Nucleic Acids and Protein Synthesis
All Materials © Cmassengale

Cell à Nucleus à Chromosomes à Genes à DNA
Proteins
- Organic molecules (macromolecules) made by cells
- Make up a large part of your body
- Used for growth, repair, enzymes, etc.
- Composed of long chains of small units called amino acids bonded together by peptide bonds
- Twenty amino acids exist
DNA
- Deoxyribonucleic acid is a coiled double helix carrying hereditary information of the cell

- Contains the instructions for making proteins from 20 different amino acids
- Appears as chromatin when cell not dividing

- Structure discovered by Watson & Crick in 1953
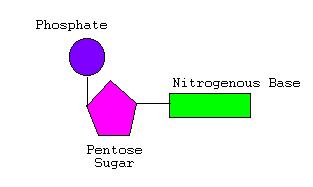
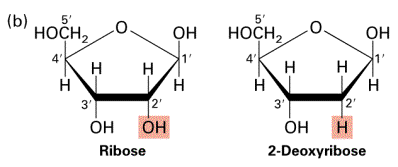
- Sides made of pentose (5-sided) sugars attached to phosphate groups by phosphodiester bonds
- Pentose sugar called Deoxyribose
- Steps or rungs of DNA made of 4 nitrogen-containing bases held together by weak hydrogen bonds
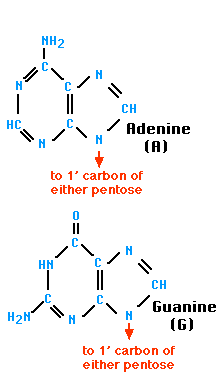
- Purines (double carbon-nitrogen rings) include adenine (A) and guanine (G)
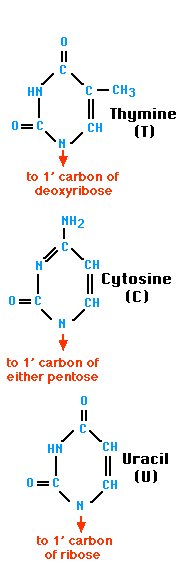
- Pyrimidines (single carbon-nitrogen rings) include thymine (T) and cytosine (C)
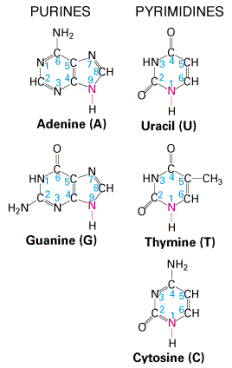
- Base pairing means a purine bonds to a pyrimidine (Example: A — T and C — G)
- Coiled, double stranded molecule known as double helix
- Make up chromosomes in the nucleus
- Subunits of DNA called nucleotides
- Nucleotides contain a phosphate, a Deoxyribose sugar, and one nitrogen base (A,T,C, or G)

- Free nucleotides also exist in nucleus
- Most DNA is coiled or twisted to the right
- Left twisted DNA is called southpaw or Z-DNA
- Hot spots which can result in mutations occur where right & left twisted DNA meet

History of DNA discovery
- Freidrich Miescher (1868) found nuclear material to be ½ protein & ½ unknown substance
- 1890’s, unknown nuclear substance named DNA
- Walter Sutton (1902) discovered DNA in chromosomes
- Fredrick Griffith (1928) working with Streptococcus pneumoniae conducted transformation experiments of virulent & nonvirulent bacterial strains
- Levene (1920’s) determined 3 parts of a nucleotide
- Hershey & Chase (1952) used bacteriophages (viruses) to show that DNA, not protein, was the cell’s hereditary material
- Rosalind Franklin (early 1950’s) used x-rays to photograph DNA crystals
 |
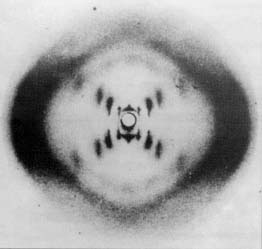 |
- Erwin Chargraff (1950’s) determined that the amount of A=T and amount of C=G in DNA; called Chargaff’s Rule
- Watson & Crick discovered double helix shape of DNA & built the 1st model

