| Chapter 17 From Gene to Protein | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Objectives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BACK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chapter 17 From Gene to Protein | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Objectives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BACK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
 CHAPTER 8, CELL REPRODUCTIONSECTION 8-1, CHROMOSOMES
CHAPTER 8, CELL REPRODUCTIONSECTION 8-1, CHROMOSOMES
DNA is a long thin molecule that stores Genetic Information. The DNA in a human cell is estimated to consist of six billion pairs of nucleotides.
OBJECTIVES: Describe the structure of a chromosome. Compare prokaryotic chromosomes with eukaryotic chromosomes. Explain the differences between sex chromosomes and autosomes. Give examples of diploid and haploid cells.
CHROMOSOME STRUCTURE
1. During Cell Division, the DNA (CHROMATIN) in an Eukaryotic Cell’s Nucleus is coiled into very tight compact structures called CHROMOSOMES.(Figure 8-1)
2. Chromosomes are Rod Shaped structures made of DNA and Proteins.
3. The Chromosomes of stained Eukaryotic cells undergoing cell division are visible as darkened structures inside the Nuclear Membrane.
4. The DNA in Eukaryotic cells wraps tightly around Proteins called HISTONES. They help to maintain the shape of Chromosomes and aid in the tight packing of DNA.
5. Proteins called NONHISTONE Proteins Do Not participate in packing of DNA, they are involved in Controlling the Activity of Specific Regions of the DNA.
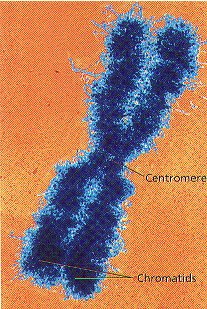
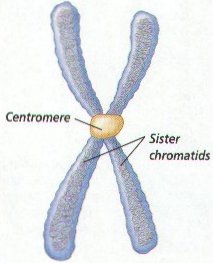 6. When preparing for Cell Division, Chromosomes form Copies of themselves, Each half of the Chromosome is called a CHROMATID or SISTER CHROMATIDS. Chromatids form as the DNA makes copies of itself before cell division. (Figure 8-2)
6. When preparing for Cell Division, Chromosomes form Copies of themselves, Each half of the Chromosome is called a CHROMATID or SISTER CHROMATIDS. Chromatids form as the DNA makes copies of itself before cell division. (Figure 8-2)
7. The constricted area of each Chromatid is called a CENTROMERE . The Centromere holds the Two Chromatids together until the separate during Cell Division.
8. Between Cell Division, DNA IS NOT so Tightly Coiled into Chromosomes. The Less tightly coiled DNA-Protein complex is called CHROMATIN .
9. Chromosomes are simpler in prokaryotes. The DNA of most Prokaryotes comprises only ONE Chromosome, which is attached to the inside of the Cell Membrane.
10. Prokaryotic Chromosomes consist of a circular DNA Molecule and associated Proteins.
CHROMOSOME NUMBERS
1. EACH HUMAN BODY CELL CONTAINS 46 CHROMOSOMES, (2n) OR TWO COMPLETE SETS.
2. ANY CELL THAT CONTAINS TWO COMPLETE SETS OF CHROMOSOMES IS CALLED A DIPLOID CELL. A Diploid Cell is commonly abbreviated as 2n.
3. THE NUMBER OF CHROMOSOMES IN A DIPLOID CELL IS CALLED THE DIPLOID NUMBER. EVERY ORGANISM HAS A CHARACTERISTIC DIPLOID NUMBER (2n).
4. EXAMPLES: FRUIT FLIES – 8, LETTUCE – 14, GOLDFISH 94, AND HUMANS 46.
5. A CELL WITH ONLY ONE COMPLETE SET OF CHROMOSOMES IS CALLED A HAPLOID CELL.
A Haploid Cell is abbreviated as 1n.
6. GAMETES, EGGS AND SPERM CONTAIN ONLY ONE COMPLETE SET. EACH HUMAN SPERM OR EGG (GAMETE) CONTAINS 23 CHROMOSOMES, THE HAPLOID NUMBER (1n) FOR ALL HUMANS.
7. WHEN AN EGG AND A SPERM OF THE SAME TYPE OF ORGANISM JOIN TO PRODUCE A NEW INDIVIDUAL, THE PROCESS IS CALLED FERTILIZATION.
8. THE SINGLE CELL THAT RESULTS FROM FERTILIZATION IS KNOWN AS A ZYGOTE. THE ZYGOTE CONTAINS TWO COMPLETE SETS OF CHROMOSOMES, ONE SET FROM EACH GAMETE, FORMING A DIPLOID CELL. IN MOST MULTICELLULAR ORGANISMS, THE ZYGOTE IS THE FIRST CELL OF THE NEW INDIVIDUAL.
 9. The Chromosomes in the Zygote exist in PAIRS. For every Chromosome that was in the egg, there is a matching Chromosome from the sperm.
9. The Chromosomes in the Zygote exist in PAIRS. For every Chromosome that was in the egg, there is a matching Chromosome from the sperm.
10. Human and Animal Chromosomes are categorized as either SEX CHROMOSOMES or AUTOSOMES.
11. SEX CHROMOSOMES are Chromosomes that Determine the SEX of an Organism.
12. In Humans, Sex Chromosomes are either X or Y. Females have TWO X Chromosomes and Males have an X and Y Chromosome.
13. All the Other Chromosomes in an Organism are called AUTOSOMES.
14. TWO of the 46 Human Chromosomes are Sex Chromosomes, while the reaming 44 are Autosomes.
 15. MATCH SET OF AUTOSOMES IN A DIPLOID CELL ARE CALLED HOMOLOGOUS PAIRS. BOTH CHROMOSOMES IN A HOMOLOGOUS PAIR CONTAIN INFORMATION THAT CODE THE SAME TRAIT (GENES). Example Eye Color.
15. MATCH SET OF AUTOSOMES IN A DIPLOID CELL ARE CALLED HOMOLOGOUS PAIRS. BOTH CHROMOSOMES IN A HOMOLOGOUS PAIR CONTAIN INFORMATION THAT CODE THE SAME TRAIT (GENES). Example Eye Color.
SECTION 8-2, CELL DIVISION
All cells are derived from preexisting cells. Cell division is the process by which cells produce offspring cells. Cell division differs in prokaryotes and eukaryotes. In eukaryotes, cell division differs in different stages of an organisms life cycle.
OBJECTIVES: Describe the events of binary fission. Describe each phase of the cell cycle. Summarize the phases of mitosis. Compare cytokinesis in animal cells with cytokinesis in plant cells.
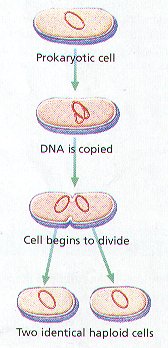 CELL DIVISION IN PROKARYOTES
CELL DIVISION IN PROKARYOTES
1. BINARY FISSION is the Division of a Prokaryotic cell INTO TWO Offspring Cells.
2. Binary Fission consist of THREE General Stages: (Figure 8-4):
STAGE 1 – The Chromosome, which is attached to the Inside of the Cell Membrane, makes a COPY of Itself, Resulting in Two Identical Chromosomes Attached to the Inside of the Inner Cell Membrane.
STAGE 2 – The Cell continues to grow until it reaches approximately TWICE its Normal Size. Then a CELL WALL Begins forms between the Two Chromosomes.
STAGE 3 – The Cell SPLITS into TWO NEW CELLS. Each New Cell contains on the Identical Chromosomes.
CELL DIVISION IN EUKARYOTES
1. The trillions of cells that make up your body came from just ONE ORIGINAL CALLED: A FERTILIZED EGG (Zygote). The Cell Theory states “CELLS COME ONLY FROM THE REPRODUCTION OF EXISTING CELLS” Chapter 4.
2. Each time A Cell Reproduces, the NEW Cells that are formed contained all the ESSENTIAL CYTOPLASM, ORGANELLES, AND NUCLEIC ACIDS NEEDED TO SURVIVE AND FUNCTION.
