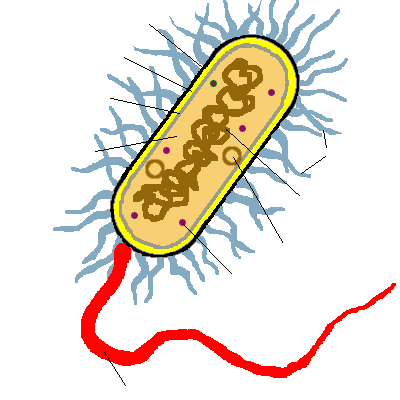
KINGDOMS ARCHAEBACTERIA & EUBACTERIA

All Materials © Cmassengale
Bacterial Evolution & Classification
- Most numerous organisms on earth
- Earliest life forms (fossils date 2.5 billion years old)
- Microscopic prokaryotes (no nucleus nor membrane-bound organelles)
- Contain ribosomes
- Infoldings of the cell membrane carry on photosynthesis & respiration
- Surrounded by protective cell wall containing peptidoglycan (protein-carbohydrate)
- Many are surrounded by a sticky, protective coating of sugars called the capsule or glycocalyx (can attach to other bacteria or host)
- Have only one circular chromosome
- Have small rings of DNA called plasmids
- May have short, hairlike projections called pili on cell wall to attach to host or another bacteria when transferring genetic material
- Most are unicellular
- Found in most habitats
- Most bacteria grow best at a pH of 6.5 to 7.0
- Main decomposers of dead organisms so recycle nutrients
- Some bacteria breakdown chemical & oil spills
- Some cause disease
- Move by flagella, gliding over slime they secrete ( e.g. Myxobacteria)
- Some can form protective endospores around the DNA when conditions become unfavorable; may stay inactive several years & then re-activate when conditions favorable
- Classified by their structure, motility (ability to move), molecular composition, & reaction to stains (Gram stain)
- Grouped into 2 kingdoms — Eubacteria (true bacteria) & Archaebacteria (ancient bacteria)
- Once grouped together in the kingdom Monera
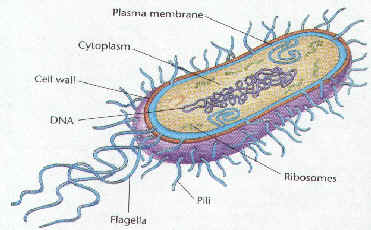
| STRUCTURE | FUNCTION |
| Cell Wall | protects the cell and gives shape |
| Outer Membrane | protects the cell against some antibiotics (only present in Gram negative cells) |
| Cell Membrane | regulates movement of materials into and out of the cell; contains enzymes important to cellular respiration |
| Cytoplasm | contains DNA, ribosomes, and organic compounds required to carry out life processes |
| Chromosome | carries genetic information inherited from past generations |
| Plasmid | contains some genes obtain through genetic recombination |
| Capsule, and slime layer | protects the cell and assist in attaching the cell to other surfaces |
| Endospore | protects the cell against harsh environmental conditions, such as heat or drought |
| Pilus (Pili) | assist the cell in attaching to other surfaces, which is important for genetic recombination |
| Flagellum | moves the cell |
Kingdom Archaebacteria
- Found in harsh environments (undersea volcanic vents, acidic hot springs, salty water)
- Cell walls without peptidoglycan
- Subdivided into 3 groups based on their habitat — methanogens, thermoacidophiles, & extreme halophiles

Methanogens
- Live in anaerobic environments (no oxygen)
- Obtain energy by changing H2 and CO2 gas into methane gas
- Found in swamps, marshes, sewage treatment plants, digestive tracts of animals
- Break down cellulose for herbivores (cows)
- Produce marsh gas or intestinal gas (methane)
Extreme Halophiles
- Live in very salty water
- Found in the Dead Sea, Great Salt Lake, etc.
- Use salt to help generate ATP (energy)
Thermoacidophiles (Thermophiles)
- Live in extremely hot (1100C) and acidic (pH 2) water
- Found in hot springs in Yellowstone National Park, in volcanic vents on land, & in cracks on the ocean floor that leak scalding acidic water
Kingdom Eubacteria (true bacteria)
- Most bacteria in this kingdom
- Come in 3 basic shapes — cocci (spheres), bacilli (rod shaped), spirilla (corkscrew shape)