DNA Replication
- Process by which DNA makes a copy of itself
- Occurs during S phase of interphase before cell division
- Extremely rapid and accurate (only 1 in a billion are incorrectly paired)
- Requires many enzymes & ATP (energy)
- Begins at special sites along DNA called origins of replication where 2 strands open & separate making a replication fork

- Nucleotides added & new strand forms at replication forks
- DNA helicase (enzyme) uncoils & breaks the weak hydrogen bonds between complementary bases (strands separate)
- DNA polymerase adds new nucleotides to the exposed bases in the 5’ to 3’ direction

- Leading strand (built toward replication fork) completed in one piece
- Lagging strand (built moving away from the replication fork) is made in sections called Okazaki fragments

OKAZAKI FRAGMENTS
- DNA ligase helps join Okazaki segments together
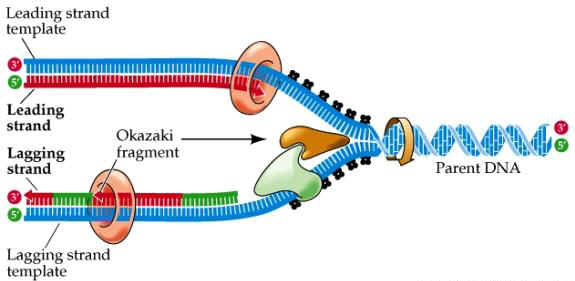
- DNA polymerase proofreads the new DNA checking for errors & repairing them; called excision repair
- Helicase recoils the two, new identical DNA molecules

RNA
- Ribonucleic acid
- Single stranded molecule

- Found in nucleus & cytoplasm
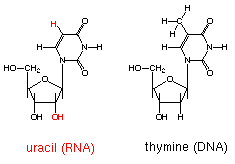
- Contains ribose sugar
- Contains the nitrogen base uracil (U) instead of thymine so A pairs with U
- Base pairings are A-U and C-G
- Three types of RNA exist (mRNA, TRNA, & rRNA)
mRNA
- Messenger RNA
- Single, uncoiled, straight strand of nucleic acid
- Found in the nucleus & cytoplasm
- Copies DNA’s instructions & carries them to the ribosomes where proteins can be made
- mRNA’s base sequence is translated into the amino acid sequence of a protein
- Three consecutive bases on mRNA called a codon (e.g. UAA, CGC, AGU)
- Reusable
tRNA
- Transfer RNA
- Single stranded molecule containing 80 nucleotides in the shape of a cloverleaf
- Carries amino acids in the cytoplasm to ribosomes for protein assembly
- Three bases on tRNA that are complementary to a codon on mRNA are called anticodons (e.g. codon- UUA; anticodon- AAU)
- Amino Acid attachment site across from anticodon site on tRNA
- Enters a ribosome & reads mRNA codons and links together correct sequence of amino acids to make a protein
- Reusable

