|
Photosynthesis |
Section 6-1 Capturing Light Energy
1. All organisms require ___________________ to carry out their life functions.
2. ___________________ is the ultimate energy for all life on earth.
3. During photosynthesis, the energy from the sun is stored within _____________________
compounds, mainly the sugar _______________________.
4. What organisms can carry on photosynthesis?
5. Name several autotrophic organisms.
6. What is a biochemical pathway and give an example?
7. What gas is used by autotrophs & what gas is produced?
8. What organisms release stored energy from organic compounds through cellular respiration?
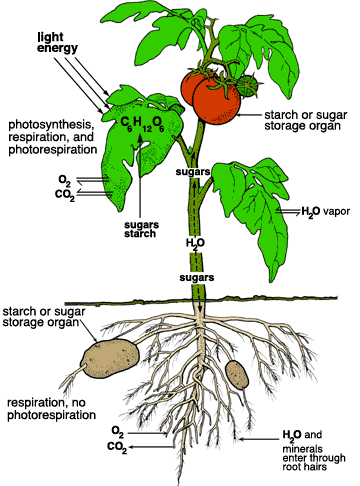
9. Draw the diagram showing energy storage & transfer between autotrophs & heterotrophs. (Figure 6.1)
10. What are the light reactions of plants and in what organelle do they occur?
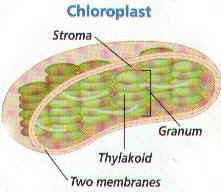
11. Draw & label the parts of a chloroplast. Tell the function of each labeled part.
12. Flattened sacs in chloroplasts are known as ____________________ and are
_______________________ to each other.
13. Thylakoid sacs in chloroplasts are called _____________________________.
14. What gel-like solution surrounds the thylakoids inside the chloroplast?
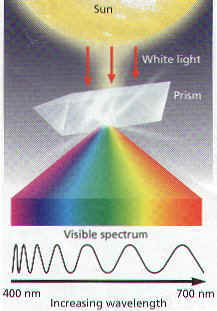
15. What is the visible spectrum?
16. Name the 7 colors that make up the visible spectrum.
17. What 3 things can happen to light that strikes an object?
18. What are pigments & what is their function in plants?
19. Is red light reflected or absorbed by an object if the object appears red to your eyes?
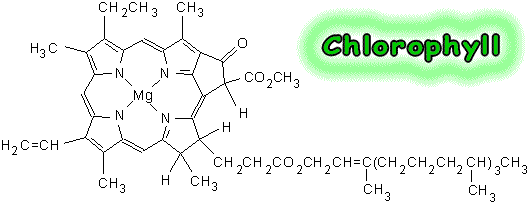
20. Name the most important chloroplast pigment & tell the 2 most important types of this pigment.
21. Only ________________________ is directly in capturing light energy.
22. Chlorophyll b is an example of an ______________________ pigment in plants.
23.Name another accessory pigment & tell what colors it includes. When could you see these colors?
24. Chlorophyll is most abundant in the _____________________ of a plant, while accessory
pigments appear more in the _________________________ and fruits.
25. The _________________________ and ________________________ pigments are grouped
into clusters in the thylakoid membrane.
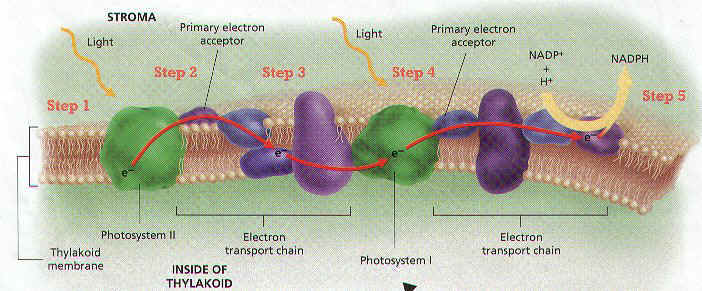
26. What is a photosystem?
27. Name the 2 types of photosystems.
28. The light reactions start when __________________ pigments absorb ______________.
29. Absorbed light is passed to a pair of ________________________ pigment molecules in
photosystem ________.
30. When light energy is absorbed by chlorophyll a molecules, what happens to its electrons?
31. Once these electrons become “excited”, they have enough energy to do what?
32. What are the chemicals called that pick up these freed electrons & where are they located?
33. These electrons lose _________________ as they are passed through a series of molecules
called the ______________________________________ chain.
34. Photosystem I chlorophyll molecules also absorb ________________, and its electrons
eventually combine with ______________________ to form NADPH.
35. What would happen if the electrons lost from photosystem II weren’t replaced?
36. ________________________ provides the replacement electrons for photosystem II when
water is __________________________.
37. Write the equation for the splitting of a water molecule.
38. What important gas is released when water is split?
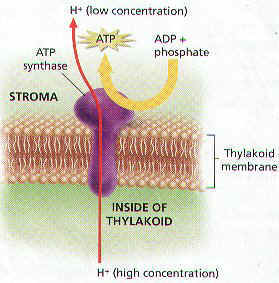
39. ______________ or energy for a cell is synthesized during the light reactions in a process
called ________________________________.
Section 6-2 Calvin Cycle
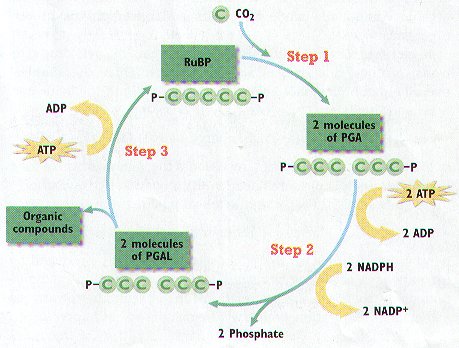
40. The _________________ cycle is the second set of photosynthetic reactions that uses energy
stored in ________________ and _____________________ to make __________________
compounds.
41. Carbon atoms from ______________ are “fixed” into organic compounds in the Calvin
cycle in a process called carbon _________________________.
42. In what part of the chloroplast does the Calvin cycle occur?
43. Carbon dioxide combines with _______________ to make two molecules of
_____________________________.
44. PGA is converted into ________________, ADP, _________________, and
phosphate.
45. Carbohydrates made from PGAL in the Calvin cycle include the monosaccharides
______________________ and ______________________, the disaccharide
_______________________, and polysaccharides such as _____________________,
________________________, and _______________________.
46. Write the balanced equation for photosynthesis. (See bottom of page 118.)
47. Plants that fix carbon through the Calvin cycle are called what type of plants?
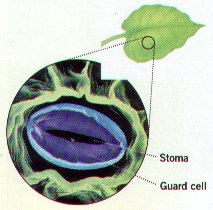
48. What are stomata & where are they located?
49. When would plant cells need to close or partially close their stomata?
50. Name 2 alternate carbon-fixing pathways used by plants in hot climates.
51. Plants that close their stomata during the hottest part of the day thus fixing carbon into four
carbon compounds are called ______________________. Name three.
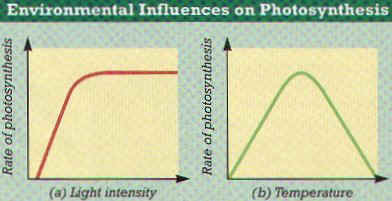
52. CAM plants open stomata at ______________ and close during the _________________.
53. Name 3 environmental factors that affect the rate of photosynthesis.