| EVOLUTION |  |
Section 14-1 Biogenesis
1. Define biogenesis.
2. What is spontaneous generation & give an example. Can spontaneous generation occur?
3. Describe Redi’s experiment and its results.
4. Describe Spallanzani’s experiment and its results.
5. Did Redi & Spallanzani’s experiments disprove spontaneous generation? Explain.
6. Describe Pasteur’s experiment and its results. Did it prove or disprove spontaneous generation?
Section 14-2 Earth’s History
7. What is the estimated age of the earth?
8. a. What is radioactive dating, and how is it used to tell the age of materials?
b. Use a science dictionary to look up and explain relative dating of fossils.
9. What is meant by half-life?
10. Name 3 radioactive isotopes and give their half-life.
11. a. What 2 scientists set up an experiment that simulated the atmosphere of early earth?
b. What molecules were made during this experiment?
Section 14-3 The First Life Forms
12. Why was there no oxygen in the early atmosphere?
13. Were the 1st cells probably aerobic or anaerobic? Why?
14. What gases did the early atmosphere contain primarily?
15. What bacteria live in harsh environments containing methane gas?
16. Oxygen didn’t become part of our atmosphere until what process started taking place?
Section 15-1 Fossil Record
17. What is a fossil?
18. In what type of rock are fossils usually found?
19. What is sedimentary rock?
20. From what part or parts of organisms do sedimentary fossils usually form?
21. How do sedimentary fossils form?
22. How do casts form?
23. What do you call imprint fossils?
24. Complete insect fossils can be found inside of a clear, golden material called ________________.
25. Who was one of the first scientists to study fossils?
26. In which rock layer or strata would the oldest fossils be found? The newest fossils?
27. Name the 4 eras of earth’s history in order beginning with the oldest & going to present day.
28. Which organisms appeared first in earth’s history?
29. Name the period in which each of these events occurred:
a. Humans appeared?
b. Mass extinction of dinosaurs?
c. Land plants appeared?
d. Birds appeared?
e. Fish appeared?
f. Reptiles appeared?
g. Modern mammals appeared?
30. What is true about fossils in the same rock layer or strata?
31. a. What is meant by mass extinction and give an example?
b. Has there been more than one mass extinction in earth’s history? Explain.
32. Radioactive isotopes are used to determine the ___________________ age of fossils.
Section 15-2 Theories of Evolution
33. What was Lamarck’s hypothesis about how species change?
34. What is meant by acquired trait?
35. What did Lamarck think had caused the webbed feet of water birds?
36. How did Lamarck think the offspring of these web-footed birds got their web-feet?
37. Can traits be passed in this way? Explain why or why not.
38. Was Lamarck’s idea of acquired traits proven or rejected?
39. Charles Darwin’s idea of how species change over time is called what?
40. Explain natural selection.
41. What other scientist came up with this same idea about evolution?
42. What was the name of the book that Darwin published with his ideas about evolution?
43. Explain how Darwin obtained all of his records & supporting evidence that led him to his theory of natural selection.
