| Extract DNA from Anything Living |  |
Introduction:
Since DNA is the blueprint for life, everything living contains DNA. DNA isolation is one of the most basic and essential techniques in the study of DNA. The extraction of DNA from cells and its purification are of primary importance to the field of biotechnology and forensics. Extraction and purification of DNA are the first steps in the analysis and manipulation of DNA that allow scientists to detect genetic disorders, produce DNA fingerprints of individuals, and even create genetically engineered organisms that can produce beneficial products such as insulin, antibiotics, and hormones.
DNA can be extracted from many types of cells. The first step is to lyse or break open the cell. This can be done by grinding a piece of tissue in a blender. After the cells have broken open, a salt solution such as NaCl and a detergent solution containing the compound SDS (sodiumdodecyl sulfate) is added. These solutions break down and emulsify the fat & proteins that make up a cell membrane. Finally, ethanol is added because DNA is soluble in water. The alcohol causes DNA to precipitate, or settle out of the solution, leaving behind all the cellular components that aren’t soluble in alcohol. The DNA can be spooled (wound) on a stirring rod and pulled from the solution at this point.
| Just follow these 3 easy steps:
Detergent, eNzymes (meat tenderizer), Alcohol
|
Objective:
To extract DNA from cells.
Materials:
Blender, split peas, salt, detergent, water, measuring cup and spoons, strainer, meat tenderizer, alcohol, test tube, glass stirring rod
Procedure:
- First, you need to find something that contains DNA such as split peas, fresh spinach, chicken liver, onion, or broccoli.

- Measure about 100 ml or 1/2 cup of split peas and place them in a blender.
- Add a large pinch of salt (less than 1 ml or about 1/8 teaspoon) to the blender.
- Add about twice as much cold water as the DNA source (about 200 ml or 1 cup) to the peas in the blender.
- Blend on high (lid on) for about 15 seconds.

- The blender separates the pea cells from each other, so you now have a really thin pea-cell soup.
And now, those 3 easy steps:
- Pour your thin pea-cell soup through a strainer into another container like a measuring cup or beaker.

- Estimate how much pea soup you have and add about 1/6 of that amount of liquid detergent (about 30ml or 2 tablespoons). Swirl to mix.

- Let the mixture sit for 5-10 minutes.
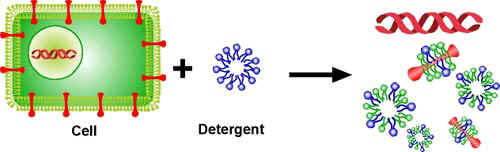
The detergent captures the proteins & lipids of the cell membrane.
- Pour the mixture into test tubes or other small glass containers, each about 1/3 full.
- Add a pinch of enzymes to each test tube and stir gently. Be careful! If you stir too hard, you’ll break up the DNA, making it harder to see. (Use meat tenderizer for enzymes. If you can’t find tenderizer, try using pineapple juice or contact lens cleaning solution.)

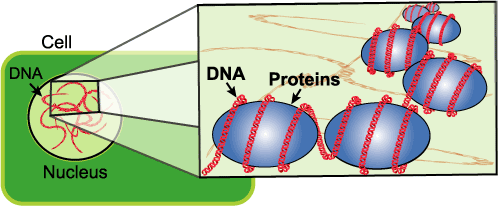
The DNA in the nucleus of the cell is molded, folded, and protected by proteins. The meat tenderizer cuts the proteins away from the DNA.
- Tilt your test tube and slowly pour rubbing alcohol (70-95% isopropyl or ethyl alcohol) into the tube down the side so that it forms a layer on top of the pea mixture. Pour until you have about the same amount of alcohol in the tube as pea mixture.

- Alcohol is less dense than water, so it floats on top forming two separate layers.
- All of the grease and the protein that we broke up in the first two steps move to the bottom, watery layer.
- DNA will rise into the alcohol layer from the pea layer. You can use a glass stirring rod or a wooden stick to draw the DNA into the alcohol.
- Slowly turning the stirring rod will spool (wrap) the DNA around the rod so it can be removed from the liquid.

Questions:
1. Does the DNA have any color?
2. Describe the appearance of the DNA.
3. Do only living things contain DNA? Explain.
Frequently Asked Questions: 1. I’m pretty sure I’m not seeing DNA. What did I do wrong?
First, check one more time for DNA. Look very closely at the alcohol layer for tiny bubbles. Often, clumps of DNA are loosely attached to the bubbles.
If you are sure you don’t see DNA, then the next step is to make sure that you started with enough DNA in the first place. Many food sources of DNA, such as grapes, also contain a lot of water. If the blended cell soup is too watery, there won’t be enough DNA to see. To fix this, go back to the first step and add less water. The cell soup should be opaque, meaning that you can’t see through it. Another possible reason for not seeing any DNA is not allowing enough time for each step to complete. Make sure to stir in the detergent for at least five minutes. If the cell and nuclear membranes are still intact, the DNA will be stuck in the bottom layer. Often, if you let the test tube of pea mixture and alcohol sit for 30-60 minutes, DNA will precipitate into the alcohol layer.
2. Why does the DNA clump together?
Single molecules of DNA are long and stringy. Each cell of your body contains six feet of DNA, but it’s only one-millionth of an inch wide. To fit all of this DNA into your cells, it needs to be packed efficiently. To solve this problem, DNA twists tightly and clumps together inside cells. Even when you extract DNA from cells, it still clumps together, though not as much as it would inside the cell.
Imagine this: the human body contains about 100 trillion cells, each of which contains six feet of DNA. If you do the math, you’ll find that our bodies contain more than a billion miles of DNA!
3. Can I use this DNA as a sample for gel electrophoresis?
Yes, but all you will see is a smear. The DNA you have extracted is genomic, meaning that you have the entire collection of DNA from each cell. Unless you cut the DNA with restriction enzymes, it is too long and stringy to move through the pores of the gel; instead, all you will end up seeing is a smear.
4. Isn’t the white, stringy stuff actually a mix of DNA and RNA?
That’s exactly right! The procedure for DNA extraction is really a procedure for nucleic acid extraction. However, much of the RNA is cut by ribonucleases (enzymes that cut RNA) that are released when the cells are broken open.