| Chapter 55 Conservation Biology | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Objectives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BACK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Chapter 55 Conservation Biology | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Objectives | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
|
||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| BACK | ||||||||||||||||||||||||||||||||||||||||||||||||||||||||||||
| Bacteria |  |
1.
What is the simplest explanation for the inheritance of these colors in chickens?
Incomplete or codominance. Feather color is controlled by 2 genes B = black and b = white. The third phenotype is the result of a 50-50 mix of black and white to produce gray.
The 15 gray, 6 black, and 8 white birds represent a 2:1:1 ratio&emdash;the result of mating two heterozygous individuals: (Bb x Bb)
What offspring would you predict from the mating of a gray rooster and a black hen?
A gray rooster (Bb ) mated to a black hen (BB ) can be represented by the following Punnett square:
50% of the offspring should be gray (Bb ) and 50% black (BB )
If flower position (axial or terminal) is inherited as it is in peas what will be the ratios of genotypes and phenotypes of the generation resulting from the following cross: axial-red (true-breeding) x terminal-white?
Note: Axial (A ) is dominant over terminal (a ).
The genotypes of the parents are AARR and aarr. Therefore the gametes of the parents must be AR and ar so the genotype for all the offspring in the F1 generation will be AaRr, and their phenotype will be axial-pink.
What will be the ratios in the F2 generation?
The ratio of genotypes can be determined by examining the Punnett square below:
| 6 axial-pink | 8 pink |
| 3 axial-red | 4 red |
| 3 axial-white | 4 white |
| 2 terminal-pink | 12 axial |
| 1 terminal-white | 4 terminal |
| 1 terminal-red |
Flower position, stem length, and seed shape were three characters that Mendel studied. Each is controlled by an independently assorting gene and has dominant and recessive expression as follows:
| Character | Dominant | Recessive |
| Flower position | Axial (A ) | Terminal (a ) |
| Stem length | Tall (T ) | Dwarf (t ) |
| Seed shape | Round (R ) | Wrinkled (r) |
If a plant that is heterozygous for all three characters were allowed to self-fertilize, what proportion of the offspring would be expected to be as follows: (Note – use the rules of probability (and show your work) instead of huge Punnett squares)
a) homozygous for the three dominant traits
AATTRR = 1/4 x 1/4 x 1/4 = 1/64
b) homozygous for the three recessive traits
aattrr = 1/4 x 1/4 x 1/4 = 1/64
c) heterozygous (assumed for each trait)
AaTtRr = 1/2 x 1/2 x 1/2 = 1/8
d) homozygous for axial and tall, heterozygous for seed shape
AATTRr = 1/4 x 1/4 x 1/2 = 1/32
What is the best explanation for this genetic situation?
Black is dominant over white
Write genotypes for the parents, gametes, and offspring.
First cross:
| Parent’s genotypes | = | BB (black) | x | bb (white) |
| gametes | = | B | b | |
| F1 offspring | = | all Bb |
Second cross:
| Parent’s genotypes | = | Bb (black) | x | bb (white) |
| gametes | = | B or b | b | |
| F1 offspring | = | Bb or bb |
There should be 50% black to 50% white offspring in this cross.
a. 318 one-pod normal, 98 one-pod wrinkled
Parental genotypes: PPLl x PPLl or PpLl x PPLl
b. 323 three-pod normal, 106 three-pod wrinkled
Parental genotypes: ppLl x ppLl
c. 401 one-pod normal
Parental genotypes: PPLL x PpLL or PPLl x PPLL or PPLL x PpLl etc (nine possible genotypes).
d. 150 one-pod normal, 147 one-pod wrinkled, 51 three-pod normal, 48 three-pod wrinkled. (a 3: 3: 1: 1 ratio)
Parental genotypes: PpLl x Ppll (see below for details)
3 One-pod wrinkled (PPll , Ppll , Ppll)
1 Three-pod normal (ppLl)
1 Three-pod wrinked (ppll)
e. 223 one-pod normal, 72 one-pod wrinkled, 76 three-pod normal, 27 three-pod wrinkled (a 9: 3: 3: 1 ratio)
Parental genotypes: PpLl x PpLl
Father = AO (or IAi)
Mother = BO (or IBi)
First Child = OO (or ii)
What other genotypes and in what frequencies, would you expect in offspring from this marriage?
Examine the Punnett square to determine the other genotypes possible.
How many phenotypes are possible in a flock of ducks that contains all the possible combinations of these three alleles?
