Chemistry
All Materials © Cmassengale
Composition of Matter
Ø Everything in the universe is made of matter
Ø Matter takes up space & has mass
Ø Mass is a measure of the amount of matter in the substance
Ø Mass & weight are NOT the same
Ø Weight is a measure of the pull of gravity on an object
Question: Is the mass of an object the same on the moon as it is on the Earth? Is its weight the same? (Hint: Gravitational pull on the moon is 1/6 of that on the Earth.)
Ø Matter exists in 4 states – solid, liquid, gas, & plasma

Ø Solids have both a definite volume & definite shape (rock)
Ø Liquids have a definite volume but no definite shape; they can be poured (water)
Ø Gases do not have a definite volume or definite shape, but they take the volume & shape of their container
Ø Plasmas have no definite volume, no definite shape, and only exist at extremely high temperatures such as the sun
Ø Chemical Changes in matter are essential to all life processes
Ø Biologists study chemistry because all living things are made of the same kinds of matter that make up nonliving things
Elements
Ø Elements are pure substances which cannot be chemically broken down into simpler kinds of matter
Ø More than 100 elements have been identified, but only about 30 are important in living things
Ø All of the Elements are arranged on a chart known as the Periodic Table
Ø Periodic charts tell the atomic number, atomic mass, & chemical symbol for every element
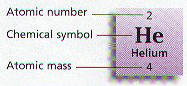
Ø Four elements, Carbon – C, Hydrogen – H, Oxygen – O, and Nitrogen – N make up almost 90% of the mass of living things
Ø Every element has a different chemical symbol composed of one to two letters
Ø Chemical symbols usually come from the first letter or letters of an element like C for Carbon and Cl for Chlorine
Ø Some chemical symbols come form their Latin or Greek name such as Na for Sodium (natrium) or K for Potassium (Kalium)
Ø Elements in the same horizontal period on the periodic table have the same number of energy levels (e.g. H & He in period 1 have only a K energy level)
![[Periodic Table]](https://biologyjunction.com/images/periodic.jpg)
All Period 2 elements have 2 energy levels
(K & L)
Ø Elements in the same vertical Family on the periodic table have the same number of electrons in their outermost energy level & react similar (e.g. Family IV, the Carbon family all have 4 electrons in their outermost energy level)
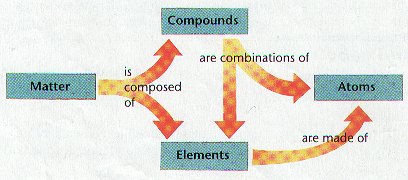
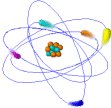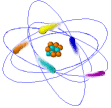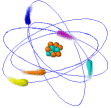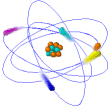
Atoms
Ø Atoms are the simplest part of an element that keeps all of the element’s properties
Ø Atoms are too small to be seen so scientists have developed models that show their structure & properties
Ø Atoms consist of 3 kinds of subatomic particles – protons & neutrons in the center or nucleus, and electrons spinning in energy levels around the center
Ø The nucleus is the center of an atom where most of the mass is concentrated
Ø Protons are positively charged ( p+ ), have a mass of 1 amu (atomic mass unit) , are found in the nucleus, and determine the atomic number of the element
Example: Carbon has 6 protons so its atomic number is 6
Ø Neutrons are neutral or have no electrical charge (n), have a mass of 1 amu, are found in the nucleus, and when added to the number of protons, determine the atomic mass of the element
Example: Sodium has 11 protons and 12 neutrons so its atomic mass is 11+12=23 amu
Ø Electrons (e-) are negatively charged, high energy particles with little mass that spin around the nucleus in energy levels
Ø Seven energy levels (K, L, M, N, O, P, & Q) exist around the nucleus and each holds a certain number of electrons
Ø The K energy level is closest to the nucleus & only holds 2 electrons, while the L – Q energy levels can hold 8 electrons

Ø Electrons in outer energy level are traveling faster & contain more energy than electrons in inner levels

Ø The number of protons (positive charges) and electrons (negative charges in an atom are equal so the net electrical charge on a atom is zero making it electrically neutral
Ø Stable or non-reactive atoms have an outer energy level that is filled with electrons