3. A Cell typically goes through PHASES during its Life, performing life processes of GROWTH AND DEVELOPMENT before it divides into new cells.
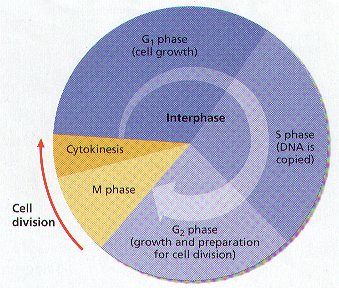 4. THE PHASES OF LIFE OF A CELL ARE CALLED THE CELL CYCLE . THE CELL CYCLE CONSISTS OF THREE PHASES:
4. THE PHASES OF LIFE OF A CELL ARE CALLED THE CELL CYCLE . THE CELL CYCLE CONSISTS OF THREE PHASES:
A. INTERPHASE
B. MITOSIS
C. CYTOKINESIS.
5. The CELL CYCLE is the Repeating Events that make up the Life of a Cell. (Figure 8-5)
6. Cell Division is One Phase of the Cell Cycle. Cell Division consists of MITOSIS AND CYTOKINESIS.
7. MITOSIS is a Series of PHASES in Cell Division during which the NUCLEUS of a Cell Divides into TWO NUCLEI WITH IDENTICAL GENETIC MATERIAL. MITOSIS OCCURS ONLY IN EUKARYOTES.
INTERPHASE
1. INTERPHASE IS THE PORTION OF THE CELL CYCLE BETWEEN DIVISION.
2. Interphase is the LONGEST Phase in the Cell Cycle of a typical Cell. Interphase used to be referred to as the “RESTING PHASE”.
3. During Interphase, calls carry on all their usual functions, such as respiration and enzyme production. The Cell also GROWS and DEVELOPS into MATURE FUNCTIONING Cells while in Interphase. It is the period of normal metabolic activity.
4. INTERPHASE CONSIST OF THREE PHASES:
A. G1 PHASE – PERIOD OF NORMAL METABOLIC CELLULAR ACTIVITIES: THE NUMBER OF ORGANELLES AND AMOUNT OF CYTOPLASM IN A CELL INCREASE. Offspring Cells Grow to Mature Size.
B. S PHASE – THE GENETIC MATERIAL (DNA) IS DUPLICATED (COPIED). THE CHROMOSOMES OF THE CELL REPLICATE.
C. G2 PHASE – Structure directly involved with mitosis are formed. The Cell makes the Organelles and substances it needs for Cell Division. A time during which the Cell prepares to divide.
 5. REPLICATION IS THE PROCESS OF COPYING GENETIC MATERIAL.
5. REPLICATION IS THE PROCESS OF COPYING GENETIC MATERIAL.
6. REPLICATION RESULTS IN TWO IDENTICAL COPIES OF A CHROMOSOME CALLED SISTER CHROMATIDS.
7. CHROMOSOMES MUST REPLICATE DURING INTERPHASE SO THERE WILL BE A COMPLETE COPY OF EACH CHROMOSOME IN EACH NEW CELL.
8. BECAUSE THE DNA CONTAINED IN CHROMOSOMES CONTROL GROWTH DEVELOPMENT, AND FUNCTION OF EVERY CELL, EACH NEW CELL MUST HAVE AN EXACT COPY OF THE ORIGINAL SET OF CHROMOSOMES.
CELL DIVISION
1. CELL DIVISION IS THE PROCESS BY WHICH ONE CELL PRODUCES TWO NEW IDENTICAL DAUGHTER CELLS.
2. CELL DIVISION INVOLVES TWO STEPS: CALLED MITOTIC CELL DIVISION.
A. MITOSIS – FIRST STEP. A SERIES OF PHASES IN CELL DIVISION DURING WHICH THE NUCLEUS OF A CELL DIVIDES INTO TWO NUCLEI WITH IDENTICAL GENETIC MATERIAL.
B. CYTOKINESIS – SECOND STEP. THE CYTOPLASM OF THE CELL DIVIDES INTO TWO NEW CELLS CALLED DAUGHTER CELLS.
3. DAUGHTER CELL NUCLEI ARE IDENTICAL TO THE PARENT CELL NUCLEUS IN EVERY WAY. LIKE THEIR PARENT CELL, SOME DAUGHTER CELLS WILL PASS THROUGH THE CELL CYCLE OF GROWTH, DEVELOPMENT, AND CELL DIVISION.
4. MULTICELLULAR ORGANISMS GROW AS MORE CELLS REPEAT THE CYCLE OF CELL DIVISION AND GROWTH.
MITOSIS
1. Mitosis is the Division of the Nucleus, which occurs during Cell Division.
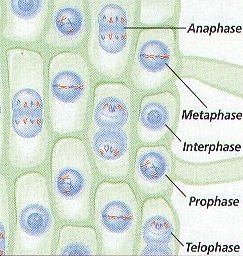
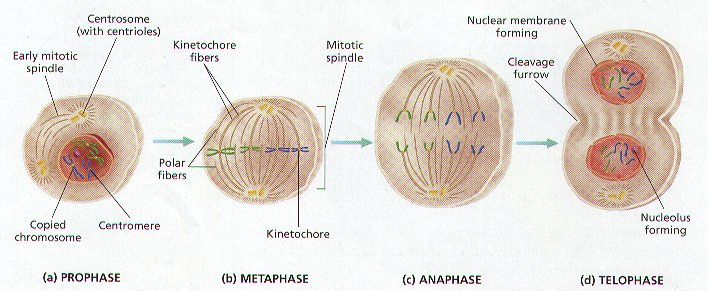
2. Biologist have named the Steps, or Phases, of Mitosis to help study the process. The FOUR Phases of Mitosis are called PROPHASE, METAPHASE, ANAPHASE, AND TELOPHASE. (Figure 8-6)
3. THE ACTUALLY PROCESS OF MITOSIS IS CONTINUOUS.
4. MITOSIS IS THE PROCESS BY WHICH A NUCLEUS GIVES RISE TO TWO IDENTICAL NUCLEI.
5. INTERPHASE PRIOR TO MITOSIS, THE PERIOD OF NORMAL METABOLIC ACTIVITY. The Chromosomes REPLICATE and the CYTOPLASM Increases as he cell prepares to divide. Interphase includes G1, S, G2 Phases of the Cell Cycle.
FOUR PHASES OF MITOSIS
PHASE 1- PROPHASE (Figure 8-6 (a))
1. Chromatin condenses into Chromosomes of TWO Sister Chromatids joined together by the CENTROMERE, and visible when viewed through a microscope.
2. THE NUCLEOLUS AND NUCLEAR MEMBRANE DISAPPEAR.
3. TWO Structures called CENTROSOMES appear next to the Disappearing Nucleus. In Animal Cells, each Centrosome contains a pair of small, cylindrical bodies called CENTRIOLES. Plant Cells lack Centrioles.
4. In BOTH Animal and Plant Cells, the Centrosomes move toward opposite poles of the cell. As they Separate, SPINDLE FIBERS made of microtubules radiate from the Centrosomes in preparation for Mitosis. The array of Spindle fibers is called the MITOTIC SPINDLE, which serves to Equally divides the Sister Chromatids between the Two Offspring Cells.
5. There are TWO Type of Spindle Fibers:
A. KINETOCHORE FIBERS – They Attached to the Centromere Region of each Sister Chromatids.
B. POLAR FIBERS – they extend across the dividing cell from Centrosome to Centrosome.
PHASE 2 – METAPHASE (Figure 8-6 (b))
1. The Chromosomes are moved to the CENTER of the CELL (Equatorial Plane) by the Kinetochore Fibers attached to the Centromeres.
2. The Two Sister Chromatids of each Chromosome are attached to Kinetochore Fibers radiating from OPPOSITE ENDS OF THE CELL.
