| Seven Life Processes | |
| Movement Reproduction Sensitivity Nutrition Excretion Respiration Growth |

MRS NERG |
| What one MAIN characteristic do ALL organisms have in common? |
|
| They’re all made of cells! | |
| Model of a Nucleotide |
Introduction
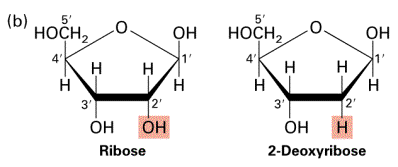
Nucleotides consist of three parts — a pentose sugar, a nitrogen-containing base, and a phosphate group. A pentose sugar is a five-sided sugar. There are 2 kinds of pentose sugars — deoxyribose and ribose. Deoxyribose has a hydrogen atom attached to its #2 carbon atom (designated 2′), and ribose has a hydroxyl group atom there. Deoxyribose-containing nucleotides are the monomers of DNA, while Ribose-containing nucleotides are the monomers of RNA.
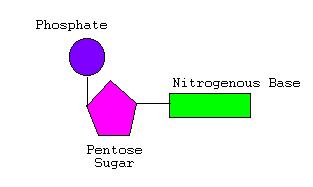
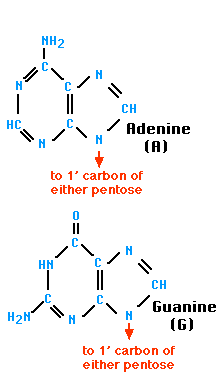
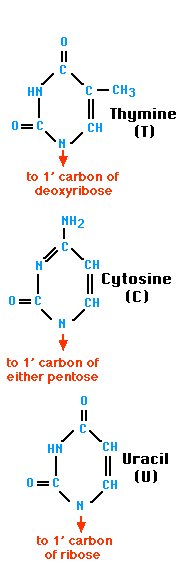
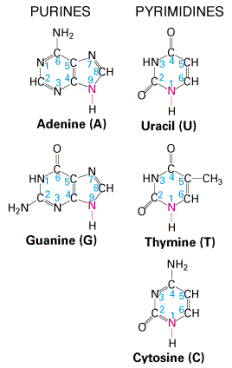
A nitrogen-containing ring structure is called a base. The base is attached to the 1′ carbon atom of the pentose. In DNA, four different bases are found — two purines, called adenine (A) and guanine (G) and two pyrimidines, called thymine (T) and cytosine (C). RNA contains The same purines, adenine (A) and guanine (G). RNA also uses the pyrimidine cytosine (C), but instead of thymine, it uses the pyrimidine uracil (U).
| The Purines | The Pyrimidines |
 |
 |
The combination of a base and a pentose is called a nucleoside. A phosphate group is attached to the 5′ carbon of the pentose sugar.
Objective
Students will construct a 3-dimensional model of a single nucleotide, the monomer of which nucleic acids are composed.
Materials
Various materials may be used for the atoms that make up a nucleotide such as styrofoam balls, plastic coke bottle caps, beads, etc. Bonds between atoms may be made from toothpicks, plastic stirring sticks, popsicle sticks, etc. Single & double bonds must be represented by the correct number of “sticks”. The atoms and bonds may NOT be made of any food item. Your model should be glued together to make the model rigid for hanging. Attach string and a label with the nucleotide’s name to your model. Models must be sturdy, light weight, and small enough to hang from the ceiling.
Color Code for atoms:
| CARBON – | BLACK |
| HYDROGEN – | YELLOW |
| OXYGEN – | RED |
| NITROGEN – | BLUE |
Structural Formulas of Nucleotides:
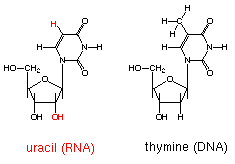
| Uracil Nucleotide (Ribose ) & Thymine Nucleotide (Deoxyribose)
|
 |
| Adenine Nucleotide (Deoxyribose) |
 |
| Cytosine Nucleotide (Deoxyribose) |
 |
| Guanine Nucleotide (Deoxyribose) |
|
|
Nucleic Acids and Protein Synthesis
All Materials © Cmassengale