- Bacteria can occur in pairs ( diplo– bacilli or cocci)
- Bacteria occurring in chains are called strepto- bacilli or cocci
- Bacteria in grapelike clusters are called staphylococci
- Most are heterotrophic (can’t make their own food)
- Can be aerobic (require oxygen) or anaerobic (don’t need oxygen)
- Subdivided into 4 phyla — Cyanobacteria (blue-green bacteria), Spirochetes, Gram-positive, & Proteobacteria
- Can be identified by Gram staining (gram positive or gram negative)
Gram Staining
- Developed in 1884 by Danish microbiologist, Hans Gram
- Bacteria are stained purple with Crystal Violet & iodine; rinsed with alcohol to decolorize; then restained with Safranin (red dye)
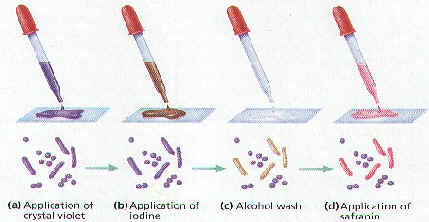
- Bacterial cell walls either stain purple or reddish-pink
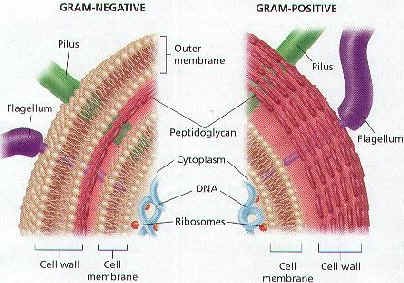
Gram-positive bacteria (Gram +)
- Thick layer of peptidoglycan (protein-sugar) complex in cell walls & single layer of lipids
- Stain purple

- Lactobacilli are used to make yogurt, buttermilk ….
- Actinomycetes make antibiotics like tetracycline & streptomycin
- Disease-causing gram + bacteria produce poisons called toxins
- Clostridium causes tetanus or lockjaw
- Streptococcus cause infections such as “strep” throat
- Staphylococci cause “staph” infections

- Also cause toxic shock, bacterial pneumonia, botulism (food poisoning), & scarlet fever
- Can be treated with penicillin (antibiotics) & sulfa drugs
Gram-negative bacteria (Gram -)
- Cell walls have a thin layer of peptidoglycan & an extra layer of lipids on the outside
- Stain pink or reddish
- Lipid layer prevents the purple stain & antibiotics from entering (antibiotic resistant)
- Some are photosynthetic but make sulfur, not oxygen
- Rhizobacteria grow in root nodules of legumes (soybeans, peanuts…) & fix nitrogen form the air for plants
- Rickettsiae are parasitic bacteria carried by ticks that cause Rocky Mountain spotted fever
- Spirochetes can cause syphilis & Lyme disease
Phylum Cyanobacteria
- Gram negative
- Carry on photosynthesis & make oxygen
- Called blue-green bacteria
- Contain pigments called phycocyanin (red & blue) & chlorophyll a (green)
- May be red, yellow, green, brown, black, or blue-green
- Some grow in chains (e.g. Oscillatoria) & have specialized cells called heterocysts that fix nitrogen

