Category: 1st Semester
Evolution PPT Questions
| Evolution ppt Questions |
History of Evolutionary Thought 
1. What were Aristotle’s early ideas about life on Earth?
2. How long did these ideas last?
3. What was Linnaeus first to do?
4. What language is used for scientific naming?
5. What are the two words called in a scientific name?
6. This naming system is known as ____________ ______________.
7. Name the contribution that each of these men made to Darwin’s ideas on evolution:
a. Charles Lyell
b. George Cuvier
c. Thomas Malthus
d. James Hutton
e. Lamarck
f. Wallace
8. Which was published first – the Origin 0f Species by Darwin or Gregor Mendel’s papers on inheritance?
9. What was the name of George Cuvier’s theory on evolution?
10. What did Cuvier study in Paris and what did he find?
11. What did Cuvier decide was responsible for the disappearance of some species?
12. James ___________ was a Scottish _________ who studied fossils of _____________ in the Paris Museum.
13. Hutton’s ideas were known as _____________.
14. Briefly state Hutton’s idea on geological change.
15. Lyell proposed the theory of _________________.
16. Describe uniformitarianism.
17. How old did Lyell propose that the Earth was? How old did most people at this time believe the Earth was?
18. How did reading Lyell’s book help darwin on his voyage on the Beagle?
19. Lamarck was one of the first scientists to understand that change occurs over ___________.
20. Lamarck believed that changes were adaptations to the ____________ that organisms _____________ in their lifetime and that he thought could be passed on to _______________.
21. Explain Lamarck’s idea of the Law of Use and Disuse.
22. Lamarck’s theory of acquiring or losing traits by using or not using them led to his theory of evolution called the _____________ of ______________ _____________.
23. According to this theory new ___________ could arise over time.
24. According to Lamarck, if a blacksmith built up his muscles then he would have what type of sons?
25. According to Lamarck, if a giraffe stretched its neck reaching for leaves, what would its offspring look like?
26. What did Lamarck NOT know that made his theory incorrect?
27. Are genes changed by life activities?
Darwin the Naturalist 
28. In what year and at what age did Darwin become the naturalist for the ship the HMS Beagle?
29. How long was the Beagle voyage around the world?
30. As Darwin sailed around the coast of __________ __________, he collected many different types of plants and animals on the mainland and on the islands.
31. Where are the Galapagos Islands and how were they formed?
32. What did Darwin discover about the animals on each type of island
33. How did the island species of finches and tortoises compare with those on the islands?
34. How did the necks of the tortoises compare with each other?
35. The island finches resembled a finch on the ___________.
36. Was the available food and habitat the same on all the islands? Explain.
37. What was different about the finches and why?
Darwin’s Observations & Conclusions 
38. List three observations Darwin made on his travels that led him to propose his revolutionary idea about the way life changes over time.
a.
b.
c.
39. Give an example of the uneven distribution of species noted by darwin.
40. Darwin collected both ___________ organisms and ____________ of organisms.
41. Give 2 examples of fossils collected by Darwin in which the species were no longer in existence.
42. Give a definition for evolution.
43. Left unchecked, what did Darwin predict would happen to the number od individuals in a population?
44. In nature, what tends to happen to the size of populations over time?
45. Competition among members of a population occur due to a limited number of ____________ _______________.
46. Only a ___________ of the offspring produced survive to the next generation.
47. The struggle for environmental resources is commonly called _____________ of the ____________.
48. How do individuals in population compare with each other?
49. Variation in a population is ______________.
50. Which organisms in a population are most likely to live offspring to pass on their traits?
51. This process is known as _____________ ___________ and was proposed by Charles ___________.
52. State Darwin’s theory of natural selection.
53. New ____________ evolve according to natural selection.
Ideas that Shaped Darwin’s Thinking 
54. _____________ was an economist in 1798 that influenced Darwin’s thinking.
55. Malthus observed what about the birth rate of babies?
56. Malthus knew population size was limited by what?
57. According to Malthus, a high birth rate and limited resources caused what to happen?
58. List several things that organisms struggle for in the environment.
59. What did Malthus say would happen if the population size continued to groww unchecked?
60. The __________ rate should increase to balance the __________ of a population and the limited _____________ in the environment.