As in the ABO blood system 4 phenotypes are possible in this case:
| Genotype | Phenotype |
| HH, Hi | (H) |
| II, Ii | (I) |
| HI | (HI) |
| ii | (i) |
Under these circumstances assume the following Punnett square to be true.
a. all three of their children will be of normal phenotype
3/4 x 3/4 x 3/4 = 27/64
b. one or more of the three children will have the disease (x)
1 – 27/64 = 37/64
| All three have x | 2 out of 3 has x | 1 out of 3 has x | |
| + | + | = | |
| x x o | o o x | ||
| 3 Combinations | x o x | o x o | |
| o x x | x o o | ||
| + 3(3/4 x 1/4 x 1/4) | + 3(3/4 x 3/4 x 1/4) | = |
Note: the probability of the disease (x) = 1/4 & the probability of being normal (o) = 3/4
c. all three children will have the disease
1/4 x 1/4 x 1/4 = 1/64
d. at least one child out of three will be phenotypically normal
(Note: Remember that the probabilities of all possible outcomes always add up to 1)
1 – 1/64 = 63/64
a. aabbccdd = x x x = 1/256
b. AaBbCcDd = x x x = 1/16
c. AABBCCDD = x x x = 1/256
d. AaBBccDd = x x x = 1/64
e. AaBBCCdd = x x x = 1/128
Just remember that the probability of a heterozygote (Xx) = 2/4 or 1/2 and the probability of a homozygote XX or xx = 1/4
How would you determine whether the curl allele is dominant or recessive?
Mate the stray to a non-curl cat. If any offspring have the “curl” trait it is likely to be dominant. If the mutation is recessive, then on ly non-curl offspring will result.
How would you select for true-breeding cats?
You know that cats are true-breeding when curl crossed with curl matings produce only curl offspring.
How would you know they are true-breeding?
A pure-bred “curl cat” is homozygous.
a. AABBCC x aabbcc —-> AaBbCc
b. AABbCc x AaBbCc —–> AAbbCC
c. AaBbCc x AaBbCc —–> AaBbCc
d. aaBbCC x AABbcc —-> AaBbCc
2/3 x 2/3 x 1/4 = 1/9.
| Genotype | |
| Father | AO |
| Mother | BO |
OO expresses the disease
The second child’s chance of having the disease is = x = 1/16
Using the Punnett square below where P = normal pigmenation and p = white
Purple (i i P_ ) = 3
Red ( i i p p) = 1
The dominant allele I is epistatic to the p locus, and thus the F1 generation will be:
9 I_P_ : colorless
3 I_pp : colorless
3 i iP_ : purple
1 i i pp: red
Recessive
Fill in the genotypes of the individuals whose genotypes you know. What genotypes are possible for each of the other individuals?
If alkaptonuria is recessive George must be a carrier. See below.
If alkaptonuria is dominant Carla could not have the disease, as indicated in the pedigree chart, since the parents do not express the trait. See Below.
Because the daughter is normal the man’s genotype must be heterozygous for the trait so:
if X = extra digits and x = normal (5) digits then:
What is the probability that Charles and Elaine will have a baby with cystic fibrosis? (Neither Charles nor Elaine has the disease)
The probability that the baby (?) has the disease (if Elaine is a carrier) is 1/4
The total probability is 2/3 x 1/4 or 1/6.
B = Black – dominant
A = Agouti – dominant
BbAa x BbAa =
|
|
The phenotypic ratio is:
9 agouti: 4 white: 3 black
The allele is most likely dominant because the #2 individual (see below) with the trait marries a woman with the trait and 50% of their offspring are normal. If the trait were recessive one would expect the following:
| |
100% of offspring would have the disease, which is not the case. |
What genotypes are possible for the individuals marked 1, 2, and 3?
