| Birds All Materials © Cmassengale |
 |
Birds:
| Well adapted to marine, freshwater, & terrestrial habitats | |
| Bodies adapted for flight | |
| Endothermic – body temperature controlled by metabolism |
Evolution:
- Evolved from reptiles
- Few fossils due to lack of preservation of feathers or thin, hollow bones
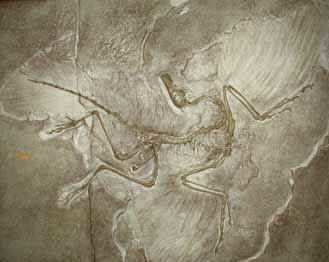
- Archaeopteryx:
1. Possible link between birds & reptiles
2. Lived during Jurassic period
3. Large skull with reptile like teeth
4. Bones not hollow
5. Claws on forelimbs
6. Long tail
7. Strong legs & rounded wings for gliding
8. Feathers
9. Furculum – fused collarbone or wishbone
- Hesperonis:
1. Bird fossils from Cretaceous period
2. Large, flightless bird
3. Had teeth like reptiles

Hesperonis
- Ichthyornis:
1. Smaller, tern like bird
2. Lived during Cretaceous period
3. Had large flight wings
![]()
Characteristics of Birds:
- Body covered with feathers made of protein called keratin
- Thin, hollow bones
- Some bones fused for extra strength
- Forelimbs modified into wings for flight
- Two hind limbs with claws to support upright body
- Scales on legs
- Toothless, horny beak
- Additional air sacs with lungs for more oxygen
- Endotherms (40 to 41 degrees Celsius body temperature)
- Four chambered heart with single, right aortic arch
- Amniote egg with calcium carbonate shell
- Oviparity with both parents often caring for eggs
- Eggs usually incubated within a nest
Feathers:
- Modified scales
- Function to provide lift for flight & help conserve body heat
- Five kinds of feathers —– down, contour, flight, filoplume, & bristles

Types of Feathers
- Down feathers:
1. Soft & fluffy
2. Cover the body of nestlings
3. Provide an undercoat insulating adult birds - Contour Feathers:
1. Give streamline shape to body
2. Provide coloration to adult birds
3. Give additional insulation to body - Flight Feathers:
1. Specialized contour feathers
2. Found on wings & tail

- Filoplumes:
1. Called pin feathers
2. Hairlike feathers under contour feathers on body
Parts of a feather:
- Develop from tiny pits in the skin called follicles
- Shaft emerges from the follicle
- Two vanes develop on either side of shaft
- Barbs branch off of each vane & have projections called barbules
- Barbules have microscopic hooks to hold barbules together
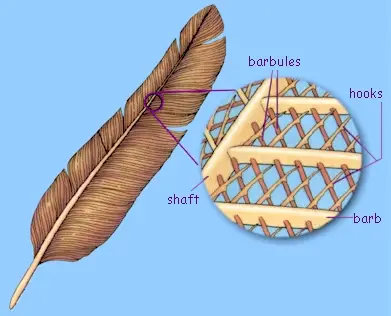
Parts of a Flight Feather