61. Did Darwin see this occurring in nature?
62. Most organisms produce ____________ offspring than can survive causing many to ________.
Darwin’s Theory of Evolution 
63. Darwin proposed that organisms descended from what?
64. Over time, according to Darwin organisms __________ their form causing evolution of new ____________.
65. ___________ __________ is the driving force for evolution.
66. During the struggle for survival, which organisms survive to pass on their traits?
Origin of Species 
67.How long after he returned to England did Darwin publish his book about evolution?
68. Why did Darwin wait so long to publish his ideas?
69. Darwin’s theory of evolution challenged both the ____________ and _____________ ideas at that time.
70. What made Darwin publish his book?
71. _______________ independently developed the same theory as Darwin.
72. Both Darwin and Wallace believed that __________ changed over time due to a _____________ for existence.
73. Both Darwin’s and Wallace’s papers were presented to the ____________ ______________ in July of __________.
74. How long after this did it take Darwin to finish writing his book?
75. Before Darwin, it was thought that species were perfectly made and _______________.
76. What group of people had been observing and using variations in organisms for a long time?
77. How were farmers using variation?
78. This process is called _____________ ______________ instead of natural selection that occurs in nature.
79. Artificial selection involves ____________ desired traits in stock or crops and __________ them to pass on the trait.
Controversy
80. Define these terms:
a. struggle for existence
b. survival of the fittest
c. descent with modification
d. Fitness
e. adaptation
81.What are the two types of adaptations?
82. Give some examples of physical adaptations.
83. Give some examples of behavioral adaptations.
84. What happens to organisms with LOW fitness?
85. How did changes in the Galapagos finches make them more FIT to survive?
86. Natural selection takes place over a _________ period of time.
87. Natural selection can be observed as changes in _______ structure, ecological _________, and ____________.
88. Do species today look them same as their ancestors?
89. Living species descended with changes from other __________ over periods of time.
90. What was a major problem in Darwin’s Theory?
91. The work of what scientists solved the problem of how variations were passed to offspring?
92. What is the complete title of Darwin’s book?
Theory of Evolution Today
93. List three main things used today to show how organisms are related.
a.
b.
c.
94. Give two examples of evolution that has occurred today in a much shorter period of time.
a.
b.
95. Define macroevolution.
96. Define microevolution.
97. Darwin argued that Earth was ____________ of years old instead of thousands of years old.
98. One of the main pieces of evidence to support this ancient age of the Earth came from ___________ collected by Darwin.
99. Fossils are found in what type of rock layers?
100. Animals on different continents living in similar habitats show similar _______________.
101. All ____________ have similar bon structures known as ______________ structures.
102. Homologous structures have the same structure but different ______________.
103. Give 3 examples of homologous structures in vertebrates.
104. __________ structures seem to have no important function.
105. Give an example of a vestigial structure in humans.
106. What is an embryo?
107. How does the embryonic development of different vertebrates compare to each other?
Enzyme Catalysis
Enzyme Catalysis
Introduction:
In general, enzymes are proteins produced by living cells, they act as catalysts in biochemical reactions. A catalyst affects the rate of a chemical reaction. One consequence of enzyme activity is that cells can carry out complex chemical activities at relative low temperatures. In an enzyme-catalyzed reaction, the substance to be acted upon ( the substrate = S ) binds reversibly to the active site of the enzyme (E). One result of this temporary union is a reduction in the energy required to activate the reaction of the substrate molecule so that the products (P) of the reaction are formed.
In summary: E + S —> ES –> E + P
Note that the enzyme is not changed in the reaction and can even be recycled to break down additional substrate molecules. Each enzyme is specific for a particular reaction because its amino acid sequence is unique and causes it top have a unique three-dimensional structure. The active site is the portion of the enzyme that interacts with the substrate, so that any substance that blocks or changes the shape of the active site affects the activity of the enzyme. A description of several ways enzyme action may be affected follows:
1. Salt Concentration. If the salt concentration is close to zero, the charged amino acid side chains of the enzyme molecules will attract to each other. The enzyme will denature and form an inactive precipitate. If, on the other hand, the salt concentration is too high, normal interaction of charged groups will be blocked, new interactions will occur, and again the enzyme will precipitate. An intermediate salt concentration such as that of human blood (0.9% ) or cytoplasm ins the optimum for many enzymes.