Cell à Nucleus à Chromosomes à Genes à DNA
Proteins
DNA




 |
 |

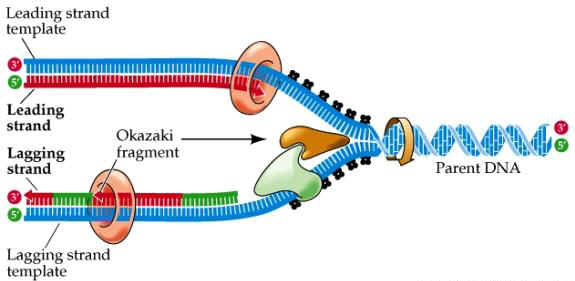
DNA Replication



OKAZAKI FRAGMENTS


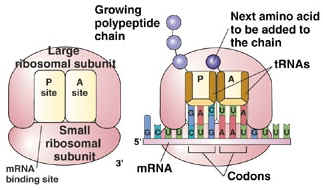
mRNA
tRNA

rRNA
Amino Acids
Genetic Code (RNA)
| Amino Acid | 3 Letter Abbreviation |
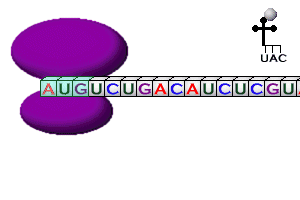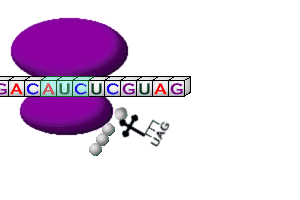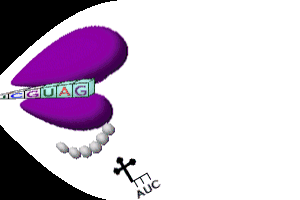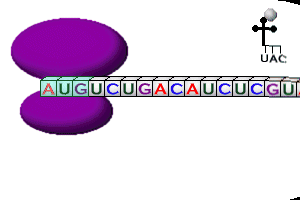
Codons |
| Alanine | Ala | GCA GCC GCG GCU |
| Arginine | Arg | AGA AGG CGA CGC CGG CGU |
| Aspartic Acid | Asp | GAC GAU |
| Asparagine | Asn | AAC AAU |
| Cysteine | Cys | UGC UGU |
| Glutamic Acid | Glu | GAA GAG |
| Glutamine | Gln | CAA CAG |
| Glycine | Gly | GGA GGC GGG GGU |
| Histidine | His | CAC CAU |
| Isoleucine | Ile | AUA AUC AUU |
| Leucine | Leu | UUA UUG CUA CUC CUG CUU |
| Lysine | Lys | AAA AAG |
| Methionine | Met | AUG |
| Phenylalanine | Phe | UUC UUU |
| Proline | Pro | CCA CCC CCG CCU |
| Serine | Ser | AGC AGU UCA UCC UCG UCU |
| Threonine | Thr | ACA ACC ACG ACU |
| Tryptophan | Trp | UGG |
| Tyrosine | Tyr | UAC UAU |
| Valine | Val | GUA GUC GUG GUU |
| Start | AUG | |
| Stop | UAA UAG UGA |
Practice Table:
| DNA Codon |
mRNA Codon |
tRNA Anticodon |
Amino Acid |
GCU |
|||
| TAC | |||
| AUU | |||
| UUU | |||
| TCA | |||
| UCU | |||
| CTT | |||
| ACU | |||
| ACU |
Protein Synthesis
Steps in Transcription
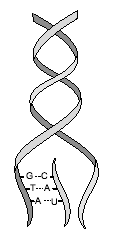
Steps in Translation

| Survival of the Fittest |  |
Introduction:
Within a population, organisms will vary. Charles Darwin stated that in the struggle for existence, those variant organisms that have favorable variations are “better adapted” to their environment and will survive and reproduce in greater numbers. Favorable variations may mean that they are faster, or stronger, or able to eat different types of food, or better camouflaged to avoid predators. In this lab you will simulate the effect of predation by a hawk on a large population of assorted mice. Your population of mice will consist of black, white, and speckled mice. You will represent the hawk.
Objectives:
–to simulate the effect of hawk predation on the appearance of mice
-to simulate the natural selection of traits
Materials:
large sheet of newspaper 4 hawks (students)
30 white mice (paper squares) 1 petri dish
30 speckled mice (paper squares)
30 black mice (paper squares)
Procedure:
mice |
mice |
||
| Hawk #1 |
|
||
| Hawk #2 |
|
||
| Hawk #3 |
|
||
| Hawk #4 |
|
||
| Total |
|
Conclusion:
Write a paragraph describing;
* the purpose of the lab
* what you thought the results would be
* what the results were (discussing numbers from data)
*how the mouse population and hawk population may change over time from natural selection
| Natural Selection Within a Species |  |
Introduction:
Natural selection is the evolutionary process by which the most adaptable individuals survive. An adaptation is an inherited variation that helps an organism to survive. When the organism survives, its chances of reproduction are increased as well as its ability to pass on its inherited traits. All members of a species are different from one another. In this activity, you will investigate two variations among peanut plants — length of shell and number of seeds per shell. Most shells contain a certain number of seeds which is an adaptation to its survival.
Objective:
Students will investigate natural selection in peanuts.
Materials:
50 peanuts in shells, metric ruler, pencil, graph paper
Procedure:
Data:
Table 1
| Length of Shell (mm) |
Number of Shells |
| 5-10 | |
| 10-15 | |
| 15-20 | |
| 20-25 | |
| 25-30 | |
| 30-35 | |
| 35-40 | |
| 40-45 | |
| 45-50 | |
| 50-55 | |
| 55-60 | |
| 60-65 | |
| 65-70 | |
| 70-75 |
Table 2
| Number of Seeds Per Shell | Number of Shells |
| 1 | |
| 2 | |
| 3 |
Title: ________________________________________________________

Title: ________________________________________________________

Questions: