| DNA Technology All Materials © Cmassengale |
 |
Introduction:
- Biotechnology refers to technology used to manipulate DNA
- The procedures are often referred to as genetic engineering
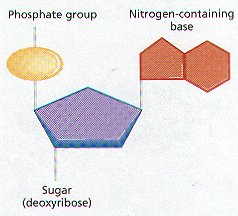
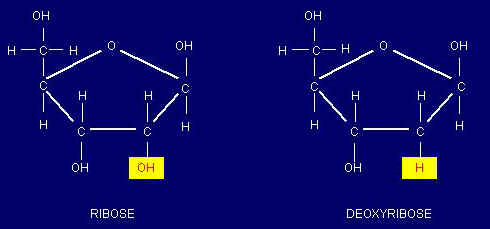
- DNA is the genetic material of all living organisms
- All organisms use the same genetic code
- Genes from one kind of organism can be transcribed and translated when put into another kind of organism
- For example, human and other genes are routinely put into bacteria in order to synthesize products for medical treatment and commercial use
- Human insulin, human growth hormone, and vaccines are produced by bacteria
- Recombinant DNA refers to DNA from two different source
- Individuals that receive genes from other species are transgenic
Viruses & their Structure:
- Viruses contain genetic material but are not living
- Host cells are required for their reproduction
- Viruses are composed of an inner nucleic acid core (genetic material) and an outer protein coat (capsid)
- Viruses that infect animals have an outer envelope (membrane) that is derived from the cell membrane of the host cell may surround the capsid
- The genetic material in some viruses is DNA; in others it is RNA

Viral Reproduction:
- When viral genetic material enters a cell, it is replicated, transcribed (mRNA is produced) and translated (proteins are produced from the mRNA) by the host cell
- By this process, the host cell uses the genetic instructions in the virus to make more viruses
Viral DNA ® mRNA ® protein
- If the viral genetic material is RNA, a DNA copy must first be made before transcription and translation can occur
- The DNA copy of the viral RNA is called cDNA.
viral RNA ® cDNA ® mRNA ® protein
Bacteriophages:
- Bacteriophages are viruses that infect bacteria
- Not surrounded by a membrane as the animal-infecting viruses
- Virus attaches to the bacteria cell, a viral enzyme digests away a part of the wall, and its viral DNA enters the host cell
- Inside the host cell, the viral DNA is transcribed, translated, and replicated
- Translation produces protein coats and the enzymes needed in the construction of new virus particles
- Viral DNA is replicated
- The protein coats and DNA are assembled into new viral particles
- The host cell wall to ruptures releasing the newly formed viruses

- Upon entering the cell, the viral DNA may instead, become integrated into the bacterial DNA
- It is replicated along with the host DNA when the host reproduces
- Eventually, it will become transcribed and translated
Retroviruses:
- Contain RNA & the enzyme reverse transcriptase
- Reverse transcriptase can make a DNA copy of the viral RNA
- The new DNA produced from the RNA template is called cDNA
- DNA synthesis follows the production of cDNA to produce a double-helix
- cDNA then becomes incorporated into the host DNA (called a prophage)
- The new viruses escape the host cell by budding
- The AIDS virus (HIV) is an example of a retrovirus
Vectors
- Vectors are used to transfer genes into a host cell
- Plasmids & viruses are the most commonly used vectors
- A vector must be capable of self-replicating inside a cell
- Viruses are the vectors of choice for animal cells
- Marker genes can be used to determine if the gene has been taken up
Plasmids:
- Small rings of DNA in bacterial cells
- Used to transfer genes to other organisms
- Host bacterium takes up the plasmid, which includes the foreign gene
- When bacteria reproduce, plasmids with the new gene are also reproduced
- This clones (copies) the gene each time the bacteria reproduces
Viruses:
- Can accept larger amounts of DNA than plasmids
- Once the virus enters the host cell, it also reproduces the foreign gene it carries
- The copied gene is “cloned”
Restriction enzymes:
- Restriction enzymes were discovered in bacteria
- Bacteria use them as a defense mechanism to cut up the DNA of viruses or other bacteria
- Hundreds of different restriction enzymes have been isolated
- Each restriction enzyme or RE cuts DNA at a specific base sequence
- For example, EcoRI always cuts DNA at GAATTC as indicated below
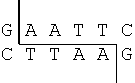
- The sequence GAATTC appears three times in the DNA strand below. As a result, the strand is cut into four pieces
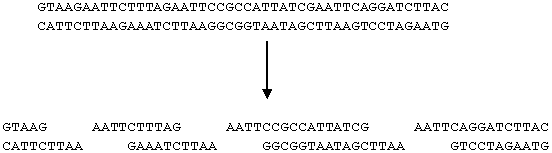
- Other restriction enzymes cut at different sites, some examples are listed below
| Enzyme | Cutting Site |
| Bam HI | GGATCC |
| Hae III | GGCC |
| Pst I | CTGCAG |
| Hind I | GANTC |
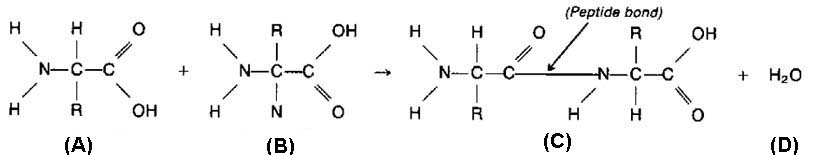
Sticky Ends & Recombinant DNA:
- Fragments of DNA that has been cut with restriction enzymes have unpaired nucleotides at the ends called sticky ends
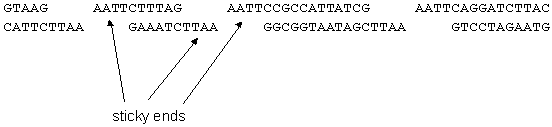
- Sticky ends have complimentary bases, so they could rejoin
- If the vector and the gene to be cloned are both cut with the same restriction enzyme, they will both have complimentary sticky ends
- After cutting, the 2 DNA samples are mixed
- Fragments with complementary sticky ends join together forming recombinant DNA (contains gene from vector & the gene to be cloned)
- Enzyme DNA ligase seals the fragments together
- Bacteria such as Escherichia coli are capable of taking up DNA from their environment
- This process is called transformation
- CaCl2 and a procedure called heat shock are used to make E. coli cells more permeable so that they take up the modified plasmids more readily
Genomic Libraries:
- A genome is all of the genes in a particular organism
- Bacteria or virus vectors can be used to store fragments of the DNA from another species
- The DNA is cut up into fragments, and the different fragments are inserted into bacteria or viruses
- The collection of bacteria or viruses is called a genomic library
Polymerase Chain Reaction (PCR):
- Used to make many copies of small pieces of DNA
- Procedure requires primers, DNA polymerase, and nucleotides
- Primers are short chains of about 20 nucleotides that are complimentary to a region in the DNA to be amplified
- DNA polymerase cannot continue the process unless it has already been started by primers
- Nucleotides are needed because DNA is composed of nucleotide “building blocks”