PHASE 3 – ANAPHASE (Figure 8-6 (c))
1. The Centromeres of Each Chromosome are pulled by the Kinetochore Fibers toward the ends of the cell (OPPOSITE POLES).
2. THE SISTER CHROMATIDS ARE THUS SEPARATED FROM EACH OTHER. They are now Considered to be Individual Chromosomes.
PHASE 4 – TELOPHASE (Figure 8-6 (d))
1. After the Chromosomes reach opposite ends of the Cell, the Spindle Fibers Disassemble.
2. The Chromosomes return to less tightly coiled Chromatin State.
3. New Nuclear Envelope begins to form around the Chromosomes at each end of the cell.
4. CYTOKINESIS BEGINS.
5. THE PROCESS OF MITOSIS IS NOW COMPLETE. THE CELL MEMBRANE BEGINS TO PINCH THE CELL IN TWO AS CYTOKINESIS BEGINS.
CYTOKINESIS
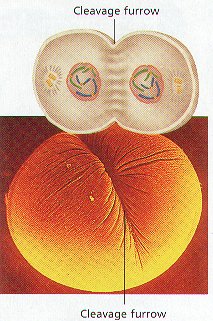 1. Following the last phase of Mitosis, Cytokinesis COMPLETES the process of Cell Division.
1. Following the last phase of Mitosis, Cytokinesis COMPLETES the process of Cell Division.
2. During Cytokinesis, the Cytoplasm of a cell and its ORGANELLES SEPARATE INTO TWO NEW DAUGHTER CELLS.
3. Cytokinesis proceeds differently in animal and plant cells.
4. CYTOKINESIS OF ANIMAL CELLS: The Cytoplasm Divides when a GROOVE called the CLEAVAGE FURROW forms through the Middle of the Parent Cell. The Cleavage Furrow Deepens until the parent cell pinches into TWO New Identical Cells. The New Cells are Now in INTERPHASE.
5. CYTOKINESIS OF PLANT CELLS: In a Plant Cell, the material for NEW CELL WALL CALLED THE CELL PLATE AND MEMBRANES GATHER AND FUSE ALONG THE EQUATOR, OR MIDDLE OF THE CELL, BETWEEN TWO NUCLEI. Forming TWO New Identical Cells.
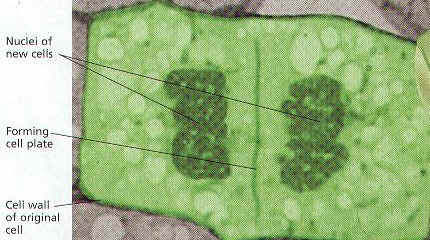
6. In Both Animal and Plant Cells, New Offspring Cells are approximately equal in Size.
SECTION 8-3, MEIOSIS
Meiosis is a process of nuclear division that Reduces the number of chromosomes in new cells to Half the number in the original cell. The Halving of the chromosome number counteracts a fusion of cells later in the life cycle of the organism. For example, in humans, meiosis produces haploid reproductive cells called GAMETES. Human gametes are sperm and egg cells, each which contains 23(1n) chromosomes. The fusion of sperm and egg results in a zygote that contains 46 (2n) chromosomes.
OBJECTIVES: List and describe the phases of meiosis. Compare the end products of mitosis with those of meiosis. Explain crossing-over and how it contributes to the production of unique individuals. Summarize the major characteristics of spermatogenesis and oogenesis.
1. Most organisms are capable of COMBINING CHROMOSOMES FROM TWO PARENTS TO PRODUCE OFFSPRING.
2. WHEN CHROMOSOMES OF TWO PARENTS COMBINE TO PRODUCE OFFSPRING, THE PROCESS IS KNOWN As SEXUAL REPRODUCTION.
3. THE CHROMOSOMES THAT COMBINE DURING SEXUAL REPRODUCTION ARE CONTAINED IN SPECIAL REPRODUCTIVE CELLS CALLED GAMETES.
4. IN MOST ORGANISMS, GAMETES CAN BE EITHER EGG OR SPERM .
5. EGGS are larger than sperm and contain a lot of Cytoplasm. An egg is nonmotile.
6. SPERM Cells contain very little Cytoplasm, have Flagella, that helps them swim to the nonmotile egg.
7. The Chromosomes of Two Gametes are added together when they join. The number of Chromosomes in the offspring DOES NOT DOUBLE WITH EACH GENERATION, BUT REMAINS THE SAME BECAUSE OF MEIOSIS.
8. MEIOSIS IS THE WAY MANY ORGANISMS PRODUCE GAMETES THROUGH A TYPE OF CELL REPRODUCTION.
9. MEIOSIS IS A TYPE OF NUCLEAR DIVISION IN WHICH THE CHROMOSOME NUMBER IS HALVED. LIKE MITOSIS, MEIOSIS IS FOLLOWED BY CYTOKINESIS.
10. IN HUMANS SPECIALIZED REPRODUCTIVE CELLS WITH 46 CHROMOSOMES (2n) (DIPLOID CELL) UNDERGO MEIOSIS AND CYTOKINESIS TO GIVE RISE TO EGG OR SPERM THAT HAVE ONLY 23 CHROMOSOMES (1N) (HAPLOID CELL) EACH.
11. MEIOSIS ONLY OCCURS IN EUKARYOTIC CELLS IN PHASES SIMILAR TO THE PHASES OF MITOSIS.
12. MEIOSIS IS DIFFERENT FROM MITOSIS IN SOME VERY IMPORTANT WAYS.
A. The process of meiosis results in the production of Daughter Cells that have HALF THE NUMBER OF CHROMOSOMES OF THE PARENT CELL (HAPLOID CELL).
B. Daughter Cell produced by meiosis ARE NOT ALL ALIKE. THE DAUGHTER CELLS MAY HAVE DIFFERENT CHROMOSOMES FROM EACH OTHER.
C. The NUMBER OF CELLS PRODUCED BY MEIOSIS IS DIFFERENT.
(1) Mitosis – One Parent Cell PRODUCES TWO DIPLOID DAUGHTER CELLS.
(2) Meiosis – One Parent Cell PRODUCES FOUR HAPLOID DAUGHTER CELLS.
STAGES OF MEIOSIS
1. THE PROCESS OF MEIOSIS SEPARATES THE PAIRS OF CHROMOSOMES IN A DIPLOID CELL TO FORM HAPLOID CELLS.
2. ONE PARENT CELL DIVIDES TWICE TO PRODUCE FOUR HAPLOID DAUGHTER CELLS.
3. DURING MEIOSIS, THE NUMBER OF CHROMOSOMES IN EACH CELL IS REDUCED FROM DIPLOID TO HAPLOID BY SEPARATING HOMOLOGOUS PAIRS OF CHROMOSOMES.
4. MEIOSIS PROCEEDS IN TWO MAIN STAGES:
A. MEIOSIS I HOMOLOGOUS PAIRS ARE SEPARATED.
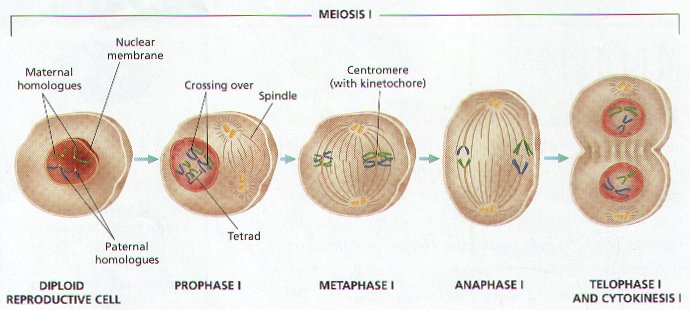
B. MEIOSIS II THE SISTER CHROMATIDS OF EACH CHROMOSOME ARE SEPARATED.
MEIOSIS I (Figure 8-9)
1. AT THE START OF MEIOSIS I EACH CHROMOSOME CONSIST OF TWO STRANDS OF SISTER CHROMATIDS CONNECTED AT THE CENTROMERE.
2. HOMOLOGOUS PAIRS OF CHROMOSOMES COME TOGETHER BEFORE MEIOSIS BEGINS, AN EVENT THAT DOES NOT OCCUR IN MITOSIS. THIS EVENT IS CALLED SYNAPSIS .
3. Each Pair of Homologous Chromosomes is called a TETRAD .
PROPHASE I.