rRNA
- Ribosomal RNA
- Globular shape
- Helps make up the structure of the ribosomes
- rRNA & protein make up the large & small subunits of ribosomes
- Ribosomes are the site of translation (making polypeptides)
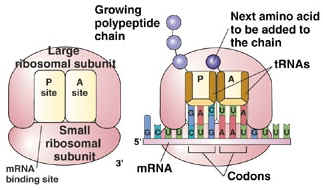
- Aids in moving ribosomes along the mRNA strand as amino acids are linked together to make a protein
Amino Acids
- 20 exist
- Linked together in a process called protein synthesis in the cytoplasm to make polypeptides (subunits of proteins)
- DNA contains the instructions for making proteins but is too large to leave the nucleus
- Three consecutive bases on DNA called a triplet (e.g. TCG, ATG, ATT)
- mRNA codon table tells what 3 bases on mRNA code for each amino acid (64 combinations of 3 bases)
- Methionine (AUG) on mRNA is called the start codon because it triggers the linking of amino acids
- UAA, UGA, & UAG on mRNA signal ribosomes to stop linking amino acids together
Genetic Code (RNA)
| Amino Acid | 3 Letter Abbreviation |
Codons |
| Alanine | Ala | GCA GCC GCG GCU |
| Arginine | Arg | AGA AGG CGA CGC CGG CGU |
| Aspartic Acid | Asp | GAC GAU |
| Asparagine | Asn | AAC AAU |
| Cysteine | Cys | UGC UGU |
| Glutamic Acid | Glu | GAA GAG |
| Glutamine | Gln | CAA CAG |
| Glycine | Gly | GGA GGC GGG GGU |
| Histidine | His | CAC CAU |
| Isoleucine | Ile | AUA AUC AUU |
| Leucine | Leu | UUA UUG CUA CUC CUG CUU |
| Lysine | Lys | AAA AAG |
| Methionine | Met | AUG |
| Phenylalanine | Phe | UUC UUU |
| Proline | Pro | CCA CCC CCG CCU |
| Serine | Ser | AGC AGU UCA UCC UCG UCU |
| Threonine | Thr | ACA ACC ACG ACU |
| Tryptophan | Trp | UGG |
| Tyrosine | Tyr | UAC UAU |
| Valine | Val | GUA GUC GUG GUU |
| Start | AUG | |
| Stop | UAA UAG UGA |
Practice Table:
| DNA Codon |
mRNA Codon |
tRNA Anticodon |
Amino Acid |
GCU |
|||
| TAC | |||
| AUU | |||
| UUU | |||
| TCA | |||
| UCU | |||
| CTT | |||
| ACU | |||
| ACU |
Protein Synthesis
- Consists of 2 parts — Transcription & Translation
- Begins in the nucleus with mRNA copying DNA’s instructions for proteins (transcription)
- Completed in the cytoplasm when tRNA enters ribosomes to read mRNA codons and link together amino acids (translation)
Steps in Transcription
- DNA helicase (enzyme) uncoils the DNA molecule
- RNA polymerase (enzyme) binds to a region of DNA called the promoter which has the start codon TAC to code for the amino acid methionine
- Promoters mark the beginning of a DNA chain in prokaryotes, but mark the beginning of 1 to several related genes in eukaryotes
- The 2 DNA strands separate, but only one will serve as the template & be copied
- Free nucleotides are joined to the template by RNA polymerase in the 5’ to 3’ direction to form the mRNA strand
- mRNA sequence is built until the enzyme reaches an area on DNA called the termination signal
- RNA polymerase breaks loose from DNA and the newly made mRNA
- Eukaryotic mRNA is modified (unneeded sections snipped out by enzymes & rejoined) before leaving the nucleus through nuclear pores, but prokaryotic RNA isn’t
- All 3 types of RNA called transcripts are produced by this method
Steps in Translation
- mRNA brings the copied DNA code from the nucleus to the cytoplasm
- mRNA attaches to one end of a ribosome; called initiation
- tRNA’s attach the correct amino acid floating in the cytoplasm to themselves
- tRNA with its attached amino acid have 2 binding sites where they join the ribosome
- The tRNA anticodon “reads” & temporarily attaches to the mRNA codon in the ribosome
- Two amino acids at a time are linked together by peptide bonds to make polypeptide -chains (protein subunits); called elongation
- Ribosomes) move along the mRNA strand until they reach a stop codon (UAA, UGA, or UAG); called termination
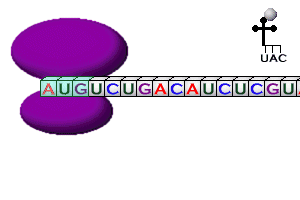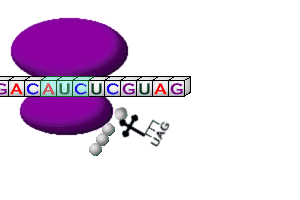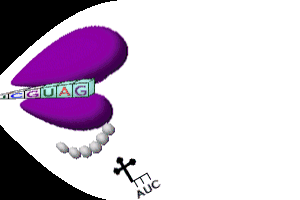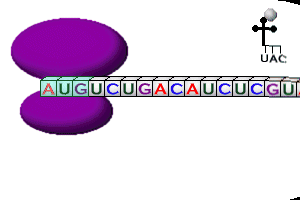
- tRNA’s break loose from amino acid, leave the ribosome, & return to cytoplasm to pick up another amino acid