Microscopic Hooks on Barbules
- Birds preen their feathers to clean them & coat them with oil
- Preen glands – oil glands located at the base of the tail
- Birds shed or molt feathers periodically:
1. Molting usually in late summer between breeding & migration
2. Flight feathers replaced
3. Some birds molt before courtship
Beaks and Feet:
- Adapted to habitat & feeding
- Hawks & eagles have hooked beaks & talons for tearing meat
 |
||
| Talons | Hooked Beak | Penguin Flippers |
- Swifts have tiny beaks that open wide to catch insects in midair
- Flightless birds like ostriches have legs & feet modified for running & walking
- Penguins have wings modified into flippers for swimming
- Ducks & geese with webbed feet
 |
 |
| Running Legs of Ostrich | Webbed Feet on Duck |
- Legs of some birds such as herons & egrets turn vivid colors to attract mates; caused by hormones
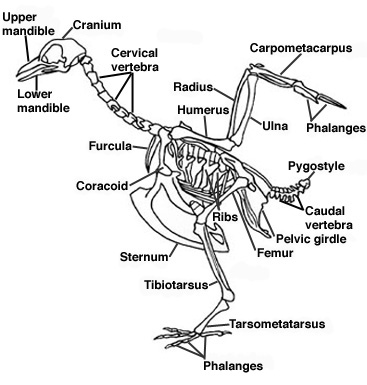
Skeleton and Muscles:
- Pelvic & pectoral girdles fused for strength
- Bones thin & hollow so bird lighter
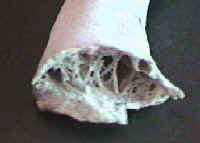
Hollow Bones
- Furculum or wishbone is a fused collarbone that stabilizes bird in flight
- Lighter beak replaces heavy teeth & jaws
- Lower vertebrae fused so no heavy ligaments needed
- Enlarged eye sockets reduce skull weight
- Keeled sternum for attachment of large flight muscles
- Pygostyle – terminal vertebrae support tail & aids in flight (lift, steering, & braking)
- Two digits in forelimbs lost & other three digits fused to form wings
- Wings shaped like air foils (thicker in front & tapering to back) so air moves faster on top causing lift
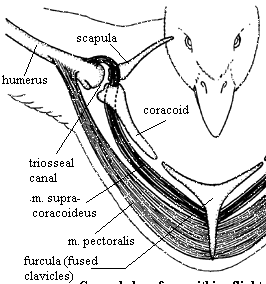
- Powerful muscles make up 50% of body weight
- each wing movement uses different set of muscles
- Flight muscles called pectorals & are attached to wing & keeled sternum
- When large pectorals contract, wings move down
- When large pectorals relax & small pectorals contract, wings move upward
Body Temperature:
- Metabolism generates body heat (endothermic)
- Enables birds to survive in warm & cold environments
- Rapid breathing & increased air sacs in lungs bring in more oxygen
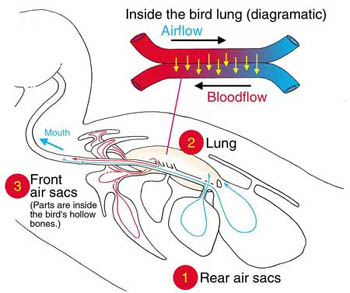
Air Sacs in Bird Lungs
- Ingest large amounts of food for energy
- Fluff out feathers to trap air for insulation
- Aquatic birds have thin layer of fat for insulation
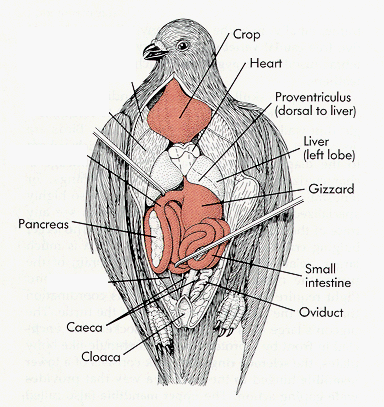
Digestive System:
- Fast & efficient digestion (mouse digested in 3 hours)
- No chewing
- Crop for temporary food storage
- Two part stomach — proventriculus & gizzard
- Proventriculus is 1st chamber where digestive juices added
- Gizzard is 2nd part for crushing food
- Small stones & gravel eaten by birds aids grinding in gizzard
- Pyloric sphincter valve at lower end of gizzard controls food movement into intestines
- Duodenum – beginning of small intestine where bile (digests fats) & pancreatic juice are added & digested food is absorbed
Excretory System:
- Paired kidneys filter nitrogen wastes (uric acid) from blood
- No urinary bladder to store liquid wastes
- Uric acid travels down ureters to cloaca where intestinal wastes & reproductive products added
- Uric acid secreted in white, semi solid mass
- Shorebirds have salt secreting glands above the eyes & secrete excess salt through their nostrils
Respiratory System:
- Fly at high altitudes where there is less oxygen so need efficient respiratory system
- High metabolic rate requires large amount of oxygen
- Nine air sacs associated with lungs increase oxygen level & decrease density
- Air sacs connected to air spaces in hollow bones
- One way flow of air in lungs & air sacs so more oxygen is removed
- Air pathway:
air enters body through nostrils on beaktrachea (windpipe)
syrinx (voice box)
2 primary bronchi
75% of air into two posterior air sacs and 25% of air into lungs
air from lungs into other seven air sacs
- When carbon dioxide exhaled, oxygen from posterior air sacs moves into lungs to always keep fresh oxygen supply
Circulatory System:
- Four chambered heart
- Right side of heart pumps deoxygenated blood from body cells to lungs
- Left side of heart receives oxygenated blood from lungs & pumps it to the body cells
- Single aortic arch
- Rapid heartbeat (hummingbird 600X/minute & chickadee 1000X/minute)
- Less active birds such as ostrich have slower heart rates (70X/minute)
Nervous System:
- Large brains relative to size of bird
- Most highly developed brain areas control flight
- Cerebellum coordinates movement
- Cerebrum controls navigation, mating, nest building, & care of young
- Optic lobes receive & interpret visual stimuli
- Keen vision
- Have color vision for locating food
- Large eyes located on side of head for wide field of vision in most birds
- Some birds such as owls with eyes on front of head for binocular vision (depth perception)
- No external ears, but have feathers around ear openings to direct sounds into ear canals
- Tympanic membrane or eardrum for picking up sound vibrations
- Semicircular canals in inner ear regulate balance
- Poorly developed sense of smell except in ducks & flightless birds
- Sense of taste helps avoid bitter tasting or toxic foods
Reproductive System:
- Testes in males produces sperm that travels by the vas deferens to cloaca
- Females have single ovary that makes eggs
- Eggs are fertilized in the oviducts
- Shell added by shell gland & then egg moves into
- In mating, male presses cloaca to female to transfer sperm (internal fertilization)
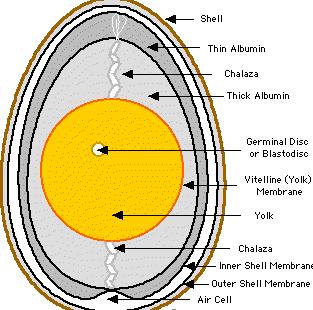
- Lay an amniote egg:
1. Embryo suspended in fluid called albumen (white of egg)
2. Chalaza – rope like strands suspending embryo in albumen
3. Chorion is membrane inside of shell
4. Yolk is stored food surrounded by yolk sac
Incubation & development of Egg:
- Eggs incubated by one or both parents
- Brood patch – thickened, featherless patch of skin on abdomen of bird used to warm eggs
- Membranes grow out of embryo’s digestive tract & surround yolk
- Membranes make digestive enzymes to dissolve proteins & lipids in yolk
- Yolk sac has blood vessels to carry food to embryo
- Wastes from embryo collect in membrane called allantois
- Chorion membrane lines the shell & allows gas exchange
- Young birds may be precocial or altricial
- Precocial young:
1. Have longer incubations
2. More eggs laid
3. Active as soon as hatch
4. Nestlings can swim, walk, & feed themselves
5. Need some parental care
6. Includes ducks, geese, & swans - Altricial young:
1.Lay fewer eggs
2. Hatch quickly
3. Hatchlings are blind, naked, & helpless
4. Depend on parents for warmth & food for several weeks
5. Includes songbirds, woodpeckers, hawks, pigeons, doves, raptors
 |
 |
| Altricial Young | Precocial Young |
Behavior:
- Longer parental care allows more complex learning (courtship, nesting, migration, etc.)
- Territoriality allows males to establish & defend breeding areas
- Courtship behaviors are used by males to attract mates:
1. Brightly colored feathers
2. Flight displays
3. Songs