OSCILLATORIA
- First bacteria to re-enter devastated areas
- Anabaena that live on nitrates & phosphates in water can overpopulate & cause “population blooms” or eutrophication
- After eutrophication, the cyanobacteria die, decompose, & use up all the oxygen for fish
Phylum Spirochetes
- Gram positive
- Have flagella at each end so move in a corkscrew motion
- Some are aerobic (require oxygen); others are anaerobic
- May be free-living, parasitic, or live symbiotically with another organism
Phylum Gram Positive bacteria
- Most are Gram +, but some are Gram –
- Lactobacilli grow in milk & make lactic acid (forms yogurt, cottage cheese, buttermilk) & also found on teeth & cause tooth decay
- Actinomycetes grow in the soil & make antibiotics
- Gram + members are found in the oral & intestinal cavities & slow the growth of disease-causing bacteria
Phylum Proteobacteria
- Largest & most diverse bacterial group
- Subdivided into Enteric bacteria, Chemoautotrophic bacteria, & Nitrogen-fixing bacteria
Enteric bacteria
- Gram negative heterotrophs
- Can live in aerobic & anaerobic environments
- Includes E. coli that lives in the intestinal tract making vitamin K & helping break down food
- Salmonella causes food poisoning
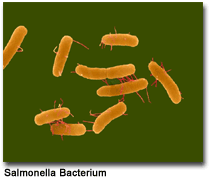
Chemoautotrophs
- Gram negative bacteria that obtain energy from minerals
- Iron-oxidizing bacteria found in freshwater ponds use iron salts for energy
Nitrogen-Fixing bacteria
- Rhizobium are Gram negative & live in legume root nodules
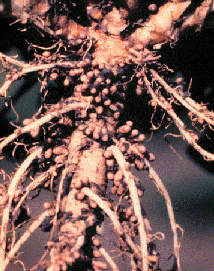
- 80% of atmosphere is N2, but plants can’t use nitrogen gas
- Nitrogen-fixing bacteria change N2 into usable ammonia (NH3)
- Important part of the Earth’s nitrogen cycle
Methods of Nutrition
- Saprobes feed on dead organic matter
- Parasites feed on a host cell
- Photoautotrophs use sunlight for energy, but get carbon from organic compounds (not CO2) to make their own food
- Chemoautotrophs obtain food by oxidizing inorganic substances like sulfur, instead of using sunlight
Methods of Respiration
- Obligate aerobic bacteria can’t live without oxygen; (tuberculosis bacteria)
- Obligate anaerobes die if oxygen is present; (tetanus bacteria that causes lockjaw)
- Facultative anaerobes do not need oxygen, but don’t die if oxygen is present; (E. coli)
- Anaerobes carry on fermentation, while aerobes carry on cellular respiration
Bacterial Reproduction & Genetic Recombination
- Most bacteria reproduce asexually by binary fission (chromosome replicates & then the cell divides)
- Bacteria replicate (double in number) every 20 minutes under ideal conditions
- Bacteria contain much less DNA than eukaryotes
- Bacterial plasmids are used in genetic engineering to carry new genes into other organisms
- Bacteria recombine genetic material in 3 ways — transformation, conjugation, & transduction
Conjugation
- Sexual reproductive method
- Two bacteria form a conjugation bridge or tube between them
- Pili hold the bacteria together
- DNA is transferred from one bacteria to the other
Transformation
- Bacteria pick up pieces of DNA from other dead bacterial cells
- New bacterium is genetically different from original
Transduction
- A bacteriophages (virus) carries a piece of DNA from one bacteria to another
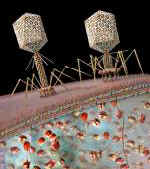
- Human insulin is produced in the lab by this method
Pathogenic bacteria
- Known as germs or pathogens
- Cause disease
- Can produce poisonous toxins
- Endotoxins are made of lipids & carbohydrates by Gram – bacteria & released after the bacteria die (cause high fever, circulatory vessel damage…)
- E. coli produce endotoxins
- Exotoxins are made of protein by Gram + bacteria
- Clostridium tetani produce exotoxins
- Antibiotics interfere with cellular functions (Penicillin interferes with synthesis of the cell wall; tetracycline interferes with protein synthesis)
- Some antibiotics are made by bacteria or fungi
- Broad-spectrum antibiotics affect a wide variety of organisms
- Bacteria can mutate and become antibiotic resistant (often results from overuse of antibiotics)