| Protozoan Review |  |
1. Protists with animal-like characteristics are called ___________________________.
2. Protozoans are all ______________________ organisms.
3. One convent way to classify protozoans is based on the way they _______________.
4. _____________________ move by extending their ___________________. ______________________ propel themselves by ______________________. _____________________ move by hairlike ____________, and _________________________ do ___________ move by themselves at all.
5. Protozoans that move by extending lobes of cytoplasm are called _______________________________.
6. The lobes of cytoplasm that sarcodinians extend are called _____________________ which means “_____________ ________________”.
7. When conditions are unfavorable, many amoebas survive by becoming hard ______________. The ____________ can withstand drought, heat, or being eaten by other organisms.
8. Not all sarcodinians are soft “Naked”, some have hard shells or _______________ made of __________________________________ or _______________________. They are called ____________________________ and _______________________________.
9. Sarcodinians are protozoans that move by extending lobes of _________________________.
10. ________________________________ are protozoans that move by means of flagella.
11. Some zooflagellates are free-living ________________________ or ____________________ organisms; many can live inside other organisms in _____________________ relationship. Zooflagellates may have a ________________________ or _________________________________ relationships with other organisms.
12. Which zooflagellate parasite causes African Sleeping Sickness in humans? ____________________________. The disease is spread by the bite of the __________________ ___________________.
13. _________________________ are protozoans that have bodies covered with short hairlike projections called _________________. They beat like ___________ to propel these protists through the water.
14. The ____________________________ is a common freshwater ciliate.
15. Paramecium gathers food with its _____________. The ____________ sweeps food particles into the ______________ _________________, the Mouth _____________ opens into a ___________________ which pinches off around them to form a ____________________ ______________________. It ejects wastes through an opening called the ______________________ __________________________.
16. Water is constantly enters the Paramecium cell by _______________________. They would burst if they did not have a way to get rid of excess water. ______________________________ _____________________________ collect the excess water and pump it outside.
17. Like all ciliates, Paramecium has ______________ distinct kinds of nuclei, each with a different function. The ___________________________ controls ongoing functions of the cell and __________________________ reproduction. Ciliates reproduce _______________________ by cell division. The _____________________________ is involved in genetic exchange during ___________________________ reproduction.
18. When a Paramecium reproduces sexually, it exchanges genetic information by ________________________________________.
19. The protozoans that have NO structures for movement, and lives by being a parasite in animals are the ______________________________. They are ____________-__________________ ____________________ protozoans.
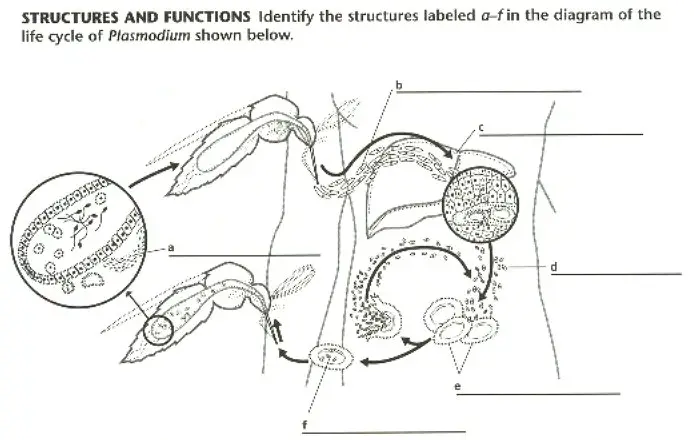
20. The protozoan that causes malaria is named ________________________ and is carried within _________________ ____________________.
21. Protozoans can grow and reproduce only in _____________ environments.
22. _____________________ is a collection of mostly microscopic organisms that float near the surface of the ocean and lakes.
23. Type of sarcodina that moves by pseudopodia is an _________________________.
24. The sporozoan that causes malaria is _______________________________.
25. The ciliophora that moves by cilia ________________________________.
26. Types of sarcodinians that are covered by a protective test are __________________________ and ______________________________.
27. The insect that transmits malaria to humans is the _____________________.
28. The sporozoan that is found in cats is ________________________ _____________________.
29. The zooflagellate that causes Chaga’s disease is ___________________________ _______________________.
30. The zooflagellate that is known to contaminate stream water in the U.S. is _______________________ __________________________.
31. ________________________ is a process of sexual reproduction in ciliates.
32. Protozoa are thought to have descended from ________________________ Eukaryotes.
33. Protozoan habitats are characterized by the presence of _______________________.
34. An adaptation to extreme environments is called ________________ _______________________________.
35. Sarcodines use their pseudopodia for ________________________________________, _____________________________________, and ________________________________.
36. Certain sarcodines affect Earth’s geology by having mineralized shells that form __________________________ _________________ after they die.
37. What do trypanosomiasis, Chaga’s disease, leishmaniasis, and giardiasis have in common? (Hint) They are all caused by __________________________________.
38. Pseudopodia are extensions of a sarcodine’s ____________________________.
39. In Paramecium, the macronucleus contains _______________________ _______________________ of _____________________.
40. What Two terms best describes members of the Kingdom Protista? (Hint) They are _________________________ – _______________ and ____________________________.
41. Most protists are made up of ______________________________ cell(s).
42. Most protists live in a ________________________________ environment.
43. Some protists undergo sexual reproduction only at times of environmental ________________________________.
44. Some protists have __________________________________ that contain light sensitive pigments.
45. Sleeping sickness is caused by a group of ____________________________ called trypanosomes.
46. _________________________________ has been used since the 1600s to relieve the symptoms of malaria.
47. Disease causing protists are transmitted mainly by insects and contaminated ______________________________.
DIRECTIONS: Answer the questions below as completely and as thoroughly as possible. Answer the question in essay form (not outline form), using complete sentences. You may use diagrams to supplement your answers, but a diagram alone without appropriate discussion is inadequate.
1. What kind of organisms are found in the kingdom Protista? What characteristics do they share?
2. Explain how parasitic zooflagellates infect their hosts. Give two examples.
3. Describe the life cycle of Plasmodium. What features typical of sporozoans does this life cycle exhibit.
4. Describe the four phyla of protozoa.
5. Would a motile or nonmotile protozoan be more likely to be free-living? Explain your answer.
6. Distinguish between the terms Protist and Protozoan.
7. What is conjugation? How is this process advantageous for ciliates, such as Paramecium?
8. Describe the process of ameboid movement and how it helps with the amoeba’s nutrition.
9. What are pseudopodia? What functions do they serve in sarcodines?
10. Describe Three means of locomotion among protozoa.
11. What is a cyst? Under what conditions might certain protozoa form cysts?
12. Explain how Conjugation in protozoa (Paramecium) differs from conjugation in bacteria?
13. Explain the role of protozoa in aquatic ecosystem food chains?
14. How have sarcodines built geological features of the environment?
15. What adaptive significance does the contractile vacuole have in fresh water sarcodine?
16. What Kinds of diseases can zooflagellates cause in humans?