|
|
| Vertebrate Worksheet |  |
1. In what phylum & kingdom are the vertebrates found?
2. List the classes of vertebrates.
3. Discuss the characteristics of chordates & vertebrates.
4. What were the 1st vertebrates & describe them?
5. Sketch a lamprey & describe the characteristics of this fish. Where are they found?
6. Describe a hagfish.
7. In what group are lampreys & hagfish found & why?
8. Do agnathans have paired fins?
9. What were the 1st jawed fish & describe them.
10. What are the 2 classes of jawed fish?
11. What is in the class Chondrichthyes & what traits do they have in common.
12. Sketch & describe sharks.
13. Sketch a ray or skate & describe them.
14. Name the class for bony fish.
15. Name the 2 groups of bony fish.
16. Give several examples of ray-finned fish & describe them.
17. Name 2 lobe-finned fish & describe both of them.
18. What was the 1st group of vertebrates to move onto land? What is in this group?
19. Describe characteristics of amphibians.
20. Amphibians are ectotherms. What does this mean?
21. How are amphibians still linked to water?
22. What is in the class Reptilia?
23. Reptiles do not need water for reproduction. Explain why this is true.
24. Describe the amniote egg of reptiles. Include a labeled sketch of the egg.
25. What reptile group is thought to be the ancestors of mammals?
26. What were pterosaurs?
27. What 3 groups of retiles are still alive today?
28. Describe characteristics of the reptiles.
29. How can snakes swallow such large prey?
30. What is the purpose of the Jacobson’s organ in snakes?
31. What takes the place of teeth in turtles?
32. Describe crocodiles & alligators & tell some of their habits.
33. What class contains birds?
34. From what did birds probably evolve?
35. What are the distinguishing features of birds?
36. Sketch & label the parts of a feature.
37. Birds are endotherms. What does this mean?
38. Name some flightless birds.
39. Name some swimming birds.
40. What are the 3 main characteristics of all mammals?
41. What in female mammals produces milk?
42. What is mammalian hair made of & give its 4 functions.
43. What bones are modified in mammals to help them hear sounds?
44. Name a flying mammal.
45. Give examples of mammals that are herbivores.
46. Give examples of mammals that are carnivores.
47. What mammal is a thinker & toolmaker?
48.Name 7 adaptations of mammals.
49.Give examples of monotremes & tell their characteristics. Tell where they are found.
50. Give examples of marsupials & tell their characteristics. Tell where most of them are found.
51. Most mammals are placentals. What does this mean?
52. What is gestation? Do all mammals have the same gestation period?
53. What is the purpose of the chorion?
54.Name the 12 orders of placental mammals & give an example of an animal in each order.
Virus Models
Use viral pictures from your textbook or from a website to construct a 3-dimensional model of a virus. Your model must meet the following criteria:
The following rules for constructing you model must also be followed:
Types of viruses that make good models:
Models will be graded based on the following:
| Viruses All Materials © Cmassengale |
 |
Discovery of Viruses
 |
 |
| Wendell Stanley | Tobacco Leaf with Virus |
Viral Characteristics
 |
 |

Ebola Virus
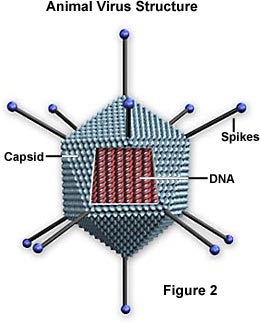
Viral Structure
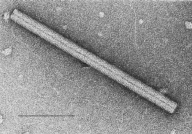
Bacteriophages or T-Phages
Retroviruses

HIV Virus
Viroids

Viroid Attack on Potatoes
Prions

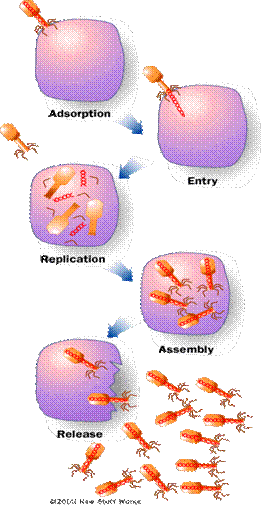
Lytic Cycle
Phases of the Lytic Cycle of a Virulent Virus:
lysis
|
Source: http://science.howstuffworks.com/virus-human.htm
Lysogenic Cycle
The Lysogenic Cycle of a Temperate Virus:
|