- The DNA is heated to approximately 95o C to separate the two strands of the double helix

- After the strands are separated, the DNA is cooled to about 50o C, and the primers attach
- The temperature is raised to approximately 70o C so the polymerase will attach to & copy the strand


- The DNA replication process repeats itself as the solution is then heated and cooled at regular intervals

DNA Fingerprinting (RFLP Analysis):
- In RFLP analysis, the DNA of an organism is cut up into fragments using restriction enzymes producing a large number of short fragments of DNA
- Because no two individuals have identical DNA, no two individuals will have the same length fragments
- Gel electrophoresis is a technique used to separate the DNA fragments according to their size
- The fragments are placed in wells on a sheet of gelatin, and an electric current is applied to the sheet
- DNA is negatively charged and will move in an electric field toward the positive pole

- The smallest fragments will move the fastest because they are able to move through the pores in the gelatin faster
- Bands will be produced on the gelatin where the fragments accumulate
- Shortest fragments will accumulate near one end of the gelatin (furthest from the wells), and the longer, slower-moving ones will remain near the other end
- DNA bands must be stained to make them visible

Gene Products & Uses of Genetic Engineering:
- E. coli is used to produce proteins such as insulin by genetic engineering because it is easily grown
- To recover the product, E. coli must be lysed or the gene must be linked to a gene that produces a naturally secreted protein
- Yeasts can be genetically engineered and are likely to secrete the gene product continuously
- Mammalian cells can be engineered to produce proteins such as hormones for medical use
- Plant cells take up a plasmid from Agrobacterium
- Plant cells can be engineered and used to produce plants with new properties such as Roundup Ready soybeans
- Pseudomonas bacteria has been engineered to produce Bacillus thuringiensis or BT
- BT bacteria make a toxin against insects, thus producing a natural insecticide (example – B.T. cotton)
- Animal viruses can be engineered to carry a gene for a pathogen’s surface protein so the virus can be used as a vaccine
- Genetic engineering techniques are being used to map the human genome through the Human Genome Project
- Could provide tools for diagnosis and possible repair of genetic disease
- Recombinant DNA techniques can be used for genetic fingerprinting
- Gene therapy can be used to cure genetic diseases by replacing the defective or missing gene
- Bovine growth hormone (BGH) increases milk production in cows by about 10%
Safety and Ethical Issues:
- Harmful organisms may be accidentally produced
- Organisms that are intended to be released in the environment may be engineered with genes that will eventually kill them
- There is little legislation on the use of genetic screening and information produced by screening
- The technology is increasing the ability to diagnose genetic diseases pre-natally, adding new complexity to the abortion controversy
- Ethical questions have been raised over whether we should modify the genes of humans
- Genetic screening and gene therapy are expensive and may be unavailable to the poor
- Biological weapons could be created using biotechnology




![[Glucose Straight Structure]](https://biologyjunction.com/images/glucose1.jpg)




![[Lecithin]](https://biologyjunction.com/images/lecithin.jpg)
![[Phospholipid Bilayer]](https://biologyjunction.com/images/membrane.jpg)