1. Chromosomes become thick and visible, the chromosomes of each homologous pair are tangled together.
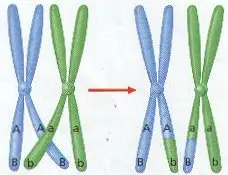 2. Portions of Chromatids may Break Off and attach to Adjacent Chromatids on the homologous Chromosome – a process called CROSSING-OVER. (Figure 8-10)
2. Portions of Chromatids may Break Off and attach to Adjacent Chromatids on the homologous Chromosome – a process called CROSSING-OVER. (Figure 8-10)
3. Crossing-Over results in Genetic Recombination by producing a New Mixture of Genetic Material.
4. Each pair consists of FOUR CHROMATIDS, BECAUSE EACH CHROMOSOME IN THE PAIR HAD REPLICATED BEFORE MEIOSIS BEGAN.
5. The Nucleoli and the Nuclear Envelope disappear and the spindle fibers form.
METAPHASE I. Homologous pairs (Tetrads) are still together and arrange in the middle of the cell.
ANAPHASE I. The homologous pairs of chromosomes separate from each other, spindle fibers pull one member from each pair to opposite ends of the cell. The Random separation of the Homologous Chromosomes is called INDEPENDENT ASSORTMENT.
TELOPHASE I. Cytokinesis takes place; each new cell is haploid, containing one chromosome
from each pair.
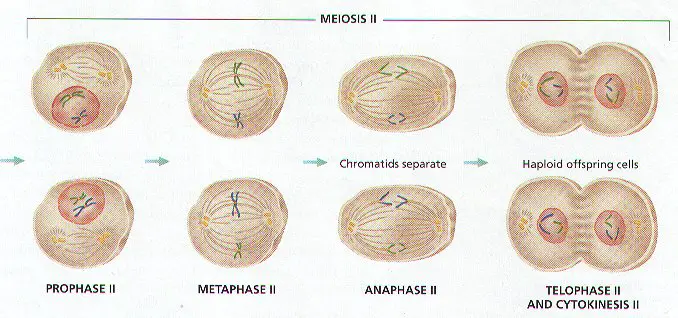
MEIOSIS II (Figure 8-11)
1. CHROMOSOMES DO NOT REPLICATE BEFORE BEGINNING THE SECOND PHASE MEIOSIS II WILL DIVIDE CHROMOSOMES INTO HAPLOID CELLS CALLED GAMETES.
2. Each Diploid Cell from Meiosis I will go through a second division, forming the FOUR GAMETES HAPLOID CELL. (Review Figure 8-11)
CROSSING-OVER
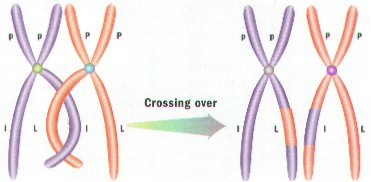 1. CHROMOSOMES OF ALL ORGANISMS CONTAIN REGIONS CALLED GENES .
1. CHROMOSOMES OF ALL ORGANISMS CONTAIN REGIONS CALLED GENES .
2. EACH GENE CODES FOR ONE TRAIT, OR CHARACTERISTIC, OF THE ORGANISM.
3. ONE VERY IMPORTANT EVENT THAT CAN OCCUR DURING MEIOSIS I IS CROSSING- OVER.
4. CROSSING-OVER IS THE EXCHANGE OF GENES BETWEEN PAIR OF HOMOLOGOUS CHROMOSOMES.
5. CROSSING-OVER OCCURS ONLY DURING PROPHASE I (ONLY!) WHEN HOMOLOGOUS PAIRS ARE STILL JOINED TOGETHER. THESE PAIRS CAN SOMETIMES BREAK WHERE THEY MEET AN EXCHANGE GENES. (Figure 8-10)
FORMATION OF GAMETES
 1. In Animals, meiosis produces haploid reproductive cells called GAMETES.
1. In Animals, meiosis produces haploid reproductive cells called GAMETES.
2. Meiosis occurs within the Reproductive Organs, in the TESTES or OVARIES.
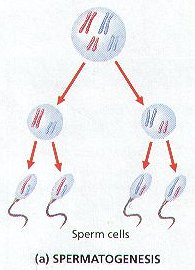
3. In the Testes, meiosis is involved in the production of Male Gametes known as Sperm Cells or Spermatozoa.
4. In the development of Sperm Cells, a Diploid Reproductive Cell divides Meiotically to form FOUR Haploid Cells called SPERMATIDS.
5. Each Spermatid then develops into a Mature Sperm Cell.
6. The production of Sperm Cells is called SPERMATOGENESIS . (Figure 8-12 (b))
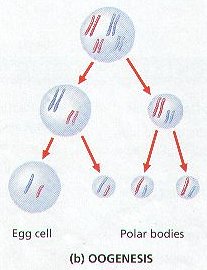 7. OOGENESIS is the production of Mature Egg Cells or OVA. (Figure 8-12 (c))
7. OOGENESIS is the production of Mature Egg Cells or OVA. (Figure 8-12 (c))
8. Notice that the Female only produces ONE EGG (OVUM) under normal circumstances.
9. Although creating 4 Haploid Cells through meiosis, only One Becomes the Egg, the other Three products of meiosis are called POLAR BODIES ,and Degenerate. This is due to the unequal dividing of the cytoplasm during Cytokinesis I & II.
ASEXUAL AND SEXUAL REPRODUCTION
1. EVOLUTION IS THE PROCESS OF CHANGE IN LIVING POPULATIONS OVER TIME.
2. ASEXUAL REPRODUCTION is the production of Offspring from ONE PARENT.
3. Asexual reproduction DOES NOT Usually involve Meiosis or the Union of Gametes.
4. In Unicellular Organisms, such as bacteria, New Organisms are created by either BINARY FISSION or MITOSIS.
5. Asexual Reproduction in multicellular organisms results from BUDDING OFF a Portion of Their Bodies. (Plants)
6. The Offspring From Asexual Reproduction are Genetically Identical to the Parent.
7. SEXUAL REPRODUCTION is the Production of Offspring through Meiosis and the Union of a Sperm and an Egg.
8. MEIOSIS AND SEXUAL REPRODUCTION RESULTS IN NEW COMBINATIONS OF CHARACTERISTICS WITHIN A POPULATION.
9. ORGANISMS IN A POPULATION THAT REPRODUCE SEXUALLY ARE NOT ALL ALIKE.
10. DIFFERENCES AMONG MEMBERS OF A POPULATION ARE COLLECTIVELY CALLED VARIATION. WHICH RESULTS FROM THE RECOMBINATION OF GENES DURING MEIOSIS AND FERTILIZATION.
11. MEIOSIS AND FERTILIZATION SHUFFLE THE GENES FROM PARENT ORGANISMS, PRODUCING NEW COMBINATIONS OF GENES IN THE OFFSPRING.
12. AN ORGANISMS CHARACTERISTICS ENABLE IT TO SURVIVE IN IT’S ENVIRONMENT. THE CONDITIONS OF THE ENVIRONMENT DETERMINE WHICH CHARACTERISTICS OR TRAITS BENEFIT THE SURVIVAL AND WHICH DO NOT.
13. THE ORGANISMS WITH THE TRAITS TO SURVIVE WILL THEN REPRODUCE TO PASS THOSE POSITIVE TRAITS ON TO THEIR OFFSPRING.
14. OVER TIME THIS PROCESS LEADS TO THE CHANGE IN THE POPULATIONS, BECAUSE ONLY THOSE WITH POSITIVE TRAITS TO PASS ON WILL REPRODUCE. NATURAL SELECTION.
