The Floating Leaf Disk Assay for Investigating Photosynthesis
Brad Williamson
Introduction:
Trying to find a good, quantitative procedure that students can use for exploring photosynthesis is a challenge. The standard procedures such as counting oxygen bubbles generated by an elodea stem tend to not be “student” proof or reliable. This is a particular problem if your laboratory instruction emphasizes student-generated questions. Over the years, I’ve found that the floating leaf disk assay technique to be reliable and understandable to students. Once the students are familiar with the technique they can readily design experiments to answer their own questions about photosynthesis. I plan to add to this page as I have time to elaborate on the technique and provide suggestions for modifications.
Materials:
| 1. Sodium bicarbonate (Baking soda)
2. Liquid Soap 3. Plastic syringe (10 cc or larger)—remove any needle! 4. Leaf material 5. Hole punch 6. Plastic cups 7. Timer 8. Light source
|
Optional:
Buffer Solutions Colored Cellophane or filters Leaf material of different ages Variegated leaf material Clear Nail polish
|
Procedure:
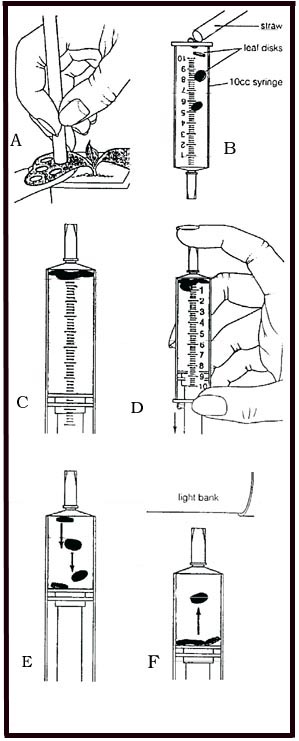
- Prepare 300 ml of bicarbonate solution for each trial.
- The bicarbonate serves as an alternate dissolved source of carbon dioxide for photosynthesis. Prepare a 0.2% solution. (This is not very much—it’s about 1/8 of a teaspoon of baking soda in 300 ml of water.) Too much bicarbonate will cause small bubbles (CO2)to form on the surface of the leaf which will make it difficult to sink the leaf disk.
- Add 1 drop of dilute liquid soap to this solution. The soap wets the hydrophobic surface of the leaf allowing the solution to be drawn into the leaf. It’s difficult to quantify this since liquid soaps vary in concentration. Avoid suds. If your solution generates suds then dilute it with more bicarbonate solution.
- Cut 10 or more uniform leaf disks for each trial

-
- Single hole punches work well for this but stout plastic straws will work as well
- Choice of the leaf material is perhaps the most critical aspect of this procedure. The leaf surface should be smooth and not too thick. Avoid plants with hairy leaves. Ivy, fresh spinach, Wisconsin Fast Plant cotyledons—all work well. Ivy seems to provide very consistent results. Any number of plants work. My classes have found that in the spring, Pokeweed may be the best choice.
- Avoid major veins.
- Infiltrate the leaf disks with sodium bicarbonate solution.
-
- Remove the piston or plunger and place the leaf disks into the syringe barrel. Replace the plunger being careful not to crush the leaf disks. Push on the plunger until only a small volume of air and leaf disk remain in the barrel (< 10%).
-
- Pull a small volume of sodium bicarbonate solution into the syringe. Tap the syringe to suspend the leaf disks in the solution.


-
- Holding a finger over the syringe-opening, draw back on the plunger to create a vacuum. Hold this vacuum for about 10 seconds. While holding the vacuum, swirl the leaf disks to suspend them in the solution. Let off the vacuum. The bicarbonate solution will infiltrate the air spaces in the leaf causing the disks to sink. You will probably have to repeat this procedure several times in order to get the disks to sink. You may have difficulty getting the disks to sink even after applying a vacuum three or four times. Generally, this is usually an indication that you need more soap in the bicarbonate solution. Some leaf surfaces are more water repellent than others are. Adding a bit more soap usually solves the problem.

- Pour the disks and solution into a clear plastic cup. Add bicarbonate solution to a depth of about 3 centimeters. Use the same depth for each trial. Shallower depths work just as well.

-
- This experimental setup includes a control. The leaf disks in the cup on the right were infiltrated with a water solution with a drop of soap—no bicarbonate.
- Place under the light source and start the timer. At the end of each minute, record the number of floating disks. Then swirl the disks to dislodge any that are stuck against the sides of the cups. Continue until all of the disks are floating.


-
- The control is on the left in each image. In the experimental treatment, on the right, leaf disks are rising and floating on the surface.
- Sample results:
| Time (minutes) | Disk Floating | |
| 1 | 0 | |
| 2 | 0 | |
| 3 | 0 | |
| 4 | 0 | |
| 5 | 0 | |
| 6 | 0 | |
| 7 | 1 | |
| 8 | 1 | |
| 9 | 1 | |
| 10 | 1 | |
| 11 | 4 | |
| 12 | 7 | |
| 13 | 8 | |
| 14 | 10 |
- The point at which 50% of the leaf disks are floating is the point of reference for this procedure. By interpolating from the graph, the 50% floating point is about 11.5 minutes. Using the 50% point provides a greater degree of reliability and repeatability for this procedure.