Compounds
Ø Most elements do not exist by themselves; Most elements combine with other elements
Ø Compounds are made of atoms of two or more elements chemically combined
Ø Chemical Formulas represent a compound & show the kind & number of atoms of each element (e.g. H2O has 2 hydrogen & 1 oxygen)
Ø Compounds have different physical & chemical properties than the atoms that compose them (e.g. hydrogen & oxygen are gases but H2O is a liquid)
Ø The number & arrangement of electrons in an atom determines if it will combine to form compounds
Ø Chemical reactions occur whenever unstable atoms (outer energy level not filled) combine to form more stable compounds
Ø Chemical bonds form between atoms during chemical reactions
Types of Chemical Bonds
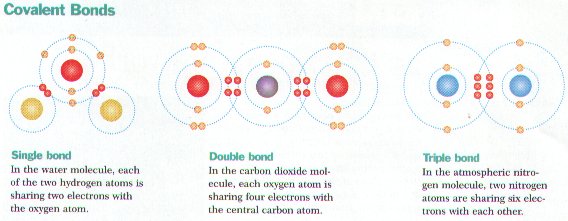
Ø Covalent bonds form between atoms whenever they share 1 or more pairs of electrons (e.g. H2O)
Ø Molecules form from covalent bonding & are the simplest part of a compound (e.g. NaCl, H2O, O2)
Ø Ionic bonding occurs between a positively & negatively charged atom or ion
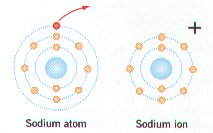
Ø Positively charged ions have more electrons (-) than protons (+); negatively charged ions have more protons than electrons
Ø Table salt (NaCl) forms when the 1 outer electron of Na is transferred to the outer energy level of chlorine that has 7 electrons (e-)
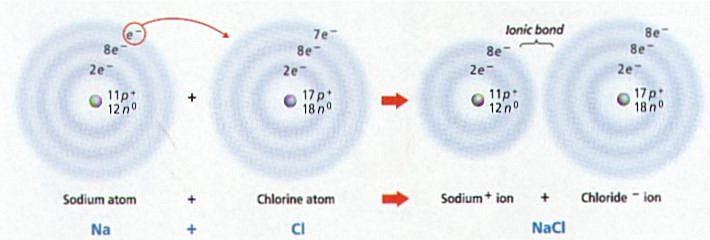
Ø Sodium (Na) with 1 less e- becomes positively charged, while Chlorine (Cl) with 1 more e- becomes negatively charged; the + and – charges attract & form the ionic bond holding NaCl together
Ø Other types of chemical bonding include hydrogen bonding
Energy
Ø Energy is the ability to do work
Ø Energy occurs in several forms & may be converted from one form to another
Ø Sunlight is the ultimate energy for all life on earth
Ø Forms of energy include chemical, electrical, mechanical, thermal, light, & sound
Ø Free energy is the energy available for work (e.g. cells have energy to carry out cell processes)
Ø Cells convert the chemical energy stored in food into other types of energy such as thermal & mechanical
Ø Energy is used to change matter form one state into another (e.g. liquid into a gas)
Chemical Reactions
Ø Living things undergo thousands of chemical reactions
Ø Chemical equations represent chemical reactions
Ø CO2 + H20—–goes to—–H2CO3 (carbonic acid) is a sample Chemical Reaction in living things
Ø Reactants are on the left side of the equation, while products are on the right side

Ø Activation energy is required to start many reactions

Ø Chemical bonds are broken, atoms rearranged, and new bonds form in chemical reaction
Ø Plants use sunlight to produce sugars such as C6H12O6 glucose; the chemical energy from the sun is stored in the chemical bonds of glucose
Ø Organisms eat plants, break down the sugars, and release energy along with CO2 & H2O
Ø Exergonic reactions involve a net release of energy; while endergonic reactions involve a net absorption of energy
Ø Energy must be added to the reactants for most chemical reactions to occur; called activation energy
Ø Enzymes are chemical substances in living things that act as catalysts & reduce the amount of activation energy needed
Ø Organisms contain thousands of different enzymes
Ø Most enzymes end with –ase (e.g. lipase is the enzyme that acts on lipids)

Reduction-Oxidation (Redox) reactions
Ø Reactions in which e- are transferred between atoms is a redox or reduction-oxidation reaction (e.g. formation of table salt NaCl)
Ø In oxidation reactions, a reactant loses 1 or more e- & becomes positively (+) charged (e.g. Sodium atom becomes a Na+ ion)
Ø In a reduction reaction, a reactant gains 1 or more e- and becomes negatively (-) charged (e.g. Chlorine atom becomes a Cl- ion)
Ø REDOX reactions always occur together; the electron(s) from the oxidation reaction are then accepted by another substance in the reduction reaction
Solutions
Ø A large percentage of the mass of organisms is water & many of the chemical reactions of life occur in water
Ø A solution is a uniform mixture of one substance in anther
Ø Solutions may be mixtures of solids, liquids, or gases
Ø The solute is the substance uniformly dissolved in the solution & may be ions, molecules, or atoms
Ø The solvent is the substance in which the solute is dissolved
Ø Water is known as the universal solvent
Ø Dissolving one substance in another does not alter their chemical properties
Ø The concentration of a solution is a measure of the amount of solute dissolved in a given volume of solvent
Ø Increasing the amount of solute increases the solution’s concentration
Ø Aqueous solutions are solutions in which water is the solvent; these are important in living things (e.g. blood, cytoplasm of cell…)
Acids and Bases
Ø The degree of acidity or alkalinity (basic) is important in organisms
Ø The force of attraction between molecules is so strong that the oxygen atom of one molecule can actually remove the hydrogen from other water molecules; called Dissociation
Ø H20—–GOES TO—– H+ + OH-
Ø OH- called hydroxide ion; H+ called hydrogen ion
Ø Free H+ ion can react with another water molecule to form H3O+ (hydronium ion)
Ø Acidity or alkalinity is a measure of the relative amount of H+ and OH- ions dissolved in a solution
Ø Neutral solutions have an equal number of H+ and OH- ions
Ø Acids have more H3O+ ions than OH- ions; taste sour; and can be corrosive
Ø Bases contain more OH- ions than H3O+ ions; taste bitter; & feel slippery
Examples of Common Acids
|
Examples of Common Bases
|
PH Scale
Ø Compares the relative concentration of H3O+ ions and OH- ions
Ø Scale ranges from 0 to 14; 0-3 is very acidic; 7 is neutral; 11-14 is very basic or alkaline

Ø Litmus paper, phenolphthalein, pH paper, & other indicators that change color can be used to measure pH

Buffers
Ø Control of pH is important to organisms
Ø Enzymes function only within a narrow pH range; usually neutral
Ø Buffers neutral acids or bases in organisms to help control pH
| Chemistry Study Guide | Chemistry On-line |
| BACK |