15. THE ACCUMULATION OF SUCH GENES AND TRAITS IN EACH GENERATION IS THE BASIS OF EVOLUTION.
16. SINCE ASEXUAL OFFSPRING HAVE THE EXACT SAME GENES AND TRAITS AS THE PARENT, GENETIC VARIATION RARELY OCCURS.
17. A CHANGE IN THE ENVIRONMENT THAT CAN DESTROY ONE INDIVIDUAL COULD DESTROY THE ENTIRE POPULATION.
|
Name:
|
|
True/False
Indicate whether the sentence or statement is true or false.
|
||||||||||||||||||||||
| 1. |
When a tRNA anticodon binds to an mRNA codon, the amino acid detaches from the tRNA molecule and attaches to the end of a growing protein chain.
|
|||||||||||||||||||||
| 2. |
Only ribosomal RNA plays a role in translation.
|
|||||||||||||||||||||
| 3. |
During DNA replication, the molecule unzips and the exposed DNA nucleotides pair with other
specific nucleotides present in the nucleus
|
|||||||||||||||||||||
| 4. |
Humans pass exact copies of their DNA to their offspring.
|
|||||||||||||||||||||
| 5. |
Watson and Crick proposed a model of DNA
|
|||||||||||||||||||||
| 6. |
Amino acids are linked together by hydrogen bonds.
|
|||||||||||||||||||||
| 7. |
During transcription, the information on a DNA molecule is “rewritten” into an mRNA molecule.
|
|||||||||||||||||||||
| 8. |
All codons encode amino acids.
|
|||||||||||||||||||||
|
Multiple Choice
Identify the letter of the choice that best completes the statement or answers the question.
|
||||||||||||||||||||||
| 9. |
Purines and pyrimidines are
|
|||||||||||||||||||||
| 10. |
Chargaff’s rules, or the base-pairing rules, state that in DNA
|
|||||||||||||||||||||
| 11. |
ATTG : TAAC ::
|
|||||||||||||||||||||
| 12. |
Which of the following types of RNA carries instructions for making proteins?
|
|||||||||||||||||||||
| 13. |
Which of the following is not found in DNA?
|
|||||||||||||||||||||
| 14. |
Suppose that you are given a polypeptide sequence containing the following sequence of amino acids: tyrosine, proline, aspartic acid, isoleucine, and cysteine. Use the portion of the genetic code given in the table below to determine the DNA sequence that codes for this polypeptide sequence.
|
|||||||||||||||||||||
| 15. |
In order for protein synthesis to occur, mRNA must migrate to the
|
|||||||||||||||||||||
| 16. |
After the primary structure of a protein has been completed
|
|||||||||||||||||||||
| 17. |
Which of the following is not part of a molecule of DNA?
|
|||||||||||||||||||||
| 18. |
During replication in a molecule of DNA, one separation likely to occur is between
|
|||||||||||||||||||||
| 19. |
A gene may be described as
|
|||||||||||||||||||||
| 20. |
The synthesis of a new double strand of DNA begins when the two strand of the original DNA helix
|
|||||||||||||||||||||
| 21. |
Genes(DNA) affect cell structure and function by directing the synthesis of
|
|||||||||||||||||||||
| 22. |
Protein molecules are made up of
|
|||||||||||||||||||||
| 23. |
During, DNA replication, DNA
|
|||||||||||||||||||||
| 24. |
Which is not true about proteins?
|
|||||||||||||||||||||
| 25. |
Molecules of DNA are composed of long chains of
|
|||||||||||||||||||||
| 26. |
Watson and Crick were the first scientists to state that DNA
|
|||||||||||||||||||||
| 27. |
The two chains of a DNA molecule are connected by
|
|||||||||||||||||||||
| 28. |
All nucleotide molecules contain the same kind of
|
|||||||||||||||||||||
| 29. |
After DNA replication, the two DNA molecules that are made
|
|||||||||||||||||||||
| 30. |
Sixty-four codons for 20 amino acids requires that
|
|||||||||||||||||||||
| 31. |
Which of the following combines with amino acids
|
|||||||||||||||||||||
| 32. |
rRNA has a function in
|
|||||||||||||||||||||
| 33. |
The DNA code consists of sequences of nucleotides arranged in groups of
|
|||||||||||||||||||||
| 34. |
Unlike mRNA, the DNA molecule is
|
|||||||||||||||||||||
| 35. |
The number of bases in a row in a gene that codes a protein composed of 200 amino acids is
|
|||||||||||||||||||||
| 36. |
A DNA molecule unzips during
|
|||||||||||||||||||||
| 37. |
A DNA chain has the following sequence of bases, TAG. The corresponding messenger RNA
chain should have the sequence
|
|||||||||||||||||||||
| 38. |
Unlike DNA, RNA
|
|||||||||||||||||||||
| 39. |
Which molecule contains deoxyribose
|
|||||||||||||||||||||
| 40. |
Each combination of three nitrogenous bases on messenger RNA forms a (an)
|
|||||||||||||||||||||
| 41. |
In RNA, uracil is complementary to:
|
|||||||||||||||||||||
| 42. |
Once a molecule of transfer RNA has released its amino acid, the tRNA
|
|||||||||||||||||||||
| 43. |
If the sequence of bases in a segment of a DNA strand were cytosine, guanine, adenine, thymine, adenine, then the sequence in a complimentary strand of newly-made mRNA would be
|
|||||||||||||||||||||
| 44. |
Which sugar is present in RNA
|
|||||||||||||||||||||
| 45. |
RNA differs from DNA, in that RNA
|
|||||||||||||||||||||
| |
|
Chapter 11 Cell Communication Lecture Outline
Overview
· Cell-to-cell communication is absolutely essential for multicellular organisms.
° Cells must communicate to coordinate their activities.
· Communication between cells is also important for many unicellular organisms.
· Biologists have discovered universal mechanisms of cellular regulation involving the same small set of cell-signaling mechanisms.
° The ubiquity of these mechanisms provides additional evidence for the evolutionary relatedness of all life.
· Cells most often communicate by chemical signals, although signals may take other forms.
A. An Overview of Cell Signaling
· What messages are passed from cell to cell? How do cells respond to these messages?
· We will first consider communication in microbes, to gain insight into the evolution of cell signaling.
1. Cell signaling evolved early in the history of life.
· One topic of cell “conversation” is sex.
· Saccharomyces cerevisiae, the yeast of bread, wine, and beer, identifies potential mates by chemical signaling.
° There are two sexes, a and a, each of which secretes a specific signaling molecule, a factor and a factor, respectively.
° These factors each bind to receptor proteins on the other mating type.
· Once the mating factors have bound to the receptors, the two cells grow toward each other and undergo other cellular changes.
· The two cells fuse, or mate, to form an a/a cell containing the genes of both cells.
· The process by which a signal on a cell’s surface is converted into a specific cellular response is a series of steps called a signal-transduction pathway.
° The molecular details of these pathways are strikingly similar in yeast and animal cells, even though their last common ancestor lived more than a billion years ago.
° Signaling systems of bacteria and plants also share similarities.
· These similarities suggest that ancestral signaling molecules evolved long ago in prokaryotes and have since been adopted for new uses by single-celled eukaryotes and multicellular descendents.
2. Communicating cells may be close together or far apart.
· Multicellular organisms release signaling molecules that target other cells.
· Cells may communicate by direct contact.
° Both animals and plants have cell junctions that connect to the cytoplasm of adjacent cells.
° Signaling substances dissolved in the cytosol can pass freely between adjacent cells.
° Animal cells can communicate by direct contact between membrane-bound cell surface molecules.
° Such cell-cell recognition is important to such processes as embryonic development and the immune response.
· In other cases, messenger molecules are secreted by the signaling cell.
° Some transmitting cells release local regulators that influence cells in the local vicinity.
° One class of local regulators in animals, growth factors, includes compounds that stimulate nearby target cells to grow and multiply.
° This is an example of paracrine signaling, which occurs when numerous cells simultaneously receive and respond to growth factors produced by a single cell in their vicinity.
· In synaptic signaling, a nerve cell produces a neurotransmitter that diffuses across a synapse to a single cell that is almost touching the sender.
° The neurotransmitter stimulates the target cell.
° The transmission of a signal through the nervous system can also be considered an example of long-distance signaling.
· Local signaling in plants is not well understood. Because of their cell walls, plants must have different mechanisms from animals.
· Plants and animals use hormones for long-distance signaling.
° In animals, specialized endocrine cells release hormones into the circulatory system, by which they travel to target cells in other parts of the body.
° Plant hormones, called growth regulators, may travel in vessels but more often travel from cell to cell or move through air by diffusion.
· Hormones and local regulators range widely in size and type.