Source: http://science.howstuffworks.com/virus-human.htm
Viral Control
| Back |
Viruses, Viroids, & Prions
ppt Questions
Living or Nonliving?
1. Why might some people consider viruses alive?
2. Can viruses be “killed”?
3. Can a virus maintain homeostasis like cells?
4. Are viruses cellular or noncellular?
5. Define virus.
6. Name the 2 main things that make up a virus.
Viral History
7. Who coined the term virus and what does it mean?
8. Explain how viruses were first discovered.
9. What discovery did Wendell Stanley make about viruses? What type of virus was he studying?
10. __________ is an example of a viral disease.
11. Who found the vaccine against this viral disease? What milder virus was used to make the vaccine?
12. What is meant by a virulent virus?
13. Smallpox has been ________________ in the world today. What does this mean?
14. How does the size of a virus compare with that of a cell?
15. What metric units are used to measure the size of viruses?
16. What technology had to be developed before viruses can be seen?
17. Give the size of these viruses.
a. bacteriophage
b. polio virus
c. adenovirus
d. Ebola virus
Viral Structure
18. Are viruses made of cells? Are they living?
19. What covers the outside of a virus and what is it called?
20. What is in the core of a virus?
21. When & how can a virus reproduce?
22. What protective covering is around the capsid of some viruses?
23. What is the purpose of spikes and do all viruses have them?
24. Viruses only attack ___________ host cells.
25. Sketch and label the parts of a virus.
26. Describe the capsid of viruses.
27. What are capsomeres?
28. Are all viruses the same shape?
29. Outside of a host cell, viruses are ______________.
30. Do viruses have ribosomes like cells?
31. Do viruses have enzymes like cells?
32. Viruses use the _________ __________ and ____________ of its host cell to be able to ________________.
33. Does the HIV virus have spikes for attachment?
34. Besides smallpox and AIDS, name 6 other viral diseases.
35. Name a type of cancer thought to be caused by viruses.
36. What is the shape of each of these viruses:
a. ebola?
b. influenza?
37. Label the parts of these viruses.
 |
 |
 |
Taxonomy of Viruses
38. Family names for viruses end in what suffix?
39. Genus names for viruses end in ___________.
40.What is meant by a viral species?
41. ____________ names are used for different species of viruses.
42. How is the subspecies for a virus designated?
43. Give the family, genus, and species for the HIV and Herpes viruses.
44. What virus causes blisters that may appear around the mouth?
45. What virus is responsible for the common cold?
46. What virus causes warts?
47. Name 4 things used to identify viruses.
a.
b.
c.
d.
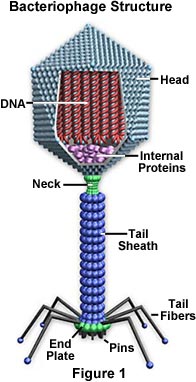
Bacteriophages
48. What is a bacteriophage?
49. Give 3 characteristics of T-phages.
a.
b.
c.
50. What are the most commonly studied T-phages?
51. T-phages often attack what bacterial cell? Where is the host cell found?
52. How does a bacteriophage attach to its host cell?
53. What is the only part of the virus that actually enters the host cell?
54. What is the shape of the bacterial host cell that bacteriophages attack?
55. How many sides does the head or capsid of a bacteriophage have?
56. T-bacteriophages have what nucleic acid at their core?
57. What are the head and tail fibers made of?
Retroviruses