Male Scarlet Tanager Breeding Plumage
- Nest building holds eggs, conceals & shelters young birds, may help attract mates
- Nests are built in sheltered, well-hidden spots in trees, on the ground, etc. & are made of twigs, mud, grass, feathers…

- Migration to new areas is triggered by dropping temperatures & dwindling food supplies
- Birds use migration clues including:
1. Position of sun & stars
2. Topographical landmarks
3. Magnetic clues
4.Air pressure changes
5. Low frequency sounds
| Section 2 Review |
Classification:
- Class Aves
- 27 orders
- Gaviiformes – loons
- Pelecaniformes – pelicans & cormorants
- Ciconiiformes – wading birds like ibises & herons
- Anseriformes – ducks, geese, & swans
- Falconiformes – falcons, eagles, hawks, vultures
- Galliformes – turkey, quail, pheasants
- Gruiformes – cranes, coots, & rails
- Charadriiformes – snipes, sandpipers, gulls, terns
- Columbiformes – pigeons & doves
- Psittaciformes – parrots, parakeets, & macaws
- Cucluiformes – cuckoos & roadrunners
- Strigiformes – owls
- Caprimulgiformes – whippoorwill & nighthawk
- Apodiformes – hummingbird & swifts
- Coraciiformes – kingfishers
- Piciformes – woodpeckers, sapsuckers, & flickers
- Passeriformes – perching birds like robins, cardinals, blue jays
 |
 |
| Pygmy Owl | Brown Pelican |
 |
 |
| Macaw | Female Cardinal |
Food & Habitat Adaptations:
- Anseriformes (ducks, geese, & swans) have webbed feet for swimming & flattened bills; young are precocial but need some parental care
- Strigiformes (owls) have sharp, hooked beaks & talons (claws) for meat eating, keen hearing & eyesight, & forward facing eyes
- Apodiformes (hummingbirds) are small, fast-flying birds with tiny feet & long tongues for drinking nectar; found only in western hemisphere
- Psittaciformes (parrots, cockatoos, parakeets…) have a strong, hooked beak for seed opening & two forward & two rear facing toes for perching & climbing
- Piciformes (woodpeckers, toucans, & flickers) have two rear facing toes for dwelling in tree cavities & sharp, chisel like bills for drilling into trees
- Falconiformes or raptors ( hawks, eagles, vultures) have hooked beaks & talons & keen vision for seeing prey
- Passeriformes or songbirds (blue jays, cardinals, sparrows, robins …) have enlarged rear facing toe to grip branches, a syrinx or voice box in males to produce songs, & a variety of beak shapes to feed on seeds, nectar, fruits, & insects; known as passerines or perching birds
- Columbiformes (pigeons & doves) have small heads & bills, a crop that makes “pigeon’s milk” for feeding young, short incubation period (2 weeks)
- Ciconiiformes (herons, ibises, & egrets) have long legs for wading & sharp pointed bills for piercing frogs & fish
- Galliformes (turkeys, quail, pheasants, & chickens) have plump bodies with limited flying &a large gizzard for grinding grains
- Sphenisciformes (penguins) have wings modified into flippers, an extra layer of body fat for insulation, & webbed feet for swimming
- Struthioniformes (ostrich) are the largest birds that can’t fly but have long legs with only two toes adapted for fast running
| Section 3 Review |

 Birds & Mammals Study Guide
Birds & Mammals Study Guide