AP Biology PowerPoints
| Chapter 1 Introduction | Chapter 20 Biotechnology | Chapter 39 Plant Responses |
| Chapter 2 Biochemistry | Chapter 21 Genomes | Chapter 40 Animal Form & Structure |
| Chapter 3 Water | Chapter 22 Darwin Evolution | Chapter 41 Animal Nutrition |
| Chapter 4 Carbon Chemistry | Chapter 23 Population Evolution | Chapter 42 Circulation & Respiration |
| Chapter 5 Macromolecules | Chapter 24 Origin of Species | Chapter 43 Immune System |
| Chapter 6 The Cell | Chapter 25 Earth History | Chapter 44 Osmoregulation |
| Chapter 7 Cell Membranes | Chapter 26 Phylogeny | Chapter 45 Endocrine System |
| Chapter 8 Metabolism | Chapter 27 Archea & Bacteria | Chapter 46 Animal Reproduction |
| Chapter 9 Cellular Respiration | Chapter 28 Protists | Chapter 47 Animal Development |
| Chapter 10 Photosynthesis | Chapter 29 Plant Diversity I | Chapter 48 Neurons |
| Chapter 11 Cell Communication | Chapter 30 Plant Diversity II | Chapter 49 Nervous System |
| Chapter 12 Cell Cycle | Chapter 31 Fungi | Chapter 50 Senses |
| Chapter 13 Meiosis | Chapter 32 Introduction to Animals | Chapter 51 Behavior |
| Chapter 14 Mendel | Chapter 33 Invertebrates | Chapter 52 Ecology |
| Chapter 15 Chromosomes | Chapter 34 Vertebrates | Chapter 53 Population Ecology |
| Chapter 16 Molecular Inheritance | Chapter 35 Plant Structure | Chapter 54 Community Ecology |
| Chapter 17 From Gene to Protein | Chapter 36 Transport in Plants | Chapter 55 Ecosystems |
| Chapter 18 Gene Expression | Chapter 37 Plant Nutrition | Chapter 56 Conservation |
| Chapter 19 Viruses | Chapter 38 Angiosperms |
| ENZYME CATALASE |  |
Introduction:
Enzymes are proteins one of the four macromolecules: carbohydrates, proteins, lipids, and nucleic acids. They are produced by cells to act as a catalyst in chemical reactions. Enzymes are made by the ribosomes within a cell. The enzymes that need to stay within the cell are made by the free ribosomes in the cytoplasm. The enzymes that are made to be exported to other cells are created by the ribosomes on the Rough E.R. The purpose of enzymes is to lower the activation energy (the energy that is put in at the beginning of the reaction) of a reaction; therefore speeding the reaction rate up.
Catalase is one of the enzymes found in the cells of the human body. It is made up of five hundred amino acids in four polypeptide chains. It is found in the perioxisome microbody in the cells of eukaryotic organisms. The perioxisomes make hydrogen peroxide (H2O2). It is a byproduct of cellular respiration. This is very toxic to cells. That is why catalase is there. Catalase breaks down the hydrogen peroxide before it damages the cell. The formula for the reaction of decomposition of hydrogen peroxide by catalase is:
H2O2 H2O2 H2O + O2 H2O + O2
Substrates are the substances that attach to the enzyme. They attach at the enzyme’s active site with induced fit. The combining of and enzyme and substrate requires ATP. The reaction when an enzyme and a substrate join is reversible. The reaction can make more products or it can make more substrate.
Enzymes are affected by many things; such as salt concentration, pH, temperature, substrate concentration, product concentration, activators, and inhibitors. These things can cause the enzyme to denature (change shape). If the enzyme denatures, then the substrate isn’t able to attach to the active site of the enzyme.
The typical salt concentration for an enzyme is intermediate. This allows the substrate to attach to the enzyme correctly. If the concentration is lowered the R groups of the enzyme attract to each other and pull the enzyme into a different shape (changing the active site). If the concentration of salt is raised, then the active site of the enzyme is blocked. Therefore the substrate can’t attach to the enzyme.
Enzymes function best in an environment with a pH of seven. When the pH of the enzyme’s environment become more acidic (H+ ions increase) the enzyme denatures. This causes the active site to change its shape and the substrate can’t attach. The same thing happens when the enzyme’s environment gains H+ ions (becomes more basic).
When heat is added to anything the molecules increase is movement. With an enzyme, when ten degrees Celsius is added the reaction rate is doubled. This will continue until optimum temperature is reached. Then the enzyme is destroyed. Enzymes denature at approximately forty-fifty degrees Celsius.
Enzymes follow the Law of Mass Action. This determines the rate and direction of an enzyme and substrate reaction. If there is a high amount of substrate and a low amount of products the reaction will continue to make more products. If there is a low amount of substrate and a high amount of products, then the reaction will stop to get more substrate to continue the reaction. This is true except when products are immediately metabolized or exported from the cell.
Activators enable the substrate to fit the active site better. They increase the reaction. Inhibitors block the active site or cause the enzyme to denature. They slow down the reaction.