58. What nucleic acid do retroviruses contain?
59. What enzyme do retroviruses contain that let them make DNA from RNA?
60. What 2 things do retroviruses inject into their host cells when they attack them?
61. Give an example of a retrovirus.
Viroids and Prions
62. What is a viroid?
64. What is the host for viroids?
65. Viroids are responsible for causing what major problem in Europe?
66. Viroids resemble ____________ pieces of DNA that are cut out because they do not code for any proteins.
67. Prions are infectious pieces of _____________.
68. Describe how prions occur.
69. Do prions have a nucleic acid core?
70. What protein is responsible for most mammalian prion diseases?
71. What happens when prions get into the brain?
72. What does BSE stand for and what is the common name for this prion disease?
73. Explain the prion disease caused kuru.
Viral Replication
74. Viruses are host specific. What does this mean?
75. Do the viruses that attack most animals also attack most humans?
76. What is the envelope of viruses often made from? What is the function of an envelope?
77. List the 5 steps of the Lytic cycle in viruses.
(1)
(2)
(3)
(4)
(5)
78. What structures help a virus attach to a host cell?
79. What does the virus inject into its host?
80. What viral parts are made inside a cell? where do the raw materials come from?
81. What is meant by cell lyses?
82. Place these steps of the lytic cycle in order: maturation, penetration, release, biosynthesis, and attachment.
83. Label the stages of the lytic cycle. Also label the capsid, host cell, and DNA.
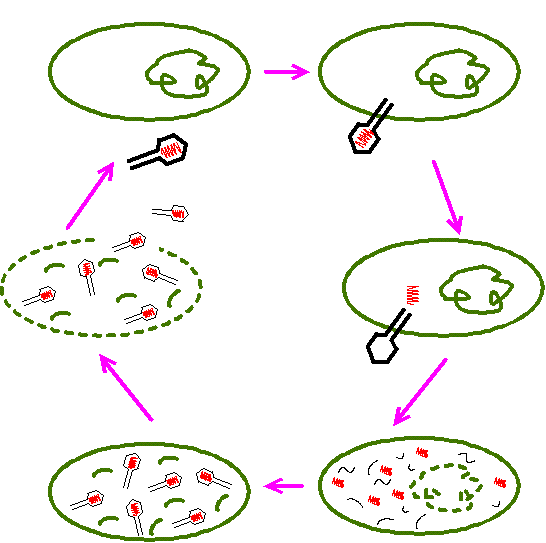
84. What are latent viruses?
85. How long can a latent virus remain inactive?
86. what activates latent viruses?
87. Give 2 examples of latent viruses.
88. During the lysogenic cycle of a virus, what happens to the viral nucleic acid after it is injected into the host cell?
89. What is a prophage?
90. What causes the phage or viral DNA to start replicating?
91. Does the prophage start replicating right after it is injected into the host cell? Explain.
92. Viral DNA along with the host cell DNA is replicated during each ___________ ___________.
93. After a long period of time, __________ cells form that contain ________________.
94. Once a prophage cell is activated, what happens?
95. Once a prophage is active, the host cell is ______________ making the virus deadly or _______________.
96. Label the viral DNA, host DNA, prophage, bacteriophage, host cell, lytic cycle, and lysogenic cycle.
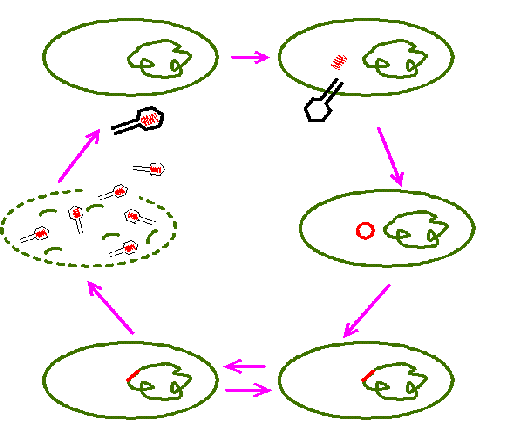
97. Give an example of a virus that remains dormant in the nervous system tissues for many years.
98. This virus may reappear later in life as a disease called ___________. Describe the disease.
99. ___________ infections also remain dormant in the nervous system.
100. How long does a herpes infection last?
101. Genital herpes is called Herpes ______________, while cold sores or fever blisters are known as Herpes ______________.
102. Viruses make us sick because they ___________ healthy host cells.
Treatment for Viral Disease
103. What is an attenuated virus?
104. How are viruses attenuated?
105. How are some viral vaccines made?
106. How does an attenuated virus help protect us from disease?
107. What proteins do our cells make to help protect us from viruses?
108. What is AZT?
109. How do protease inhibitors work to stop viruses?