2. pH. Amino acid side chains contain groups such as – COOH and NH2 that readily gain or lose H+ ions. As the pH is lowered an enzyme will tend to gain H+ ions, and eventually enough side chains will be affected so the enzyme’s shape is disrupted. Likewise, as the pH is raised, the enzymes will lose H+ ions and eventually lose its active shape. Many of the enzymes function properly in the neutral pH range and are denatured at either an extremely high or low pH. Some enzymes, such as pepsin, which acts in the human stomach where the pH is very low, have a low pH optimum.
3. Temperature. Generally, chemical reactions speed up as the temperature is raised. As the temperature increases, more of the reacting molecules have enough kinetic energy to undergo the reaction. Since enzymes are catalysts for chemical reactions, enzyme reactions also tend to go faster with increase temperature. However, if the temperature of an enzyme-catalyzed reaction is raised still further, a temperature optimum is reached; above this value the kinetic energy of the enzyme and water molecules is so great that the conformation of the enzyme molecules is disrupted. The positive effect of speeding up the reaction is now more than offset by the negative effect of changing the conformation of more and more enzyme molecules. Many proteins are denatured by temperatures around 40-50 degrees C, but some are still active at 70-80 degrees C, and a few even withstand boiling.
4. Activation’s and Inhibitors. Many molecules other than the substrate may interact with an enzyme. If such a molecule increases the rate of the reaction it is an activator, or if it decreases the reaction rate it is an inhibitor. These molecules can regulate how fast the enzymes acts. Any substance that tends to unfold the enzyme, such as an organic solvent or detergent, will act as an inhibitor. Some inhibitors act by reducing the -S-S- bridges that stabilize the enzyme’s structure. Many inhibitors act by reacting with the side chains in or near the active site to change its shape or block it. Many well known poisons such as potassium-cyanide and curare are enzyme inhibitors that interfere with the active site of critical enzymes.
The enzyme used in this lab, catalase, has four polypeptide chains, each composed of more than 500 amino acids. This enzyme is ubiquitous in aerobic organisms. One function of catalase within cells is to prevent the accumulation of toxic levels of hydrogen peroxide formed as a by-product of metabolic processes. Catalase might also take part in some of the many oxidation reactions that occur in the cell.
2H2O ——-> 2 H2O + O2 (gas )
In the absence of catalase, this reaction occurs spontaneously, but very slowly. Catalase speeds up the reaction considerably. In this experiment, a rate for this reaction will be determined. Much can be learned about enzymes by studying the kinetics of enzyme-catalyzed reactions. For example, it is possible to measure the amount of product formed, or the amount of substrate used, from the moment the reactants are brought together until the reaction has stopped. If the amount of product formed is measured at regular intervals and this quantity is plotted on a graph, a curve like the one that follows is obtained.
Figure 2.1 Enzyme Activity

Study the solid line of the graph of this reaction. At time 0 there is no product. As time progresses the production of product increases at a steady rate. After a period of time this rate slows down and at a certain point the reaction rate is very slow.
General Procedure:
In this experiment the disappearance of the substrate, H2O2, is measured as follows:
1. A purified catalase extract is mixed with substrate ( H2O2) in a beaker. The enzyme catalyzes the conversion of H2O to H2O and O2 (gas ).
2. Before all the H2O2 is converted to H2O and O2 , the reaction is stopped by adding sulfuric acid ( H2SO4 ). The sulfuric acid lowers the pH, denatures the enzyme, and thereby stops the enzyme’s catalytic activity.
3. After the reaction is stopped, the amount of substrate (H2O2) remaining in the beaker is measured. To measure this quantity, potassium permanganate is used. Potassium permanganate (KMnO4), in the presence of H2O2 and H2SO4 reacts as follows:
5 H2O2 + 2 KMnO4 + 3 H2SO4 ————–> K2SO4 + 2 MnSO4 + 8 H2O + 5 O2
Note that H2O2 is a reactant for this reaction. Once all the H2O2 has reacted, any more KMnO4 added will be in excess and will not be decomposed. The addition of excess KMnO4 causes the solution to have a permanent pink or brown color. Therefore, the amount of H2O2 remaining is determined by adding KMnO4, until the whole mixture stays a faint pink or brown, permanently. Add no more KMnO4 after this point.