° The plant hormone ethylene (C2H4), which promotes fruit ripening and regulates growth, is a hydrocarbon of only six atoms, capable of passing through cell walls.
° Insulin, which regulates blood sugar levels in mammals, is a protein with thousands of atoms.
· What happens when a cell encounters a signal?
° The signal must be recognized by a specific receptor molecule, and the information it carries must be changed into another form, or transduced, inside the cell before the cell can respond.
3. The three stages of cell signaling are reception, transduction, and response.
· E. W. Sutherland and his colleagues pioneered our understanding of cell signaling.
° Their work investigated how the animal hormone epinephrine stimulates breakdown of the storage polysaccharide glycogen in liver and skeletal muscle.
° Breakdown of glycogen releases glucose derivatives that can be used for fuel in glycolysis or released as glucose in the blood for fuel elsewhere.
° Thus one effect of epinephrine, which is released from the adrenal gland during times of physical or mental stress, is mobilization of fuel reserves.
· Sutherland’s research team discovered that epinephrine activated a cytosolic enzyme, glycogen phosphorylase.
° However, epinephrine did not activate the phosphorylase directly in vitro but could only act via intact cells.
° Therefore, there must be an intermediate step or steps occurring inside the cell.
° The plasma membrane must be involved in transmitting the epinephrine signal.
· The process involves three stages: reception, transduction, and response.
° In reception, a chemical signal binds to a cellular protein, typically at the cell’s surface or inside the cell.
° In transduction, binding leads to a change in the receptor that triggers a series of changes in a series of different molecules along a signal-transduction pathway. The molecules in the pathway are called relay molecules.
° In response, the transduced signal triggers a specific cellular activity.
B. Signal Reception and the Initiation of Transduction
1. A signal molecule binds to a receptor protein, causing the protein to change shape.
· The cell targeted by a particular chemical signal has a receptor protein on or in the target cell that recognizes the signal molecule.
° Recognition occurs when the signal binds to a specific site on the receptor that is complementary in shape to the signal.
· The signal molecule behaves as a ligand, a small molecule that binds with specificity to a larger molecule.
· Ligand binding causes the receptor protein to undergo a change in shape.
· This may activate the receptor so that it can interact with other molecules.
° For other receptors, this causes aggregation of receptor molecules, leading to further molecular events inside the cell.
· Most signal receptors are plasma membrane proteins, whose ligands are large water-soluble molecules that are too large to cross the plasma membrane.
2. Some receptor proteins are intracellular.
· Some signal receptors are dissolved in the cytosol or nucleus of target cells.
° To reach these receptors, the signals pass through the target cell’s plasma membrane.
° Such chemical messengers are either hydrophobic enough or small enough to cross the phospholipid interior of the plasma membrane.
· Hydrophobic messengers include the steroid and thyroid hormones of animals.
· Nitric oxide (NO) is a gas whose small size allows it to pass between membrane phospholipids.
· Testosterone is secreted by the testis and travels through the blood to enter cells throughout the body.
° The cytosol of target cells contains receptor molecules that bind testosterone, activating the receptor.
° These activated proteins enter the nucleus and turn on specific genes that control male sex characteristics.
· How does the activated hormone-receptor complex turn on genes?
· These activated proteins act as transcription factors.
· Transcription factors control which genes are turned on—that is, which genes are transcribed into messenger RNA.
· mRNA molecules leave the nucleus and carry information that directs the synthesis (translation) of specific proteins at the ribosome.
· Other intracellular receptors (such as thyroid hormone receptors) are found in the nucleus and bind to the signal molecules there.
3. Most signal receptors are plasma membrane proteins.
· Most signal molecules are water-soluble and too large to pass through the plasma membrane.
· They influence cell activities by binding to receptor proteins on the plasma membrane.
° Binding leads to changes in the shape of the receptor or to the aggregation of receptors.
° These cause changes in the intracellular environment.
· There are three major types of membrane receptors: G-protein-linked receptors, receptor tyrosine kinases, and ion-channel receptors.
· A G-protein-linked receptor consists of a receptor protein associated with a G protein on the cytoplasmic side.
° Seven alpha helices span the membrane.
° G-protein-linked receptors bind many different signal molecules, including yeast mating factors, epinephrine and many other hormones, and neurotransmitters.
· The G protein acts as an on/off switch.
° If GDP is bound to the G protein, the G protein is inactive.
° When the appropriate signal molecule binds to the extracellular side of the receptor, the G protein binds GTP (instead of GDP) and becomes active.
° The activated G protein dissociates from the receptor and diffuses along the membrane, where it binds to an enzyme, altering its activity.
° The activated enzyme triggers the next step in a pathway leading to a cellular response.
· The G protein can also act as a GTPase enzyme to hydrolyze GTP to GDP.
° This change turns the G protein off.
· Now inactive, the G protein leaves the enzyme, which returns to its original state.
· The whole system can be shut down quickly when the extracellular signal molecule is no longer present.
· G-protein receptor systems are extremely widespread and diverse in their functions.
° They play important roles during embryonic development.
° Vision and smell in humans depend on these proteins.
· Similarities among G proteins and G-protein-linked receptors of modern organisms suggest that this signaling system evolved very early.
· Several human diseases involve G-protein systems.
° Bacterial infections causing cholera and botulism interfere with G-protein function.
· The tyrosine-kinase receptor system is especially effective when the cell needs to trigger several signal transduction pathways and cellular responses at once.
° This system helps the cell regulate and coordinate many aspects of cell growth and reproduction.
· The tyrosine-kinase receptor belongs to a major class of plasma membrane receptors that have enzymatic activity.
° A kinase is an enzyme that catalyzes the transfer of phosphate groups.
° The cytoplasmic side of these receptors functions as a tyrosine kinase, transferring a phosphate group from ATP to tyrosine on a substrate protein.
· An individual tyrosine-kinase receptor consists of several parts:
° An extracellular signal-binding site.
° A single alpha helix spanning the membrane.
° An intracellular tail with several tyrosines.
· The signal molecule binds to an individual receptor.
° Ligands bind to two receptors, causing the two receptors to aggregate and form a dimer.
· This dimerization activates the tyrosine-kinase section of the receptors, each of which then adds phosphate from ATP to the tyrosine tail of the other polypeptide.
· The fully activated receptor proteins activate a variety of specific relay proteins that bind to specific phosphorylated tyrosine molecules.
° One tyrosine-kinase receptor dimer may activate ten or more different intracellular proteins simultaneously.
° These activated relay proteins trigger many different transduction pathways and responses.
· A ligand-gated ion channel is a type of membrane receptor that can act as a gate when the receptor changes shape.
· When a signal molecule binds as a ligand to the receptor protein, the gate opens to allow the flow of specific ions, such as Na+ or Ca2+, through a channel in the receptor.
° Binding by a ligand to the extracellular side changes the protein’s shape and opens the channel.
° When the ligand dissociates from the receptor protein, the channel closes.
· The change in ion concentration within the cell may directly affect the activity of the cell.
· Ligand-gated ion channels are very important in the nervous system.
° For example, neurotransmitter molecules released at a synapse between two neurons bind as ligands to ion channels on the receiving cell, causing the channels to open.
° Ions flow in and trigger an electrical signal that propagates down the length of the receiving cell.
· Some gated ion channels respond to electrical signals, instead of ligands.
C. Signal-Transduction Pathways
· The transduction stage of signaling is usually a multistep pathway.
· These pathways often greatly amplify the signal.
° If some molecules in a pathway transmit a signal to multiple molecules of the next component in the series, the result can be large numbers of activated molecules at the end of the pathway.
· A small number of signal molecules can produce a large cellular response.
· Also, multistep pathways provide more opportunities for coordination and regulation than do simpler systems.
1. Pathways relay signals from receptors to cellular responses.
· Signal-transduction pathways act like falling dominoes.
° The signal-activated receptor activates another protein, which activates another, and so on, until the protein that produces the final cellular response is activated.
· The relay molecules that relay a signal from receptor to response are mostly proteins.
° The interaction of proteins is a major theme of cell signaling.
