DNA & Replication
ppt Questions
History of DNA
1. Early scientists believed that _________ was the genetic material of the cell. Explain why.
2. Proteins are made of 20 different ________________.
3. Long chains of amino acids make up _________________.
4. Fred Griffith worked with what type of bacteria?
5. What did he find to be true after his experiments with the S and R strains of bacteria?
6. This process of picking up DNA from the environment is called ____________________.
7. Did Griffith’s experiment prove DNA was the genetic material?
8. What 2 main things make up chromosomes?
9. What did Hershey and Chase use in their experiments to prove DNA was the cell’s genetic material?
10. Hershey and Chase radioactively tagged the viral DNA with _______________ and the protein capsid with ______________.
11. Which radioactive substance was injected into and took over the host cell’s DNA?
12. What scientists showed the amount of the 4 nitrogen bases present in DNA?
13. Name the bases and their amounts found in somatic or body cells of humans.
14. What bases are complementary (pair with each other) on DNA?
15. What type of bonds join base pairs on DNA?
16. Are these strong or weak bonds?
17. What was Rosalind Franklin’s contribution to finding DNA’s structure?
18. Who built the first model of DNA and what did they use to help get the correct measurements for the molecule?
DNA Structure
19. DNA is two coiled strands known as a ___________________.
20. What makes up the sides of a DNA molecule?
21. What bonds the deoxyribose sugars to the phosphate groups?
22. Where are the nitrogen bases found on DNA & how are they bonded?
23. Most DNA has a ______________ twist with ________ base pairs in a complete turn.
24. DNA stands for ___________________________.
25. ______________ are the subunits making up DNA.
26. Name the 3 parts of a nucleotide.
27. Why is deoxyribose called a pentose sugar?
28. Sketch a pentose sugar and include the carbons correctly numbered.
29. The sugars on one DNA strand of DNA are from 5′ to _________, while the sugars on the other strand are antiparallel running from _________ to ___________.
30. Double ring nitrogen bases are called ____________, while single ring nitrogen bases are called ________________.
31. Name the purines.
32. Name the pyrimidines.
33. Purines will only pair with ________________.
34. __________ hydrogen bonds are required to join guanine to cytosine, while only ________ bonds join adenine to thymine.
35. If there is 30% adenine, how much cytosine is present?
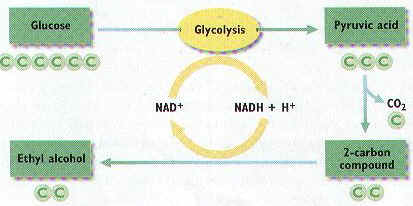
DNA Replication
36. Cells must copy their DNA before they do what? Explain why.
37. During what part of the cell cycle is DNA copied?
38. In eukaryotes, where are the copies of DNA made?
39. The process of copying DNA is known as DNA ________________.
40. Replication of DNA begins at points called __________________________.
41. The two DNA strands ______________ at origins of replication forming Y-shaped areas called _______________________.
42. New DNA strands grow at the _____________.
43. As the two strands open at the origins of replication, replication _____________ form.
44. Prokaryotic chromosomes have a ___________ replication bubble, while eukaryotic cells have ___________ bubbles.
45. What enzyme uncoils DNA so it can be replicated or copied?
46. What other job does this enzyme perform?
47. What is the job of single-strand binding proteins?
48. What enzyme relieves stress on the DNA strands at the replication forks?
49. An RNA __________ must be present to start the addition of new nucleotides.
50. What enzyme makes the RNA primer?
51. DNA ______________ is the enzyme that adds new complementary base pairs.
52. DNA polymerase can only add nucleotides to what end of the DNA molecule?
53. Which DNA strand is synthesized continuously as a single strand?
54. The leading strand is made from the ______________ toward the _________________ as it opens.
55. The ____________ strand is synthesized ____________ the overall direction of replication.
56. The lagging strand is made in __________ short segments from the _____________ fork toward the _____________ of replication.
57. The short segments of the lagging strand are called ______________ fragments.
58. What enzyme joins Okazaki fragments together into one strand?
59. Sketch half of a replication bubble. Label the point of origin and the replication fork. Draw two arrows showing the leading strand and the lagging strand.
60. DNA polymerase initially makes how many errors?
61. Proofreading enzymes correct mistakes in DNA to a one in ______________ base pairing errors?
62. Explain the semiconservative model for DNA replication.
63. Who developed this idea about replication?
64. Chemicals and _____________ radiation can damage DNA in our body cells so it must be ________________ repaired.
65. What is excision repair?
66. What 2 enzymes replace damaged sections of DNA and rebind the molecule?
67. What is the complementary DNA strand for: 5′ -CGTATG -3′ ?