80. How many times stronger is a pH of 3 than a pH of 5?
81. A change of one pH unit reflects a __________ change.
82. Why is controlling the pH range important to organisms?
83. How do organisms control their pH levels?
84. What is a buffer?
85. Give an example of a human body fluid that is: a. acidic? b. alkaline?
| Student Name Date Period |
| Chapter 2 Chemistry Outline | |
| I. Composition of Matter | |
| A. Matter | |
| * 1.1 1. Makes up everything | |
| 2. Occupies space & has mass | |
| 3. Mass – quantity of matter making up an object | |
| 4. Pull of gravity produces weight | |
| 5. Chemical changes in matter necessary for life processes | |
| B. Elements and Atoms | |
| 1. Pure substances that can’t be chemically broken down | |
| 2. about 30 elements important to organisms | |
| 3. Most common elements are C, H, O, N | |
| 4. Symbols (1-2 letters) stand for elements | |
| 5. May use Latin or Greek e.g. Na – sodium (natrium) | |
| *1.2 6. Simplest part of an element with the same properties | |
 |
|
| C. Nucleus | |
| 1. Most of the mass | |
| 2. Central part of an atom | |
| 3. | |
 |
|
Chemistry PowerPoint Notes 
1. What makes up everything in the universe?
2. Name 2 properties of all matter.
3. Define mass.
4. How does weight differ from mass?
5.What are elements?
6. Name the 4 elements that make up most of a living thing.
7. What is used to represent an element?
8. Name the smallest part of an element.
9. What are the 2 main regions of an atom?
10. Where is the nucleus of an atom found and what does it contain?
11. What is the charge on a proton? A neutron?
12. All atoms of the same element have the ________ number of protons.
13. The number of protons in an atom determines the _________________________.
14. What is the charge on an electron?
15. If you know the number of protons in an atom, how can you determine the number of electrons?
16. Do all the atoms of the same element have the same number of neutrons?
17. Atoms of the same element with different numbers of neutrons are called ___________.
18. The mass of an atom is centered in the ___________.
19. The number of protons PLUS neutrons in an atom determines its ___________________.
20. What atomic particle has a negative charge?
21. Why isn’t the mass of an electron used to determine the atomic mass of an element?
22. Where are electrons found?
23. Which electrons in an atom have the most energy?
24. How many energy levels are there & name them?
25. How many electrons will each energy level hold?
26. Elements are arranged on a __________________ by their atomic _____________.
27. What are Periods & what do they tell you about elements?
28.What are Families & what do they tell you about elements?
29. Two or more elements combined together make a _____________.
30. Chemical ______________ represent compounds.
31. _____________ are the smallest part of a compound.
32. ______________ in chemical formulas tell the number of atoms of each element.
33. _____________ in a formula tell the number of molecules.
34. Compounds have ____________ properties than its elements.
35. The outermost _______________ in elements determine if they will combine.
36. Elements with ___________ outer energy levels are stable & won’t react.
37. Why do elements tend to react with other elements?
38. Chemical _____________ represent chemical reactions.
39. ______________ appear on the right of the equation, while ___________ are on the left.
40. Chemical bonds store ____________.
41. How do covalent bonds form?
42. How do ions form?
43. Positive ions _________ electrons, while negative ions ___________ electrons.
44. Define energy.
43. Give some examples of different forms of energy.
44. Atoms are in constant ________________, which determines the atom’s ____________.
45. List the 3 main states of matter.
46. _______________ energy must be added or removed to change the state of matter.
47. In reactions the amount of product must ____________ the amount of reactants.
48. Most of an organism’s energy comes from _____________ in foods.
49. _______________ reactions release energy.
50. Give an example of an exergonic reaction in cells.
51. _________________ reactions store energy.
52. Give an example of an endergonic reaction in cells.
53. Most reactions in cells are _________________.
54. _________________ energy is the energy needed to start a reaction.
55. Catalysts _____________ the amount of activation energy needed.
56. Catalysts in organisms are called ______________ and are usually ______________.
57. Enzymes act on ____________ which join at the _______________ site.
58. Can enzymes be reused?
59. Redox reactions stands for _________________________ reactions.
60. The oxidized substance _____________ electrons & becomes _____________ charged.
61. The reduced substance _____________ electrons & becomes _____________ charged.
62. Define solution.
63. The _____________ is dissolved in the ____________ in a solution.
64. _______________ is the universal solvent.
65. A solution becomes _____________ when no more solute will dissolve.
66. ______________ solutions have water as the solvent & are important to ______________.
67. What is the dissociation of water?
68. Write the equation for this.
69. OH is the ______________ ion, while H+ is the ______________ ion.
70. Acids produce _____________ ions, while bases have an excess of ________ ions.
71. The _______ scale measures the concentration of H+ ions and goes from ____________.
72. From 0 to 7 are ___________, a pH of 7 is __________, & above 7 to 14 are _________.
73. _____________ are used in the body to control pH and keep it near a pH of ______ or neutral.