44. Describe the voyage of the H.M.S. Beagle.
45. What book did Darwin read that influenced his ideas when he sailed on the Beagle? Who was the author?
46. What unusual information did Darwin collect about the Galapagos finches on his voyage?
47. What forced Darwin to move ahead and publish his ideas?
48. State the 2 theories that sum up Darwin’s ideas about evolution & natural selection.
49. State Darwin’s Descent with Modification theory.
50. Darwin’s theory stated that all the Galapagos finches had descended from what?
51. State Darwin’s Modification by Natural Selection theory.
52. What idea, published in a book by Thomas Malthus, did Charles Darwin use?
53. According to Darwin, what limits the growth of populations?
54. How do populations of organisms adapt to their environment?
55. What is meant by the fitness of an organism?
Section 15-3 Evolution in Process
56. What are homologous structures and give an example?
57. What are analogous structures & give an example?
58. Which structures, homologous or analogous, show that organisms are more closely related?
59. What are vestigial structures & give an example?
60. Vestigial structures show __________________________ ancestry.
61. In the early stages, how do all vertebrate embryos compare with each other? What does this indicate?
62. Organisms with homologous (similar) _________________ & __________________ acids are probably more closely related.
63. What is co-evolution & give 2 examples of organisms that co-evolve?
64. Are a shark and a porpoise closely related? Explain your answer.
65. When does convergent evolution occur?
66. What is divergent evolution?
67. Divergent evolution usually results in new ________________________.
68. What is adaptive radiation & give an example?
69. How can adaptive radiation be sped up & give an example?
Section 16-1 Genetic Equilibrium
70. Do populations change quickly or slowly?
71. What is the study of evolution using genetics called?
72. Do individuals evolve?
73. Do populations evolve?
74. Variations in traits may be due to ___________________ factors or __________________.
75. Name the 3 ways variations in genotypes arise.
76. What is meant by the gene pool?
77. How many alleles exist for each trait in a gene pool?
78. How is allele frequency determined?
79. Determine the frequency of each allele in the following genotypes:
a. AA?
b. Aa?
c. aa?
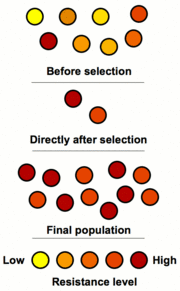
80. How is phenotypic ratio determined?
81. What is the phenotypic frequency of white, pink, & red four-o-clocks in these crosses?
a. RR x rr ?
b. Rr x Rr ?
c. Rr x rr ?
82. The frequency of alleles or phenotypes in a cross must always add up to ______________.
83. What does the Hardy-Weinberg theory state about allele frequencies in a population?
84. List the 5 conditions that must hold true in the ideal population for Hardy-Weinberg to be correct.
85. Would the Hardy-Weinberg law apply to real populations?
Section 16-2 Disruption of Genetic Equilibrium
86. What effect does evolution have on a population’s genetic material?
87. Does evolution affect allele frequencies?
88. Any violations of the 5 conditions necessary for Hardy-Weinberg can result in _____________.
89. A change in genetic material is known as a _______________________.
90. Mutations occur constantly at _______________ rates unless an organism is exposed to ___________.
91. Most mutations are __________________, but some may be _____________________ & help the population evolve & survive.
92. Compare & contrast population immigration & emigration.
93. What is gene flow & give an example?
94. Does the Hardy-Weinberg law apply to small and medium sized populations?
95. What is genetic drift?
96. In what size populations does genetic drift apply & explain why?
97. Do all populations mate randomly? Explain.
98. What is the effect of matings of related individuals?
99. What is the most significant factor that affects genetic equilibrium?
100. Name the 4 types of natural selection & explain each.
a.
b.
c,
d.
101. Give an example of stabilizing selection using body size of lizards.
102. Give an example of directional selection using tongue length of anteaters.
103. Give an example of disruptive selection using limpets.
104. Explain the following — “genes of successful reproducers, rather than those of successful survivors, are amplified through natural selection.”
Section 16-3 Formation of Species
105. Do new species ever form? Do old species ever disappear? Explain your answer.
106. Define speciation.
107. Are all new species similar to their ancestral species? Explain.
108. What did scientists use for many years to help classify organisms?
109. What is morphology?
110. What are some limitations of using only morphology to identify organisms?
111. What is the biological species concept?
112. Does this concept help identify extinct species & why?
113. Members of a species are _________________similar & can __________________ to produce _____________________ offspring.
114. What does speciation begin with & does it affect mating?
115. Name 2 important types of isolation.
a.
b.
116. Define geographic isolation.
117. Give an example of how this type of isolation could occur.
118. What happens to the 2 subgroups after being geographically isolated from each other?
119. Define reproductive isolation.
120. Name & describe the 2 types of reproductive isolations.
a.
b.
121. Not recognizing mating calls or having different breeding times are examples of what type of isolation?
122. An infertile mule produced when a donkey and a horse mate is an example of ______________________ isolation.
123. Speciation often requires ______________________ of years.
124. Can some species form more quickly than others? Explain.
125. Does fossil record support a slow, gradual or more “instant” change in species?
126. A more “instant” formation or change occurs in ___________________, not millions of years.
127. What is this type of quicker species formation called?
128. What does punctuated equilibrium mean?