In this lab assaying is performed. An assay measures the amount of a substance that is left after a reaction. In this case, hydrogen peroxide.
Hypothesis:
The enzyme catalase will decompose H2O2 (hydrogen peroxide) best when its environment is ideal.
Materials:
The materials used in the first part of exercise 2A are 10mL of 1.5% H2O2, one 50mL beaker, and 1mL of fresh catalase. The materials used in the second part of exercise 2A are one test tube, 5mL of fresh catalase, a hot plate, 10mL of 1.5% H2O2, and one 50mL beaker. The materials used in the third part of exercise 2A are a potato, a knife, a ruler, one 50mL beaker, and 10mL of 1.5% H2O2. The materials used in exercise 2B are 10mL of 1.5% H2O2, 1mL of water, 10mL of H2SO4 (1.0 M), a stirring rod, two 50mL beakers, 5mL syringe, and 5mL of 2% KMnO4. The materials used in exercise 2C are a 50mL beaker, 10-15mL of 1.5% H2O2, 10mL syringe, and 5-10mL of 2% KMnO4. The materials used in exercise 2D are 60mL of 1.5% H2O2, 60mL of H2SO4 (1.0M), 6mL of fresh catalase, six 50mL beakers, three 10mL syringes, six 10mL beakers, and 30mL of 2% KMnO4.
Methods:
Exercise 2A: To observe the reaction of catalase and hydrogen peroxide, transfer 10mL of 1.5% H2O2 into a 50mL beaker and add 1mL of fresh catalase. Observe the reaction and record results on Table 1. Next, the effect of boiling catalase needs to be demonstrated. Place 5mL of fresh catalase into a test tube and boil it over a hot plate for 5 minutes. Allow the boiled catalase to cool very well. Then place 10mL of 1.5% H2O2 into a 50mL beaker and add 1mL of the cooled, boiled catalase. Observe the reaction and record results on Table 1. Finally, to see the presence of catalase in living tissue, cut 1 cm3 of potato and macerate it. Then, place it in a 50mL beaker with 10mL of 1.5% H2O2. Observe the reaction and record results on Table 1.
Exercise 2B: This is the procedure for establishing a baseline. First, put 10mL of 1.5% H2O2 in a beaker. Add 1mL of water (instead of catalase). Add 10mL of H2SO4 (1.0M). Mix the solution well. Remove a 5mL sample from the solution and place it into a 50mL beaker. Using the 5mL syringe, add 2% KMnO4 one drop at a time (swirling after each drop) to the sample until a persistent pink or brown color is obtained. Record the results on Table 2.
Exercise 2C: This will determine how much H2O2 will decompose spontaneously in an uncatalyzed reaction. Place 10-15mL of H2O2 in a 50mL beaker and store it uncovered for approximately 24 hours Determine the amount of H2O2 remaining after 24 hours by running a baseline test. Use a 10mL syringe to get 10mL of 2% KMnO4. Add the KMnO4 to the solution one drop at a time (swirling after each drop) until a pink or brown color is permanently obtained. Record the results in Table 3.
Exercise 2D: If a day or more has passed since exercise 2B has been performed, reestablish the baseline by performing exercise 2B again. Record the results in Table 4. This exercise will determine the amount of H2O2 is disappearing over 10, 30, 60, 120, 180, and 360 seconds. First, place 10mL of 1.5% H2O2 in six 50mL beakers. Label the beakers 10, 30, 60, 120, 180, and 360 seconds. Obtain 6mL of fresh catalase and 60mL of H2SO4 (1.0M). Add 1mL of catalase to the 10 second beaker with one syringe. At 10 seconds, add 10mL of H2SO4 (1.0M) with another syringe. For each of the times, repeat the adding of the catalase and H2SO4. Allow the reactions to proceed for 30, 60, 120, 180, and 360 seconds respectively. After the times have passed remove a 5mL sample from each of the six beakers and place each of the samples into a separate 10mL beaker. Use 5ml of 2% KMnO4 for each of the six beakers to determine the amount of H2O2 that is left after the reaction. Add KMnO4 to the samples one drop at a time (swirling after each drop) until a pink or brown color is permanently obtained. Record results in Table 5. Graph the results.