Figure 2.2 The General Procedure for the above exercise and Exercise 2C.
The figure below represents the complete Exercise 2C.

Exercise 2A: Test of Catalase Activity:
1. To observe the reaction to be studied, transfer 10 mL of 1.5% (0.44M) H2O2 into a 50 ml glass beaker and add 1 mL of freshly made catalase solution. The bubbles coming from the reaction mixture are oxygen, which results from the breakdown of H2O2 by catalase. Be sure to keep the freshly made catalase solution on ice at all times.
a. what is the enzyme in this reaction? ____________________________________________________
b. What is the substrate in this reaction? ___________________________________________________
c. What is the product in this reaction? ____________________________________________________
d. How could you show that the gas evolved is oxygen ? _____________________________________
2. To demonstrate the effect of boiling on enzymatic activity, transfer 5 mL of purified catalase extract to a test tube and place it in a boiling water bath for five minutes. Transfer 10 mL of 1.5% H2O2 into a 50 mL glass beaker and add 1 mL of the cooked, boiled catalase solution. How does the reaction compare to the one using the unboiled catalase? Explain the reason for this difference.
_____________________________________________________________________
_____________________________________________________________________
3. To demonstrate the presence of catalase in living tissue, cut 1 cm3 of potato or liver, macerate it, and transfer it into a 50 mL beaker containing 1.5% H2O2 . What do you observe? What do you think would happen if the potato or liver was boiled before being added to the H2O2?
_____________________________________________________________________
_____________________________________________________________________
Exercise 2B: The Baseline Assay:
To determine the amount of H2O2 initially present in a 1.5% solution, one needs to perform all the steps of the procedure without adding catalase to the reaction mixture. This amount is known as the baseline and is an index of the initial concentration of H2O2 un solution. In any series of experiments, a baseline should be established first.
Procedure for Establishing Baseline:
1. Put 10 mL of 1.5% H2O2 into a clean glass beaker.
2. Add 1 mL of H2O ( instead of enzyme solution).
3. Add 10 mL of H2SO4 (1.0 M) Use Extreme care in Handling Acids.
4. Mix well.
5. Remove a 5 mL sample. Place this 5 mL sample in another beaker, and assay for the amount of H2O2 as follows: Place the beaker containing the sample over white paper. Use a burette or 5 mL pipette to add potassium permanganate a drop at a time to the solution until a persistent pink or brown color is obtained. Remember to gently swirl the solution after adding each drop. Record your data below.
Baseline calculations
Final Reading of burette ________ mL
Initial reading of burette ________mL
Baseline (Final -Initial) _________mL KMnO4
Figure 2.4: Proper Reading of a Burette
Exercise 2C: An Enzyme-Catalyzed Rate of H2O2 Decomposition
Refer to figure 2.2 to complete this section and record the data in Table 2.1 below.
Table 2.1
| Potassium Permanganate (ml) |
Time (Seconds) |
|||||
| 10 | 30 | 60 | 120 | 180 | 360 | |
| A. Baseline | ||||||
| B. Final Reading | ||||||
| C. Initial Reading | ||||||
| D. Amount of KMnO4 consumed (B-C) | ||||||
| E. Amount of H2O2 used (A-D) | ||||||
Graph the data for enzyme-catalyzed H2O2 decomposition.
Graph Title: ___________________________________________________________________
Graph 2.1

Analysis of Results:
1. Explain the inhibiting effect of sulfuric acid on the function of catalase. Relate this to enzyme structure and chemistry.
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
2. Predict the effect lowering the temperature would have on the rate of enzyme activity. Explain you prediction.
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
3. Design a controlled experiment to test the effect of varying pH, temperature, or enzyme concentration.
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
____________________________________________________________________
| AP LAB PAGE |
Enzyme PowerPoint Worksheet
Enzymes
ppt Questions
Enzyme Structure & Function
1. Most enzymes are what type of macromolecule?
2. Most enzymes are ______________ or ______________ structures.
3. Enzymes act as ___________ in reactions.
4. Are enzymes permanently changed in the chemical reactions they are involved in?
5. Will an enzyme work on any substance? Explain.
6. Can enzymes be reused?
7. What ending is found on many enzymes?
8. Give 3 examples of enzymes with this ending.
9. How does an enzyme work?
10. What effect does an enzyme have on activation energy needed to start a reaction?
11. Hydrogen peroxide H2O2 is a common waste product of cells. Enzymes called catalases in cells break this down into harmless ________________.