° Protein interaction is a unifying theme of all cellular regulation.
· The original signal molecule is not passed along the pathway and may not even enter the cell.
° It passes on information.
° At each step, the signal is transduced into a different form, often by a conformational change in a protein.
° The conformational change is often brought about by phosphorylation.
2. Protein phosphorylation, a common mode of regulation in cells, is a major mechanism of signal transduction.
· The phosphorylation of proteins by a specific enzyme (a protein kinase) is a widespread cellular mechanism for regulating protein activity.
° Most protein kinases act on other substrate proteins, unlike tyrosine kinases that act on themselves.
· Most phosphorylation occurs at either serine or threonine amino acids of the substrate protein (unlike tyrosine phosphorylation in tyrosine kinases).
· Many of the relay molecules in a signal-transduction pathway are protein kinases that act on other protein kinases to create a “phosphorylation cascade.”
· Each protein phosphorylation leads to a conformational change because of the interaction between the newly added phosphate group and charged or polar amino acids on the protein.
· Phosphorylation of a protein typically converts it from an inactive form to an active form.
° Rarely, phosphorylation inactivates protein activity.
· A single cell may have hundreds of different protein kinases, each specific for a different substrate protein.
° Fully 2% of our genes are thought to code for protein kinases.
° Together, they regulate a large proportion of the thousands of cell proteins.
· Abnormal activity of protein kinases can cause abnormal cell growth and may contribute to the development of cancer.
· The responsibility for turning off a signal-transduction pathway belongs to protein phosphatases.
° These enzymes rapidly remove phosphate groups from proteins, a process called dephosphorylation.
° Phosphatases also make the protein kinases available for reuse, enabling the cell to respond again to a signal.
· At any given moment, the activity of a protein regulated by phosphorylation depends on the balance of active kinase molecules and active phosphatase molecules.
· When the extracellular signal molecule is absent, active phosphatase molecules predominate, and the signaling pathway and cellular response are shut down.
· The phosphorylation/dephosphorylation system acts as a molecular switch in the cell, turning activities on and off as required.
3. Certain signal molecules and ions are key components of signaling pathways (second messengers).
· Many signaling pathways involve small, water-soluble, nonprotein molecules or ions called second messengers.
° These molecules rapidly diffuse throughout the cell.
· Second messengers participate in pathways initiated by both G-protein-linked receptors and tyrosine-kinase receptors.
° Two of the most widely used second messengers are cyclic AMP and Ca2+.
· Once Sutherland knew that epinephrine caused glycogen breakdown without entering the cell, he looked for a second messenger inside the cell.
· Binding by epinephrine leads to increases in the cytosolic concentration of cyclic AMP, or cAMP.
° This occurs because the activated receptor activates adenylyl cyclase, which converts ATP to cAMP.
° The normal cellular concentration of cAMP can be boosted twentyfold within seconds.
° cAMP is short-lived, as phosphodiesterase converts it to AMP.
° Another surge of epinephrine is needed to reboost the cytosolic concentration of cAMP.
· Caffeine-containing beverages such as coffee provide an artificial way to keep the body alert.
° Caffeine blocks the conversion of cAMP to AMP, maintaining the system in a state of activation in the absence of epinephrine.
· Many hormones and other signal molecules trigger the formation of cAMP.
° G-protein-linked receptors, G proteins, and protein kinases are other components of cAMP pathways.
° cAMP diffuses through the cell and activates a serine/threonine kinase called protein kinase A.
° The activated kinase phosphorylates various other proteins.
· Regulation of cell metabolism is also provided by G-protein systems that inhibit adenylyl cyclase.
° These use a different signal molecule to activate a different receptor that activates an inhibitory G protein.
· Certain microbes cause disease by disrupting G-protein signaling pathways.
° The cholera bacterium, Vibrio cholerae, may be present in water contaminated with human feces.
° This bacterium colonizes the small intestine and produces a toxin that modifies a G protein that regulates salt and water secretion.
° The modified G protein is unable to hydrolyze GTP to GDP and remains stuck in its active form, continuously stimulating adenylyl cyclase to make cAMP.
° The resulting high concentration of cAMP causes the intestinal cells to secrete large amounts of water and salts into the intestines, leading to profuse diarrhea and death from loss of water and salts.
· Treatments for certain human conditions involve signaling pathways.
° One pathway uses cyclic GMP, or cGMP, as a signaling molecule. Its effects include the relaxation of smooth muscle cells in artery walls.
° A compound was developed to treat chest pains. This compound inhibits the hydrolysis of cGMP to GMP, prolonging the signal and increasing blood flow to the heart muscle.
° Under the trade name Viagra, this compound is now widely used as a treatment for erectile dysfunction. Viagra causes dilation of blood vessels, allowing increased blood flow to the penis.
· Many signal molecules in animals induce responses in their target cells via signal-transduction pathways that increase the cytosolic concentration of Ca2+.
° In animal cells, increases in Ca2+ may cause contraction of muscle cells, secretion of certain substances, and cell division.
° In plant cells, increases in Ca2+ trigger responses such as the pathway for greening in response to light.
· Cells use Ca2+ as a second messenger in both G-protein pathways and tyrosine-kinase pathways.
· The Ca2+ concentration in the cytosol is typically much lower than that outside the cell, often by a factor of 10,000 or more.
° Various protein pumps transport Ca2+ outside the cell or into the endoplasmic reticulum or other organelles.
° As a result, the concentration of Ca2+ in the ER is usually much higher than the concentration in the cytosol.
· Because cytosolic Ca2+ is so low, small changes in the absolute numbers of ions causes a relatively large percentage change in Ca2+ concentration.
· Signal-transduction pathways trigger the release of Ca2+ from the cell’s ER.
· The pathways leading to release involve still other second messengers, diacylglycerol (DAG) and inositol trisphosphate (IP3).
° DAG and IP3 are created when a phospholipase cleaves membrane phospholipid PIP2.
° The phospholipase may be activated by a G protein or by a tyrosine-kinase receptor.
° IP3 activates a gated-calcium channel, releasing Ca2+ from the ER.
· Calcium ions activate the next protein in a signal-transduction pathway.
D. Cellular Responses to Signals
1. In response to a signal, a cell may regulate activities in the cytoplasm or transcription in the nucleus.
· Ultimately, a signal-transduction pathway leads to the regulation of one or more cellular activities.
° This may be the opening or closing of an ion channel or a change in cell metabolism.
° For example, epinephrine helps regulate cellular energy metabolism by activating enzymes that catalyze the breakdown of glycogen.
· The stimulation of glycogen breakdown by epinephrine involves a G-protein-linked receptor, a G protein, adenylyl cyclase, cAMP, and several protein kinases before glycogen phosphorylase is activated.
· Other signaling pathways do not regulate the activity of enzymes but the synthesis of enzymes or other proteins.
· Activated receptors may act as transcription factors that turn specific genes on or off in the nucleus.
2. Elaborate pathways amplify and specify the cell’s response to signals.
· Signaling pathways with multiple steps have two benefits.
They amplify the response to a signal.
They contribute to the specificity of the response.
· At each catalytic step in a cascade, the number of activated products is much greater than in the preceding step.
° In the epinephrine-triggered pathway, binding by a small number of epinephrine molecules can lead to the release of hundreds of millions of glucose molecules.
· Various types of cells may receive the same signal but produce very different responses.
° For example, epinephrine triggers liver or striated muscle cells to break down glycogen, but stimulates cardiac muscle cells to contract, leading to a rapid heartbeat.
· The explanation for this specificity is that different kinds of cells have different collections of proteins.
° The response of a particular cell to a signal depends on its particular collection of receptor proteins, relay proteins, and proteins needed to carry out the response.
° Two cells that respond differently to the same signal differ in one or more of the proteins that handle and respond to the signal.
· A signal may trigger a single pathway in one cell but trigger a branched pathway in another.
· Two pathways may converge to modulate a single response.
· Branching of pathways and interactions between pathways are important for regulating and coordinating a cell’s response to incoming information.
· Rather than relying on diffusion of large relay molecules such as proteins, many signal pathways are linked together physically by scaffolding proteins.