Results:
Table 1
Enzyme Activity
| Activity | Observations |
Enzyme Activity
|
Bubbles |
Effect of Extreme Temperature
|
Nothing |
Presence of Catalase
|
Bubbles |
Table 2
Establishing a Baseline
| Volume | |
Initial Reading
|
5mL |
Final Reading
|
1.6mL |
| Baseline (final volume – initial volume)
|
3.4mL |
Table 3
Rate of Hydrogen Peroxide Spontaneous Decomposition
| Volume | |
| Initial KMnO4
|
6mL |
Final KMnO4
|
1mL |
| Amount of KMnO4 used after 24 hours
|
5mL |
| Amount of H2O2 spontaneously decomposed
(ml baseline – ml after 24 hours) |
1.6mL |
Percent of H2O2 spontaneously decomposed(ml baseline – ml after 24 hours/ baseline) |
53% |
Table 4
Establishing a Second Baseline
| Volume | |
Initial Reading
|
5mL |
Final Reading
|
1mL |
| Baseline (final volume – initial volume)
|
4mL |
Table 5
Rate of Hydrogen Peroxide Decomposition by Catalase
|
Time ( Seconds) |
||||||
| 10 | 30 | 60 | 120 | 180 | 360 | |
| Baseline KMnO4 | 4mL | 4mL | 4mL | 4mL | 4mL | 4mL |
| Initial volume KMnO4
|
5mL | 5mL | 5mL | 5mL | 5mL | 5mL |
| Final volume KMnO4
|
2mL | 3mL | 4mL | 4.2mL | 4mL | 4.2mL |
| Amount KMnO4 used
(baseline – final) |
3mL | 2mL | 1mL | .8mL | 1mL | .8mL |
Amount H2O2 used(KMnO4 – initial)
|
1mL | 2mL | 3mL | 3.2mL | 3mL | 3.2mL |
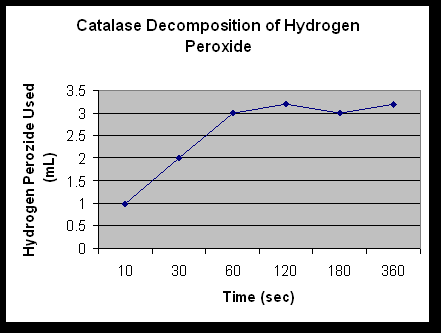
1. Determine the initial rate of the reaction and the rates between each of the time points.
0-10 – .1
10-30 – .05
30-60 – .03
60-120 – .003
120-180 – .003
180-360 – .001
2. When is the rate the highest? Explain.
They are the highest at the beginning of the reaction because; less hydrogen peroxide had been spontaneously decomposed. Therefore, there was more to be broken down.
3. When is the rate the lowest? For what reasons?
They are the lowest at the end of the reaction because more hydrogen peroxide has been decomposed.
4. Explain the inhibiting effect of sulfuric acid on the function of catalase. Relate this to enzyme structure and chemistry.
The acid lowered the pH and the change made catalase gain H+ ions which caused the active site to change shape.
5. Predict the effect lowering the temperature would have on the rate of enzyme activity. Explain.
It would decrease the reaction because changing the temperature causes the enzyme to denature. This causes the active site to change place, which slows down the reaction.
6. Design a control experiment to test the effect of varying pH, temperature, or enzyme concentration.
Label 3 test tubes 0oC, rt, and 100oC. Place an equal amount of enzyme in each tube. Put the 0oC on ice, leave the rt out and boil the 100oC tube. Observe the changes in all 3 tubes.
Error Analysis:
Some possible errors that could have occurred in this lab include taking inaccurate measurements of the substances used, making wrong calculations, and the times of testing the experiments could have been delayed (ex: instead of 60 seconds it took place at 65 seconds).
Conclusion:
The enzyme catalase is created to break down H2O2. If the environment of catalase is changed in any way; such as pH and temperature; then it can not do its job of breaking down H2O2. This is proven when catalase broke down H2O2 in 2A and produced water and oxygen when it wasn’t boiled. When the catalase was boiled, it had been denatured; therefore it wouldn’t break the H2O2 down. In 2D the sulfuric acid kept catalase from breaking down H2O2 also. It lowered the pH of the solution so the catalase denatured. You can tell by looking at Table 5. The lower times have less H2O2 used than the higher times. The higher times had more time with out the H2SO4, so the catalase was able to break down more H2O2.
The rates of the reaction in this lab were very high at the beginning. Then, they started to slow down. This is because at the beginning of the reaction there is more H2O2 to decompose. Towards the end of the reaction there is less to decompose, so the rate slows down quite suddenly.