12. What is meant by the term substrate?
13. What is meant by active site?
14. Sketch and label the enzyme-substrate complex.
15. What is meant by induced fit?
16. What induces an enzyme to change the shape of its active site?
17. List 4 factors that can affect enzyme activity.
18. What is the effect of high temperature on an enzyme (running fever)?
19. What temperature do most enzymes do best at?
20. Most enzymes like a pH near ______________.
21. To denature an enzyme means the enzyme becomes _______________ and can no longer work properly.
22. Name 3 inorganic substances (cofactors) that are often needed for enzymes to work properly.
23. Give an example of an enzyme & its needed inorganic substance.
24. Give one example of an enzyme inhibitor.
25. Explain how competitive inhibitors work.
26. If a competitive inhibitor blocks the active site, the ____________ can’t fit.
27. Explain noncompetitive inhibitors.
28. Do noncompetitive inhibitors bind to the active site? Explain.
Ecology
Ecology
All Materials © Cmassengale
Ecology is the study of interactions between organisms (biotic part) and their nonliving environment (abiotic factors)
Biotic factors includes plants, animals, fungi, & microorganisms. They may be producers, consumers, or decomposers.
Abiotic factors include climate, soil, temperature, water, air, sunlight, humidity, pH, and atmospheric gases.
Habitat is the place a plant or animal lives, while its niche is its total way of life.
Life is organized into levels:
Organism (any single living thing)
¯
Population (members of the same species living in one place)
¯
Community (all the populations living in an area)
¯
Ecosystem (community living in a similar habitat such as a forest)
¯
Biomes (ecosystems covering wide areas & with similar climates & organisms)
¯
Biosphere ( all the living & nonliving things on earth)
Producers:
| Make their own food through photosynthesis or chemosynthesis | |
| Includes plants, algal protists, & some bacteria |
Consumers:
| Can’t make their own food | |
| May be herbivores (feed only on plants), carnivores (feed only on animals), or omnivores (feed on plants & animals) |
Decomposers:
| Break down dead plants & animals (detritus) | |
| Recycle nutrients | |
| Called detritivores | |
| Include fungi & bacteria |
Sunlight is the ultimate energy for all life on earth, but only producers can get their energy directly from the sun.

Trophic levels are feeding levels of producers & consumers in an ecosystem:
| 1st Trophic Level is producers that use sunlight directly | |
| 2nd Trophic Level includes herbivores that feed directly on plants | |
| Higher Trophic Levels are carnivores feeding on each other |
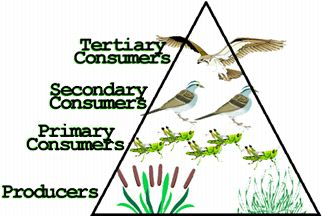
Food chains & food webs:
| Chains show who eats whom in an ecosystem. | |
| Webs are made up of several food chains. | |
| Always begin with producers absorbing sunlight. | |
| Producers store energy in the chemical bonds of the food they make. | |