° Scaffolding proteins may themselves be relay proteins to which several other relay proteins attach.
° This hardwiring enhances the speed, accuracy, and efficiency of signal transfer between cells.
· The importance of relay proteins that serve as branch or intersection points in signaling pathways is underscored when these proteins are defective or missing.
° The inherited disorder Wiskott-Aldrich syndrome (WAS) is caused by the absence of a single relay protein.
° Symptoms include abnormal bleeding, eczema, and a predisposition to infections and leukemia, due largely to the absence of the protein in the cells of the immune system.
° The WAS protein is located just beneath the cell surface, where it interacts with the microfilaments of the cytoskeleton and with several signaling pathways, including those that regulate immune cell proliferation.
° When the WAS protein is absent, the cytoskeleton is not properly organized and signaling pathways are disrupted.
· As important as activating mechanisms are inactivation mechanisms.
° For a cell to remain alert and capable of responding to incoming signals, each molecular change in its signaling pathways must last only a short time.
° If signaling pathway components become locked into one state, whether active or inactive, the proper function of the cell can be disrupted.
° Binding of signal molecules to receptors must be reversible, allowing the receptors to return to their inactive state when the signal is released.
° Similarly, activated signals (cAMP and phosphorylated proteins) must be inactivated by appropriate enzymes to prepare the cell for a fresh signal.
Chapter 3 Biochemistry of Cells PowerPoint Notes
1. What is the most abundant organic compound on Earth?
2. Approximately how much water makes up the cells of organisms?
3. ___________ is known as the universal solvent.
4. List 4 properties of water that make it so useful to organisms.
5. Besides water, what other substance makes up most of the cell?
6. ____________ chemistry is the study of carbon compounds.
7. Carbon has _______ outer electrons so it can form ___________ bonds by sharing these electrons.
8. Carbon & hydrogen make up compounds called ________________.
9. Sketch a simple hydrocarbon with the formula CH4.
10. Carbon skeletons may be straight _______________, _____________ chains, or ______________ structures.
11. Hydrocarbons in ____________ supply our bodies with energy.
12. The _______ of an organic molecule determines its function.
13. ____________ groups give different properties to the organic compound to which they attach.
14. Write the formula for the following functional groups:
a. Hydroxyl
b. Carbonyl
c. Carboxyl
d. Amino
15. Give examples of organic compounds that contain each of the functional groups from question 14.
16. Large organic molecules are called _______________.
17. Polymers are built from smaller subunits called _____________.
18. Biologists call polymers _____________________.
19. Name 4 examples of polymers found in living things.
20. Monomers linked together are called ____________.
21. The process of linking monomers together is called _______________________.
22. Dehydration synthesis links small molecules or monomers together by removing molecules of _____________.
23. Name the process used to break down large polymers into smaller monomers.
24. Hydrolysis involves ____________ a molecule of water in order to break bonds.
25. Name some foods that contain lots of carbohydrates.
26. _________________ are simple sugars.
27. Name 3 monosaccharides & give their chemical formula.
28. Monosaccharides are called hexose sugars because they contain 6 _______________.
29. __________ is the simple sugar made by plants, ___________ is the sugar found in fruits, while _______________ is known as “milk sugar”. Sugars have an __________ ending.
30. What are isomers?
31. Name 2 isomers.
32. What does aqueous mean?
33. What happens to simple sugars, monosaccharides, when they are put into aqueous solutions inside cells?
34. ___________________ serve as fuel for cells. Saccharide means ________________.
35. What is a double sugar called?
36. How are disaccharides formed? Name the BOND that joins them together.
37. Name 3 disaccharides.
38. Name the simple sugars that make up each of these disaccharides:
a. Sucrose
b. Maltose
c. Lactose
39. Complex carbohydrates are called ________________ & are made of chains of ________________________.
40. Name 3 examples of polysaccharides and tell the shape of each.
41. Plants store carbohydrate energy as ____________.
42. Name some starchy foods.
43. Animals store their carbohydrate energy as __________________.
44. Both starch & glycogen are made of monomers of ____________ or glucose.
45. Describe cellulose fibers & tell where in plants it is found.
46. Cellulose makes up __________ in plants and serves as dietary __________ in animals.
47. How are cows able to digest cellulose?
48. Since sugars dissolve in water, they are said to be _____________ or water-loving. What functional group makes them water soluble?
49. Lipids are hydrophobic. What does this mean?
50. Name 4 examples of lipids and then give 3 functions for lipids in the body.
Examples:
a.
b.
c.
51. If the bonds between carbons in a fatty acid are all single bonds, the fatty acid is ___________________. Sketch a saturated fatty acid.
52. If there is a double bond between carbons in a fatty acid, the fatty acid is ___________________. Sketch an unsaturated fatty acid.
53. _______________ are the monomers that make up lipids or fats.
54. Triglycerides are made of an alcohol called ____________ and 3 ___________ acid chains.
55. ___________ forms the backbone of the fat. Sketch glycerol.
56. Saturated fatty acids are ___________ at room temperature and include __________,
margarine, and _____________.
57. Unsaturated fats in plants exist as ________ or oils at room temperature.
58. (a) What process links the 3 fatty acid chains to the glycerol in lipids?
(b) What lipids are in cell membranes?
(c) Sketch and label a phospholipid.
(d) Phospholipid heads are _____________ and attract water, while the 2 tails are _________ and repel water.
59. Lipids called _____________ are made of four, fused rings of carbon.
60. Name 3 steroids found in organisms.
a.
b.
c.
61. Proteins are polymers made of monomers called ___________________.
62. How many different amino acids are there?
63. Give 3 jobs for proteins in cells.
a.
b.
c.
64. What four things are bonded to the central carbon of every amino acid?
65. Sketch the structure of an amino acid & label the attached groups.
66. Amino acids are linked together by ____________ synthesis and held together by _____________ bonds.
67. Many proteins act as __________ or biological catalysts.
68. Cells have _____________ of enzymes which may ___________ chemical bonds and ____________ the amount of activation energy needed for the reaction to occur.
69. Enzymes have what shape?
70. Substrates attach to an enzyme at its ___________ site. When a substrate attaches to the active site the active site changes ________________. This is called ______________ fit.
71. Can enzymes be reused?
72. The linear sequence of amino acids (chain) is the ____________ structure of a protein.
73. Protein chains are called __________________.
74. Secondary protein structures occur when proteins ___________ or ___________.
75. When polypeptides join together, the _________ groups interact with each other forming the ___________ structure of a protein forms.
76. Proteins take on a _____________ shape in the watery environment inside a cell. This is known as their _______________________ structure. Protein shape is also known as protein _____________________.
77. Denaturing a protein involves changing its __________ so it no longer works.
78. Name 2 things that denature proteins.
79. (a) What causes sickle cell anemia (disease)?
(b) What is the function of the protein hemoglobin in red blood cells?
(c) What protein controls blood sugar level?
(d) Insulin causes excess sugar to be stored in the _____________ as ________________.
(e) Proteins in the cell membrane that help cells recognize similar cells are called __________ proteins.
80. ___________ acids store hereditary information for making all of the body’s ______________.
81. Name the 2 types of nucleic acids.
82. What are the monomers for nucleic acids? Sketch a nucleotide.
83. Name the 4 bases on DNA.
84. What 2 things make up the sides of DNA?
85. DNA is ___________ stranded & coiled to make a shape called the double ____________.
86. RNA has __________ sugar instead of DEOXYRIBOSE sugar on DNA
87. RNA is a _____________ stranded molecule unlike double stranded DNA.
88. On RNA, the base ______________ replaces thymine.
89. _____________ is the cell’s energy molecule.
90. What is the monomer for ATP?
91. What does ATP stand for?
92. How is the nucleotide monomer for ATP DIFFERENT from the nucleotide monomer for nucleic acids?
93. Where is the energy stored in ATP?
94. Which bonds are considered HIGH ENERGY bonds in ATP?
95. When the last phosphate bond is broken, what is released?
96. what is the energy of ATP used for?
97. Besides energy, what two other things are formed when the last phospheta bond of ATP is broken?
98. How can ATP be reformed?