| Stored energy is passed to consumers when they eat producers or other consumers. | |
| Some energy is lost at each trophic level as heat when consumers “burn” food during cellular respiration. | |
| Both energy & nutrients must move through an ecosystem. |
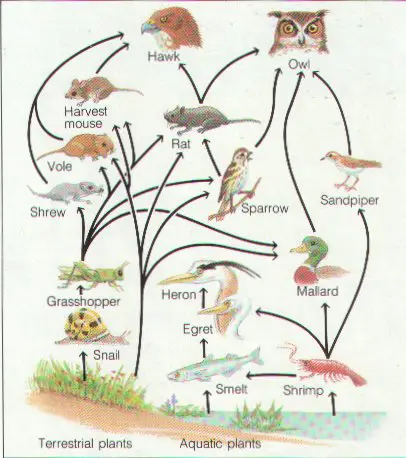
Three main elements that must move through an ecosystem:
| Water | |
| Carbon | |
| Nitrogen |
Water or Hydrologic Cycle:
| Cells are 70 – 90% water | |
| Water is needed for metabolic processes | |
| Water is most important for terrestrial organisms because of desiccation (drying out) |
Steps in the water Cycle:
Evaporation Transpiration
(water loss from lakes, rivers, oceans…) (water loss from plant leaves)
¯ ¯
Condensation
(water vapor forms clouds)
¯
Precipitation
(water returns to earth as sleet, rain, snow…)
¯
Surface Runoff
(returns water to bodies of water or to groundwater)
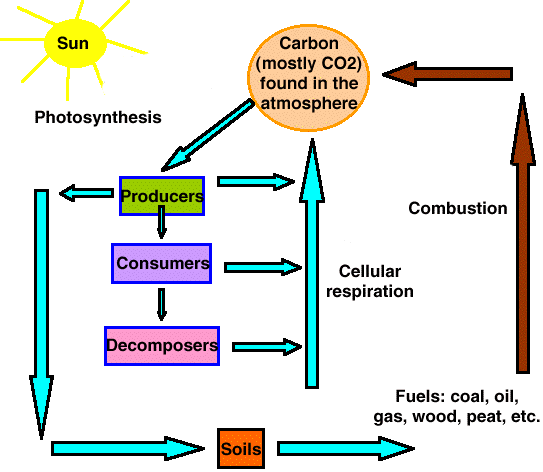
Carbon Cycle:
| Consists of photosynthesis, cellular respiration, & decomposition | |
| Begins with producers taking carbon dioxide from the air during photosynthesis | |
| Carbon dioxide used in cellular respiration | |
| Decomposing plants and animals return Carbon to the soil |
Carbon Cycle Steps:
Plant leaves take carbon dioxide from air
¯
Plants store carbon in carbohydrates or starches
(photosynthesis)
¯
Plants & animals release carbon dioxide back into the air
(cellular respiration)
¯
Decomposers return carbon to environment
(decomposition)
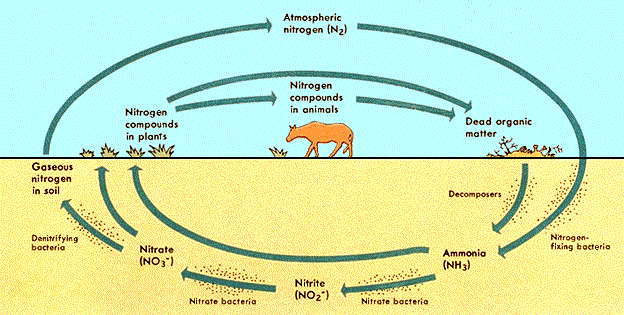
Nitrogen:
| Needed by all organisms | |
| Used to make proteins & nucleic acids (DNA & RNA) | |
| Air made up of 80% nitrogen | |
| Only Cyanobacteria & Rhizobium bacteria can use nitrogen directly from the air (nitrogen fixation) | |
| Bacteria found in the soil & on the roots of legumes (beans, peas …) |
Steps in the Nitrogen Cycle:
Cyanobacteria & Rhizobium take nitrogen from air
(nitrogen fixation)
¯
Convert nitrogen gas into ammonia
¯
Nitrifying bacteria in soil change ammonia into nitrates
¯
Plants can absorb & use nitrates to make proteins
¯
Consumers eat plants & get proteins containing nitrogen
¯
Decomposers break down dead organisms & return nitrogen to air
(called ammonification)
¯
Anaerobic bacteria in soil release nitrogen from nitrates into air
(called denitrification)
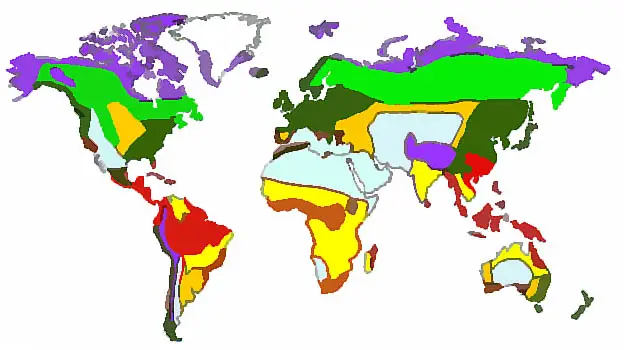
Three main types of ecosystems:
| Terrestrial (land) | |
| Freshwater (rivers, ponds, lakes …) | |
| Marine (oceans & seas) |
Terrestrial ecosystems are divided into 7 biomes with similar climates & organisms
Seven Terrestrial Biomes:
| Tropical Rain Forest (jungle) | |
| Savanna (tropical grasslands) | |
| Deserts | |
| Grasslands | |
| Deciduous Forest | |
| Taiga (coniferous forest) | |
| Tundra |

Tundra:
| Cold & dark most of the year | |
| Includes the arctic | |
| Permafrost is the top layer of soil that thaws & in which plants grow | |
| No trees, but sedges & grass, mosses, & lichens | |
| Many migratory animals | |
| Lemmings & ptarmigans are year round residents | |
| Approximately 20 cm annual rainfall |

Taiga:
- Coniferous forest
- Extends across northern Eurasia & North America
- Contains conifers or evergreens (spruce, cedar, fir, pine …)
- Needle like leaves withstand weight of snow
- Bear, deer, moose, wolves, mountain lions …
- Sequoia or redwood (largest conifer) grows here
- Bristle cone pine oldest living conifer found here

Temperate Deciduous Forest:
- South of taiga in North America, eastern Asia, & Europe
- High annual rainfall (75-150 cm)
- Moderate temperatures
- Well-defined seasons of about equal length
- Trees loose leaves in winter (deciduous)
- Show stratification (plant layers):
1. Canopy – broad leaf deciduous trees forming uppermost layer
2. Under story – shrubs
3. Forest Floor – herbaceous plants - Songbirds, deer, rabbits, foxes, squirrels, frogs 7 toads, lizards …

Tropical Rain forest:
- Near equator
- Warm climate (20 -25 degrees C)
- Plentiful rainfall (190 cm/year)
- Contains the greatest diversity of plants & animals
- Insects, monkeys & apes, snakes, tropical birds, leopards…
- Animals & plants brightly colored
- Poor soil for agriculture

Grasslands:
- Mostly grasses with a few trees due to less rainfall
- Moderate climates
- Good for agricultural crops
- Grazing & burrowing animals dominate
- Also called prairies

Savanna:
- Tropical grasslands
- Warm climate & rainy season
- Antelope, zebra, lions, wildebeests, hyenas, elephants…
- Suffer from floods & drought

Deserts:
- Low annual rainfall
- Subject to strong winds
- Days usually hot & nights cold
- Sahara desert is without vegetation
- Succulents such as cacti & other water storing plants
- Most animals nocturnal
- Lizards, snakes, roadrunners, insects, tarantula, hawks, rodents, coyotes…

Aquatic Biomes:
- May be freshwater or saltwater
- Wetlands near oceans have brackish water (mixture of fresh & salt waters)
- Part of the part water or hydrologic cycle
- Often polluted by man’s activities
Lakes & Rivers:
- Freshwater
- Oligotrophic lakes are nutrient poor (catfish, carp…)
- Eutrophic lake are nutrient rich (trout, bass…)
- Deep lakes have layers or strata where different plants & animals live
- Phototropic organisms in upper layers for light
- Estuary at mouth of river contains brackish water
Ocean Zones:
- Intertidal zone
1. Along shoreline
2. Wave action
3. Lots of light so many producers
4. Starfish, sand dollars… - Neritic Zone
1. Ocean water above continental shelf
2. Coral reef found here
3. Surrounds continents & receives light in upper layers - Oceanic Zone
1. Beyond continental shelf
2. Deepest area (up to 7 miles)
3. Bottom doesn’t receive light so animals adapted to darkness (many produce their own light, feed on other animals…)
4. Deepest area called abyss
5. Upper area gets light & called the photic zone (lots of seaweed here)
6. Floaters called plankton (microscopic organisms)
7. Swimmers such as fish called nekton
8. Bottom dwellers called benthos